Preeclampsia is a condition related to pregnancy and is characterised by new-onset hypertension after 20 weeks of gestation [1]. The global incidence is estimated to be between 2% and 10%, while studies in India have reported a prevalence of 8% to 10% [2-4]. A typical diagnosis includes blood pressure of 140 mmHg or higher (systolic) or blood pressure of 90 mmHg or higher (diastolic) measured on two occasions at least four hours apart [1,5,6]. In addition to hypertension, proteinuria is a standard biomarker of preeclampsia [7-9]. The leakage of protein in urine is attributed to damage in the glomeruli as a result of hypertension [10]. Therefore, the presence of proteinuria alongside hypertension is a strong indication of preeclampsia. Identification and early management of preeclampsia are crucial for maternal and fetal outcomes.
The UPCR has been widely used as an alternative to the 24-hour urine collection, offering the advantage of being quicker, more convenient and readily available [11-13]. By calculating the ratio of protein to creatinine in a single urine sample, UPCR can efficiently identify proteinuria without the need for prolonged urine collection. Additionally, urine protein dipsticks are extensively used for initial screening, providing a rapid, though less precise, estimate of proteinuria [14,15]. Given the life-threatening nature of preeclampsia, which can result in end-organ failure and severe complications for both the mother and foetus, the development of reliable, accurate and time-efficient diagnostic tools like UPCR and urine dipsticks is essential for improving outcomes through early detection and timely intervention [16,17]. However, there is a dearth of knowledge regarding the reliability and specificity of these time-saving diagnostic tools to ensure timely and appropriate management.
Here, the authors aimed to contribute to the existing literature by evaluating the diagnostic performance of the urine dipstick test and the UPCR against the 24-hour urine protein test.
Materials and Methods
A cross-sectional study was conducted at the Department of Obstetrics and Gynaecology at Dr. D.Y. Patil Medical College and Research Centre, Pune, Maharashtra, India, from October 2023 to October 2024. The Institutional Ethics Committee approved the study (IESC/FP/2023/54) and written informed consent was obtained from all participants.
Sample size calculation: A total of 80 patients were included in the study. The sample size was calculated using Fisher’s formula: n=Z2×P (1-P)/e2, where Z=1.96 for a 95% confidence interval, P=0.05 (prevalence of preeclampsia) [18,19] and e=0.05 (precision). With a 5% attrition rate, the sample was calculated to be 77.
Inclusion criteria: The study included hypertensive pregnant women with blood pressure levels of ≥140/90 mmHg, who had a singleton pregnancy and were beyond 20 weeks of gestation.
Exclusion criteria: Women with known renal diseases, urinary tract infections, dehydration, chronic hypertension, diabetes mellitus, molar pregnancy, or multiple pregnancies were excluded. Additionally, patients who delivered before the collection of the 24-hour urine sample were also excluded. These conditions were excluded due to their potential to alter proteinuria levels, which could confound the results and affect the outcomes of the study.
Study Procedure
Laboratory analysis: After obtaining informed consent, demographic details and obstetric parameters, including age and gravidity, were recorded. A random spot urine sample for dipstick and UPCR testing was taken from each participant, followed by the collection of 24-hour urine output for the estimation of total protein.
Urine dipstick test: The urine sample was tested using commercially available urine dipsticks (Siemens Uristix reagent strips). The dipstick results were interpreted as negative, trace, 1+, 2+, or 3+ and values of ≥1+ were considered positive for proteinuria.
Urine Protein-Creatinine Ratio (UPCR): The urine sample was analysed for the estimation of protein and creatinine concentrations. The Urinary Protein-to-Creatinine Ratio (UPCR) was calculated as (urinary protein/urinary creatinine), with a threshold value of ≥0.3 considered positive for significant proteinuria [20]. Protein was measured using the pyragallol method and creatinine levels were determined using the Jaffe method [21].
24-Hour Urine Protein Test (Gold Standard): Urine output over 24 hours was collected in a sterile container and analysed for the estimation of urine protein. Total protein excretion was measured using an automated biochemical analyser, with ≥300 mg/day considered indicative of proteinuria.
Statistical Analysis
Data were analysed using GraphPad Prism 10. Sensitivity, specificity, PPV, NPV, accuracy and F1-score were calculated for both the urine dipstick and UPCR, using the 24-hour urine protein test as the reference standard. Categorical variables were expressed as frequencies and percentages.
Results
The study population consisted of 80 hypertensive patients, of whom 35 (44%) had preeclampsia and 45 (56%) had gestational hypertension. The age distribution of the study population showed that the majority of cases were in the 20-25 years age group 38 (48%). The least represented age group was 18-20 years 2 (3%) [Table/Fig-1]. The majority of the patients with preeclampsia were within the age group of 25-30 years [Table/Fig-2].
