Hydroxychloroquine-induced Impetigo Herpetiformis Managed with Oral Cyclosporine: A Rare Case Report
Avinash Jadhav1, Reshma Gupte2, Asharbh Raman3, Kirti Deo4
1 Associate Professor, Department of Dermatology, Venereology and Leprosy, Dr. D. Y. Patil Medical College, Hospital and Research Centre, Dr. D. Y. Patil Vidyapeeth, Pune, Maharashtra, India.
2 Junior Resident, Department of Dermatology, Venereology and Leprosy, Dr. D. Y. Patil Medical College, Hospital and Research Centre, Dr. D. Y. Patil Vidyapeeth, Pune, Maharashtra, India.
3 Senior Resident, Department of Dermatology, Venereology and Leprosy, Dr. D. Y. Patil Medical College, Hospital and Research Centre, Dr. D. Y. Patil Vidyapeeth, Pune, Maharashtra, India.
4 Professor, Department of Dermatology, Venereology and Leprosy, Dr. D. Y. Patil Medical College, Hospital and Research Centre, Dr. D. Y. Patil Vidyapeeth, Pune, Maharashtra, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Reshma Gupte, E-12, Clarion Park Society, D.P. Road, Aundh, Pune-411007, Maharashtra, India.
E-mail: guptereshma@gmail.com
Impetigo Herpetiformis (IH), or Generalised Pustular Psoriasis (GPP) of pregnancy, is a rare gestational dermatosis that lacks the usual triggers for pustular psoriasis and is often associated with adverse maternal and foetal outcomes. The onset typically occurs in the third trimester and hormonal fluctuations during pregnancy might play a role in the pathogenesis. Hypocalcaemia, hypoalbuminaemia and hypoparathyroidism are often associated with this condition. A characteristic resolution of symptoms typically occurs after delivery, with a tendency for recurrence in subsequent pregnancies. Genetic susceptibility and mutations in the gene encoding the interleukin 36 receptor antagonist may constitute significant underlying pathology. Acute Generalised Exanthematous Pustulosis (AGEP) closely resembles this condition both clinically and histopathologically and should be ruled out to ensure appropriate treatment. Drug-induced cases are seldom reported and those arising from Hydroxychloroquine (HCQ) are even rarer. The patient in the present case is a 33-year-old primiparous female in her second trimester, with a history of recurrent miscarriage for which she was prescribed HCQ prophylactically. However, 15 days later, she presented with itchy erythematous patches all over her body, which later evolved into a generalised pustular eruption. Histopathological examination revealed a large sub-corneal pustule filled with neutrophils and neutrophilic debris. A diagnosis of HCQ-induced IH was made and the patient was managed successfully with cyclosporine monotherapy. HCQ, a routinely used medication for a number of conditions, is considered safe in pregnancy; however, clinicians should be aware that it can cause IH. Early immunotherapy with cyclosporine is a safe and effective option to control the disease without the need to add glucocorticoids, thereby preventing the burden of additional adverse effects.
Gestation, Immunotherapy, Pregnancy, Pustular psoriasis
Case Report
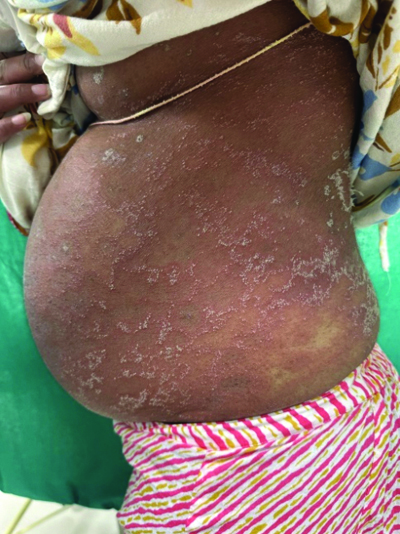
A 33-year-old primiparous female at 22 weeks of gestation presented with itchy erythematous patches all over her body for the past week. Some of these patches, located on the extremities including the palms and soles, had a dusky centre, resembling targetoid lesions. The lesions evolved over the next two days, becoming slightly enlarged, oedematous and painful, with associated swelling of the face and extremities. Subsequently, ring-shaped clusters of pinpoint pustules developed at the periphery [Table/Fig-1,2]. The scalp, joints and mucosae were uninvolved. Apart from mild body ache and malaise, fever and other systemic signs were absent. She denied any personal or family history of psoriasis. The patient revealed a history of miscarriage in her previous two pregnancies, occurring at 17 and 10 weeks, respectively. Cervical cerclage was performed in the ongoing pregnancy; however, she noticed vaginal spotting at 19 weeks of gestation. Given her history of recurrent miscarriage, the obstetrician started her on tablet HCQ 200 mg once daily prophylactically, 15 days prior to the onset of the skin lesions.
