Psoriasis is a chronic inflammatory, immune-mediated proliferative skin disorder that predominantly involves skin, nails, and joints. Chronic plaque-type psoriasis is the most common presentation, of psoriasis accounting for 90% of all cases [1]. The prevalence of psoriasis in India varies from 0.44% to 2.8% [2]. It has been associated with numerous dermatological and non-dermatological diseases, like cardiovascular diseases [3], diabetes mellitus [4], dyslipidaemia [4], and others. Pulmonary manifestations are uncommon and can include COPD, pneumonia, ILD, and nodules [5].
Interstitial Lung Diseases (ILD) or Diffuse Parenchymal Lung Diseases (DPLD) is an umbrella term used for a heterogenous group of non neoplastic lung disorders characterised by diffuse cellular infiltrates in interstitium and/or alveoli that may lead to progressive fibrosis. On the basis of aetiology and clinicoradiological features, DPLDs are classified into idiopathic interstitial pneumonias and other DPLDs with known or unknown causes. The association between Connective Tissue Disorders (CTD) and ILD is well established; but lung involvement in psoriasis is very rarely seen. The overexpression of interleukin 17 (IL-17), Tumour Necrosis Factor alpha (TNF-α), Tumour Growth Factor β (TGF-β) has been linked to the development of ILD in patients of psoriasis [6]. Psoriasis involves chronic inflammation of skin, and the excess pro-inflammatory cytokines can spill over from inflamed skin into systemic circulation, affecting organs like lung parenchyma, as almost all the blood flows into the pulmonary circulation. The IL-23/IL-17 axis plays an important role in the pathogenesis of Interstitial Pneumonia (IP) in psoriasis [7]. ILD in psoriasis is mostly reported as drug-induced pneumonitis secondary to immunosuppressants or biological therapies [8]. A few cases have been reported of the simultaneous existence of psoriasis and IP [7]. So, it is worth exploring the association between ILD and psoriasis, since both these entities share some common pathways leading to lung fibrosis. With this background, the present study was conducted with an aim to evaluate the radiological and clinical presentations of ILD in concomitant psoriasis.
Materials and Methods
This was a cross-sectional study that included 3,012 patients with psoriasis treated in the psoriasis clinic at the Department of Dermatology in SMS Medical College, Jaipur, Rajasthan, India, from March 2021 to February 2022 after taking Ethical Committee clearance (IEC NO-139/MC/EC/2021). Out of the total patients, 144 patients were referred to the Department of Respiratory Medicine with complaints of respiratory symptoms for further evaluation.
Inclusion criteria: (a) Age >18 years; (b) Diagnosed with either psoriasis or psoriatic arthritis by dermatologists and having respiratory complaints.
Exclusion criteria: Patients with Chronic Obstructive Pulmonary Disease (COPD), bronchial asthma, pneumonia, pulmonary tuberculosis, or other cardiac causes leading to shortness of breath were excluded after performing the reversibility test on spirometry and other clinical and radiological evaluation. Patients aged ≤18 years of age and immunology positive for other CTDs like lupus, rheumatoid arthritis, sarcoidosis, scleroderma, or having history of Coronavirus Disease 2019 (COVID-19) infection were also excluded from the study.
Study Procedure
A comprehensive review of history was conducted to elicit possible exposure to substances causing ILD, the autoimmune profile, and Cartridge-Based Nucleic Acid Amplification Test (CBNAAT) to rule out tuberculosis. Additionally, chest radiology (chest X-ray and HRCT), Pulmonary Function Tests (PFT), Diffusion Capacity of Lungs for Carbon Monoxide (DLCO), 2D Echo etc., were performed.
The PASI score and type of psoriasis or psoriatic arthropathy were diagnosed by a dermatologist. This score was calculated by dermatologist by quantitative evaluation of clinical severity of psoriasis. In this scoring system, four sites were included: head (h), upper limb (u), trunk (t), and lower limbs (l), and were separately scored on three parameters: erythema (E), induration (I), and scaling (D). Each one of these parameters is graded on a severity scale of 0-4 (where 0 stands for nil; 1 is mild; 2 is moderate; 3 is severe; and 4 is considered very severe). The area-wise percentage involvement of the involved sites was calculated as follows: 1=less than 10% involvement; 2=10-29%; 3=30-49%; 4=50-69%; 5=70-89%; and 6=more than 90%. The severity grading of the three symptoms is multiplied by the numerical value of the areas involved and by the various percentages of the four body areas. These values are then added to obtain the PASI score. The final formula for PASI score is: PASI=0.1 (Eh+Ih+Dh) Ah+0.2 (Eu+Iu+Du) Au+0.3 (Et+It+Dt) At+0.4 (El+Il+Dl) Al. The maximum score of the PASI is 72 [9].