Distribution of population based on age.
| Age group (in years) | n (%) |
|---|
| 18-20 | 2 (2.5) |
| 20-25 | 38 (47.5) |
| 25-30 | 21 (26.25) |
| 30-35 | 15 (18.75) |
| 35-40 | 4 (5) |
| Total | 80 (100) |
Distribution of patients with preeclampsia based on age.
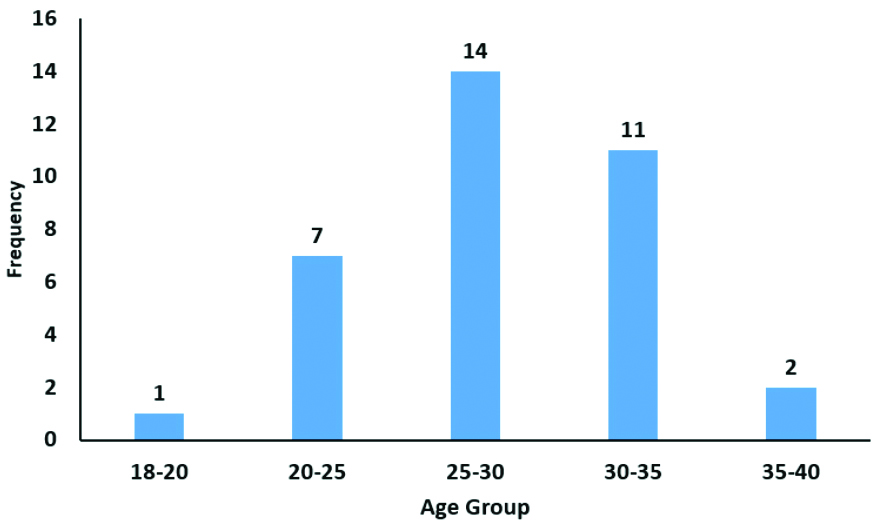
The highest proportion of participants had no prior pregnancies 25 (31%), followed by those with a gravidity of 2 24 (30%) [Table/Fig-3].
Distribution of population based on gravidity.
| Gravidity | n (%) |
|---|
| 0 | 25 (31.25) |
| 1 | 2 (2.5) |
| 2 | 24 (30) |
| 3 | 14 (17.5) |
| >3 | 15 (18.75) |
| Total | 80 (100) |
The diagnostic performance of the urine dipstick test was evaluated against the 24-hour urine protein test (the gold standard) in patients. Among the 80 patients, the urine dipstick test was positive in 42 cases, of which 15 (36%) were confirmed positive by the 24-hour urine protein test, while 27 (64%) were false positives [Table/Fig-4].
Diagnostic performance of urine dipstick test for preeclampsia.
| Preeclampsia |
|---|
| Urine dipstick | Positive | Negative | Total |
|---|
| Positive | 15 | 27 | 42 |
| Negative | 20 | 18 | 38 |
| Total | 35 | 45 | 80 |
Furthermore, the diagnostic performance of the urine UPCR was evaluated against the 24-hour urine protein test in patients. Among the 80 patients, UPCR was positive in 74 cases, of which 32 (43%) were confirmed positive by the 24-hour urine protein test, while 42 (57%) were false positives [Table/Fig-5].
Diagnostic performance of UPCR for preeclampsia.
| Preeclampsia |
|---|
| Urine Protein-Creatinine Ratio (UPCR) | Positive | Negative | Total |
|---|
| Positive | 32 | 42 | 74 |
| Negative | 3 | 3 | 6 |
| Total | 35 | 45 | 80 |
The sensitivity of the dipstick was 43%, while UPCR had a sensitivity of 91%. The PPV for the dipstick was 36%, while UPCR showed a higher PPV of 43%, demonstrating its better ability to confirm positive cases. The NPV was low for the dipstick, with values of 47% for the dipstick and 50% for UPCR. The F1-score, which reflects the balance between precision and recall, was 39% for the dipstick and 59% for UPCR [Table/Fig-6].