Generalised dusky erythema with maculopapular rash and ring-shaped clusters of peripheral pinpoint pustules over the trunk.
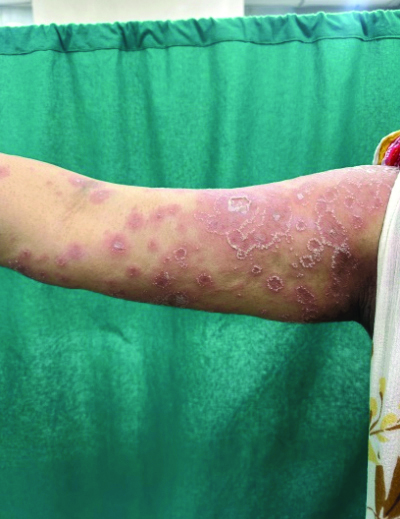
Multiple well-defined erythematous round patches studded with ring-shaped pustules at the periphery over the right arm.
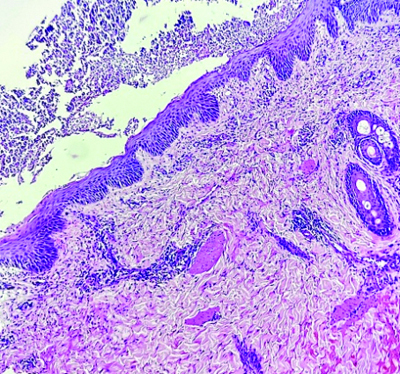
Histopathological examination {Haematoxylin and Eosin (H&E)} of a 4 mm punch biopsy specimen taken from a lesion on her arm revealed a large sub-corneal pustule filled with neutrophils and neutrophilic debris, along with mild acanthosis and spongiosis in the epidermis. The superficial dermis showed a neutrophilic infiltrate extending into the epidermis, along with a deep dermal perivascular and periadnexal neutrophilic infiltrate [Table/Fig-3]. Laboratory investigations indicated a raised Total Leucocyte Count (TLC) of 12.2×109/L (neutrophils, 89%), elevated erythrocyte sedimentation rate (64 mm/hr) and C-reactive protein (35.55 mg/L), hypocalcaemia (7.2 mg/dL), hypomagnesaemia (1.3 mg/dL), hypoalbuminaemia (2.7 g/dL) and normal parathormone levels. Anti-phospholipid antibody and antinuclear antibody tests were negative.
Histopathological examination showing mild acanthosis and spongiosis. A large sub-corneal pustule is seen filled with neutrophils and neutrophilic debris. Superficial dermis shows neutrophilic infiltrate extending into the epidermis while deep dermis shows perivascular and periadnexal neutrophilic infiltrate (H&E, 40X).
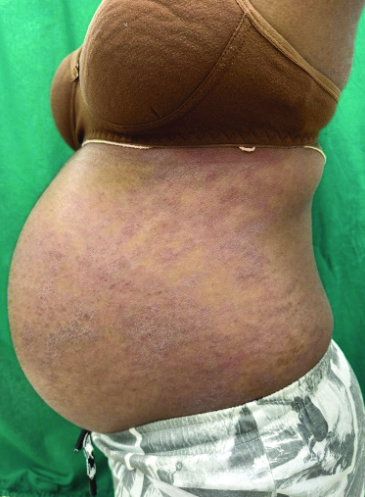
Having diagnosed the case as Impetigo Herpetiformis (IH), most likely secondary to HCQ, the culprit drug was discontinued and the patient was started on capsule cyclosporine 100 mg twice daily (3 mg/kg/day), along with calcium and protein supplementation, antihistamines and emollients. A significant symptomatic improvement ensued, evident by prompt resolution of the pustules [Table/Fig-4,5]. Two weeks later, the cyclosporine dose was tapered to 150 mg/day; however, this led to a flare-up of the skin lesions, necessitating the reinstatement of the previous dosing. Subsequently, a more gradual and careful tapering was implemented, reducing the dose by 50 mg/day every four weeks, reaching a daily dose of 50 mg at 36 weeks. The pregnancy was closely monitored for any signs of symptomatic flare-up or cyclosporine-induced adverse events. Additionally, the pregnancy was monitored for any signs of placental insufficiency or foetal distress that might warrant early induction of labour. Eventually, a healthy male child was delivered vaginally at 39 weeks, following which the cyclosporine dose was further tapered to 50 mg every alternate day for four weeks and then stopped after achieving complete symptomatic resolution. The patient followed-up a month later and there were no signs of a relapse in the mother or adverse outcomes in the child.