Outcomes Assessed
Primary outcome: Proportion/prevalence of concomitant ILD in psoriasis patients.
Secondary outcome: Correlation between PASI score and severity of ILD, and radiographic characteristics were evaluated.
Statistical Analysis
Data analysis was carried out using Statistical Package for Social Science (SPSS) v22. Categorical data was expressed as percentages (%); on the other hand, the continous data were presented as the means±SD and as median with 25th and 75th percentiles (interquartile range). Pearson’s correlation test was used to find out statistical differences between the study groups. The p-value less than 0.05 was considered significant.
Results
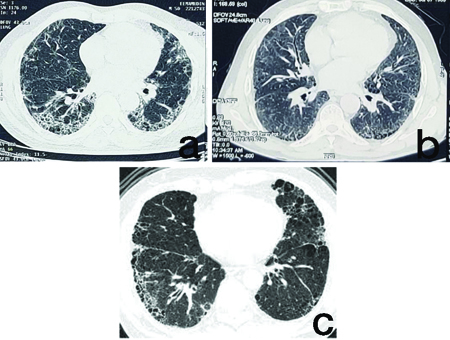
Out of the 3,012 patients, 10 patients were diagnosed with ILD. The median patient age was 51.5 years (range: 25-65 years). The median time for the development of respiratory symptoms from diagnosis of psoriasis was 24.5 months (range: 6-60 months). The median Forced Vital Capacity (FVC) percentage predicted was 58% (range: 32-85), and DLCO% predicted was 42% (range: 23-79) [Table/Fig-1]. All patients had chronic plaque-type psoriasis [Table/Fig-2]. The most common respiratory symptom at presentation was dry cough 6 (60%), followed by dyspnoea on exertion 4 (40%). Various serological testing and immunofluorescence assays to diagnose antibody patterns of various CTD were negative in all patients. A total of 5 (50%) patients had not previously or concomitantly been exposed to immunosuppressant, whereas 5 (50%) had been exposed. The most common immunosuppressants included methotrexate (n=4), followed by cyclosporine (n=1). Radiographic diagnosis were as follows: UIP pattern in 7 patients (70%, [Table/Fig-3a]), NSIP pattern in 2 patients (20%, [Table/Fig-3b]), and CPFE in 1 patient (10%, [Table/Fig-3c]). The [Table/Fig-4] demonstrates the demographic and clinical features of ILD patients with concomitant psoriasis.
| Characteristics | Median, % or range |
|---|
| Age (years) | 51.5 years (25-65 years) |
| Male | 5 (50%) |
| Female | 5 (50%) |
| Time for development of respiratory symptoms from diagnosis of psoriasis | 24.5 months (6-60 months) |
| FVC% predicted | 58% (32-85%) |
| DLCO% predicted | 42% (23-79%) |
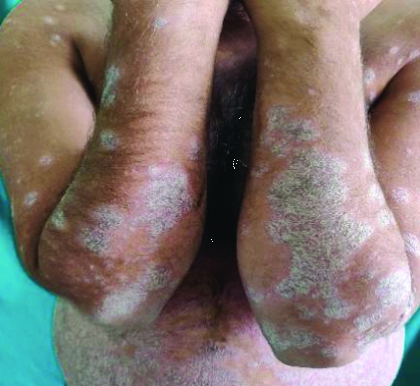
Showing chronic plaque-type psoriasis involving upper limbs and trunk.
HRCT Chest of psoriasis patients showing: (a) UIP pattern; (b) NSIP pattern; (c) CPFE pattern.