Comparison of urine dipstick and Urine Protein-to-Creatinine Ratio (UPCR) for detecting preeclampsia.
| Metric | Dipstick(95% CI) | UPCR(95% CI) |
|---|
| Sensitivity | 43% (31.51%, 54.21%) | 91% (73.67%, 109.19%) |
| Specificity | 40% (28.71%, 51.29%) | 7% (0.75%, 12.60%) |
| PPV | 36% (24.42%, 46.99%) | 43% (31.84%, 54.64%) |
| NPV | 47% (36.08%, 58.65%) | 50% (38.73%, 61.27%) |
| Accuracy | 41% (29.96%, 52.54%) | 44% (32.50%, 55.00%) |
| F1-Score | 39% (17.51%, 60.47%) | 59% (42.21%, 75.73%) |
Discussion
The present study evaluated the diagnostic performance of the urine dipstick test and the UPCR against the 24-hour urine protein test. The age distribution analysis showed that the majority of cases (48%) were within the age range of 20-25 years. Further analysis of the population based on the presence and absence of preeclampsia revealed that, for those aged over 25 years, there were more incidences of preeclampsia. These findings align with previous epidemiological studies indicating that preeclampsia predominantly affects women in this age group. A systematic review by Duckitt K and Harrington D shows that pregnant women aged over 30 years exhibit a gradual increase in the odds of developing preeclampsia [22]. Further studies by Chan TF et al., and Sheen JJ et al., are in agreement with our findings [23,24].
The urine dipstick test demonstrated a sensitivity of 43% and specificity of 40% in diagnosing preeclampsia, suggesting that it missed a significant number of cases. A meta-analysis study by Teeuw HM et al., analysing 19 studies (n=3700 urine samples), showed that the urine dipstick does not efficiently exclude preeclampsia patients from those with gestational hypertension [25]. Phelan LK et al., studied 170 hypertensive pregnant women to assess the efficacy of the dipstick method in detecting preeclampsia. The study concluded that the dipstick method has a significantly low PPV, with increased false-negative incidences [26]. A similar study by Jones C et al., concluded that the urine dipstick is not an adequate test for the diagnosis of proteinuria [27]. The present results further align with studies by White SL et al., and Siedner MJ et al., which reported that urine dipstick testing has poor sensitivity (57.8% and 63%, respectively) and relatively better specificity (95.4% and 86.1%, respectively), making it unreliable as a confirmatory diagnostic tool [28,29].
In comparison, UPCR showed significantly better sensitivity (91%) than the dipstick test (43%), although its specificity remained low at 7%. This aligns with a study by Chadha A and Tayade S which reported that UPCR has a sensitivity of 79.37%, a PPV of 92.59% and an accuracy of 75.79% [30]. A further study by Côté AM et al., which compared 13 studies in a systematic review, concluded that UPCR is an acceptable first-line diagnostic tool for identifying proteinuria, as 24-hour urine collections are frequently inaccurate, with discrepancies ranging from 13-68%, especially for those with a lean body weight. Among 161 women with serial 24-hour urine samples, 24.8% exhibited between-measurement differences of 25% or greater, exceeding both analytic and biological variation [31].
The present study findings are further supported by similar studies from Sanchez-Ramos L et al., which reported sensitivities and specificities for UPCR as 91.0% (95% CI 87.0-93.9) and 86.3% (95% CI 78.4-91.7), respectively [32]. Saudan PJ et al., also reported that UPCR had a positive correlation with 24-hour urine proteinuria (r=0.93, p<0.001) and demonstrated a sensitivity of 93% and specificity of 92% [33]. Other studies, such as that by Stefańska K et al., (sensitivity and specificity of 89% (95% CI, 75-97) and 100% (95% CI, 87-100), respectively) and Yamasmit W et al., (sensitivity, specificity and accuracy of 96.6%, 92.3% and 95.2%, respectively) also support these findings [34,35].
The F1-score, which balances sensitivity and precision, was notably higher for UPCR (59%) compared to the dipstick test (39%), reinforcing the notion that UPCR may be more useful for confirming proteinuria, although it may be less effective in detecting all true cases. These findings are in line with a study by Olisa CL et al., which demonstrated that UPCR has superior diagnostic accuracy compared with the dipstick method [19]. In a multicentre prospective study involving 2212 urine samples conducted by Baba Y et al., it was shown that the dipstick produced significantly higher false positive results compared to UPCR results [36]. The authors concluded that patients who show positive results on the dipstick should undergo further evaluation using UPCR results, even regardless of whether the patient is hypertensive or normotensive.
Limitation(s)
The present study has several limitations that should be considered when interpreting the data. The sample size of 80, being small, limits the ability to generalise the findings to a larger population. Additionally, while the 24-hour urine protein test is considered the gold standard, its accuracy remains a subject of debate [17]. The study was conducted at a single centre, underscoring the need for further multicentre studies to corroborate the findings.
Conclusion(s)
While the urine dipstick test and UPCR offer rapid alternatives, their diagnostic accuracy remains suboptimal when compared to the 24-hour urine protein test. Although UPCR is better at confirming proteinuria, it fails to reliably rule out preeclampsia due to its low NPV. These findings emphasise the need for a combination of clinical assessment, laboratory markers and the use of UPCR over the dipstick test as an adjunct.