Patient showing marked symptomatic improvement with reduced erythema, fine scaling and clearance of pustules over the trunk within two weeks of treatment with cyclosporine.
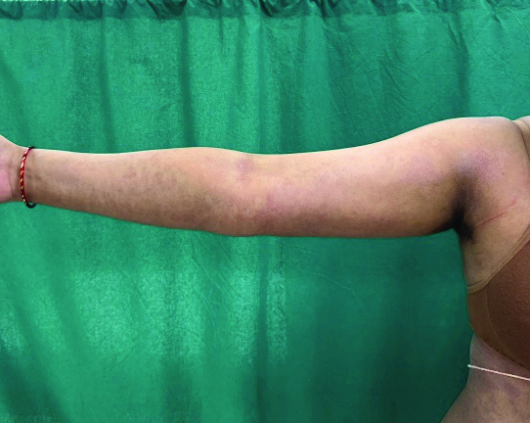
Significant improvement of symptoms with reduced erythema and prompt clearance of pustules over the right arm.
Discussion
The onset of GPP during pregnancy, also known as IH, is a rare occurrence characterised by the resolution of symptoms after delivery and a tendency to recur during subsequent pregnancies [1]. It typically occurs in the third trimester and hormonal fluctuations during pregnancy may play a role in the pathogenesis. Hypocalcaemia, hypoalbuminaemia and hypoparathyroidism are often associated with this condition [1,2]. While infection and withdrawal of systemic corticosteroids are usually associated with triggering GPP, these triggers are uncommon in cases of IH. IH is often associated with adverse maternal and foetal outcomes, necessitating early and effective disease control [1,2].
Genetic susceptibility and mutations in the gene encoding for Interleukin 36 Receptor Antagonist (IL36RN) could represent a significant underlying pathology [3]. The presence of an IL36RN mutation appears to be associated with earlier disease onset, more frequent flare-ups and a poor treatment response to standard medications [4]. Anti-malarials are well-known to aggravate or trigger new-onset psoriasis, including GPP; however, only two previous reports mention IH triggered by HCQ [2,5,6].
The patient had already experienced two miscarriages and her history of vaginal spotting was suggestive of a threatened abortion. HCQ was prescribed by the obstetrician prophylactically to prevent another miscarriage. The immunomodulatory effects of HCQ, along with its anti-thrombotic and vascular-protective actions, might explain its beneficial role in preventing recurrent miscarriages [7]. The latency period between drug intake and disease onset in the patient was two weeks, consistent with previously reported cases of HCQ-induced psoriasis. AGEP, which has also been reported to be triggered by HCQ, closely resembles GPP clinico-histopathologically and was initially considered a differential diagnosis [5]. However, the prolonged disease course and prompt resolution of symptoms after delivery reinforced the original diagnosis. Profound hypocalcaemia can rarely lead to life-threatening maternal complications, including tetany, delirium and convulsions [1], highlighting the importance of correcting underlying electrolyte disturbances.
IH is one of the few gestational dermatoses associated with adverse foetal outcomes, the most concerning being placental insufficiency, stillbirth and neonatal death [1,2,5]. Premature termination of pregnancy is often contemplated due to these complications. In fact, early onset and rapid disease progression appear to be associated with a worse prognosis and poorer foetal outcomes, warranting the use of potent immunomodulators and aggressive treatment to achieve faster disease control [2,5]. Prednisolone, cyclosporine and infliximab are generally considered first-line treatments for IH [2,5]. Cyclosporine is emerging as the preferred first-line agent, with infliximab serving as an option for resistant cases [8]. In the present case, cyclosporine monotherapy was chosen owing to its better side-effect profile for both the mother and the child. In some recalcitrant cases, biologics targeting the interleukin 17 and 23 pathways have also shown promising results [3,4]. Moreover, blocking the IL-36 pathway could emerge as an effective novel treatment option for recalcitrant IH. This pathway is well-known to play a significant role in the pathogenesis of GPP, as evidenced by excellent control of disease flare-ups with biologics targeting the IL-36 receptor [9]. Yang C et al., have recently reported successful treatment of recalcitrant IH with Spesolimab, a human monoclonal antibody against the IL-36 receptor [10].
Conclusion(s)
HCQ, a routinely used medication for a number of conditions, is considered safe in pregnancy; however, clinicians should be aware of the possibility of IH triggered by HCQ. Many practitioners still prescribe systemic glucocorticoids as the first-line treatment for IH; however, this approach may lead to undesired adverse maternal effects and birth defects, such as cleft lip and cleft palate. Cyclosporine, when started early in the disease course, is a safe and effective option to control the disease without the need to add glucocorticoids, thereby preventing the burden of additional adverse effects. In the absence of any contraindications, it should be the preferred treatment modality for IH. Systemic steroids should be avoided unless absolutely necessary and biologics should be reserved for recalcitrant cases.
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Mar 03, 2025
Manual Googling: Mar 22, 2025
iThenticate Software: Mar 24, 2025 (9%)
[1]. Wiznia LE, Pomeranz MK, Skin Changes and Diseases in PregnancyFitzpatrick’s Dermatology. Kang S (9th ed) 2019 New York, NYMcGraw Hill Education:1765-78. [Google Scholar]
[2]. Wan Y, Zhang L, Xie B, Wu J, Zhao M, Guo J, Impetigo herpetiformis in the second trimester: A case report and review of the literatureJ Int Med Res 2023 51(12):300060523121795010.1177/0300060523121795038102997PMC10725655 [Google Scholar] [CrossRef] [PubMed]
[3]. Neema S, Shrestha S, Sathu S, Vasudevan B, Excellent response to secukinumab in treatment resistant impetigo herpetiformisIndian Dermatol Online J 2022 14(1):118-19.10.4103/idoj.idoj_115_2236776161PMC9910531 [Google Scholar] [CrossRef] [PubMed]
[4]. Hsieh CY, Tsai YJ, Tsai TF, Use of biologics in Chinese pregnant patients with deficiency of interleukin-36 receptor antagonist (DITRA): A case seriesTaiwan J Obstet Gynecol 2024 63(3):405-08.10.1016/j.tjog.2024.02.00138802208 [Google Scholar] [CrossRef] [PubMed]
[5]. Liu J, Ali K, Lou H, Wang L, Wu L, First-trimester impetigo herpetiformis leads to stillbirth: A case reportDermatol Ther (Heidelb) 2022 12(5):1271-79.10.1007/s13555-022-00735-935499740PMC9059450 [Google Scholar] [CrossRef] [PubMed]
[6]. Gravani A, Gaitanis G, Zioga A, Bassukas ID, Synthetic antimalarial drugs and the triggering of psoriasis- do we need disease-specific guidelines for the management of patients with psoriasis at risk of malaria?Int J Dermatol 2014 53(3):327-30.10.1111/ijd.1223124320605 [Google Scholar] [CrossRef] [PubMed]
[7]. Dernoncourt A, Hedhli K, Abisror N, Cheloufi M, Cohen J, Kolanska K, Hydroxychloroquine in recurrent pregnancy loss: Data from a French prospective multicenter registryHum Reprod 2024 39(9):1934-41.10.1093/humrep/deae14638942601PMC11373309 [Google Scholar] [CrossRef] [PubMed]
[8]. Stanway A, Dermatoses of PregnancyRook’s Textbook of Dermatology. Griffiths CEM (10th ed) 2024 West Sussex, UKWiley-Blackwell:113.10 [Google Scholar]
[9]. Bachelez H, Choon SE, Marrakchi S, Burden AD, Tsai TF, Morita A, Trial of Spesolimab for generalized pustular psoriasisN Engl J Med 2021 385(26):2431-40.10.1056/NEJMoa211156334936739 [Google Scholar] [CrossRef] [PubMed]
[10]. Yang C, Wang Y, Li R, Tu P, Wang R, Successful treatment of recalcitrant generalized pustular psoriasis of pregnancy with spesolimabJ Dermatolog Treat 2024 35(1):233479110.1080/09546634.2024.233479138565205 [Google Scholar] [CrossRef] [PubMed]