Demographic and clinical features of ILD patients with psoriasis.
| Case | Age (years) | Sex | Type of psoriasis | PASI score | Treatment | Time of development of respiratory symptoms from diagnosis of psoriasis | Duration of treatment before development of respiratory symptoms | Radiological diagnosis of ILD (HRCT pattern) |
|---|
| 1 | 51 | M | CPP | 38 | Methotrexate | 6 months | 2 months | UIP |
| 2 | 28 | F | CPP | 32.8 | PUVAsol | 24 months | 2 months | Fibrotic NSIP |
| 3 | 62 | F | CPP | 20 | Cyclosporine | 36 months | 5 months | Cellular NSIP |
| 4 | 25 | M | CPP | 34.6 | Methotrexate | 9 months | 6 months | UIP |
| 5 | 35 | M | CPP | 24 | Methotrexate | 15 months | 6 months | UIP |
| 6 | 55 | F | CPP | 34.2 | Methotrexate | 11 months | 3 months | UIP |
| 7 | 58 | F | CPP | 36 | PUVAsol | 12 months | 3 months | CPFE |
| 8 | 65 | M | CPP | 16 | Acitretin | 60 months | 12 months | UIP |
| 9 | 52 | M | CPP | 20 | PUVAsol | 37 months | 18 months | UIP |
| 10 | 26 | F | CPP | 40 | Acitretin | 6 months | 24 months | UIP |
CPP: Chroni plaque-type psoriasis
In the present study, on comparing PASI scores among males, the mean value was 26.52±9.4, while among females, the mean value was 32.6±7.5. The mean time of development of respiratory symptoms from diagnosis of psoriasis scores among males was 21±15.4 months, and among females, it was 17.8±12.13 months. The mean duration of treatment before development of respiratory symptoms among males was 8.8±6.2 months and among females, it was 7.4±9.3 months [Table/Fig-5].
Statistical data of the various groups of patients.
| Variables | Gender | N | Mean | Std. Deviation | Std. Error mean |
|---|
| PASI score | F | 5 | 32.600 | 7.5445 | 3.3740 |
| M | 5 | 26.520 | 9.4420 | 4.2226 |
| Time of development of respiratory symptoms from diagnosis of psoriasis (months) | F | 5 | 17.80 | 12.133 | 5.426 |
| M | 5 | 21.00 | 15.411 | 6.892 |
| Duration of treatment before development of respiratory symptoms | F | 5 | 7.40 | 9.343 | 4.179 |
| M | 5 | 8.80 | 6.261 | 2.800 |
M: Males; F: Females
On applying correlation statistics, it was recorded that PASI scores and time of development of respiratory symptoms from diagnosis of psoriasis were strongly and inversely correlated (r=-0.906) (p<0.001). Among other study parameters no significant correlations was found [Table/Fig-6].
Outcomes and statistical correlations of various groups.
| Parameters | PASI score | Time of development of respiratory symptoms from diagnosis of psoriasis | Age | Duration of treatment before development of respiratory symptoms |
|---|
| PASI score | Pearson correlation | 1 | -0.906** | -0.508 | -0.134 |
| Sig. (2-tailed) | | 0.001 | 0.134 | 0.713 |
| Time of development of respiratory symptoms from diagnosis of psoriasis | Pearson correlation | -0.906** | 1 | 0.511 | 0.165 |
| Sig. (2-tailed) | 0.001 | | 0.131 | 0.649 |
| Age (years) | Pearson correlation | -0.508 | 0.511 | 1 | -0.184 |
| Sig. (2-tailed) | 0.134 | 0.131 | | 0.611 |
| Duration of treatment before development of respiratory symptoms | Pearson correlation | -0.134 | 0.165 | -0.184 | 1 |
| Sig. (2-tailed) | 0.713 | 0.649 | 0.611 | |
**Correlation is significant at the 0.01 level (2-tailed)
Discussion
Psoriasis is defined as a papulosquamous disorder characterised by disordered keratinisation arising due to T-cell mediated immune dysregulation.
In the present study, out of the 3,012 patients enrolled in the psoriasis clinic, 10 (0.33%) developed fibrotic lung disease, and 144 (4.78%) developed respiratory symptoms; however, the underlying cause of these symptoms as ILD was diagnosed in only 0.33% of the cases. The remaining patients had other respiratory ailments that led to cough and breathlessness. A similar study conducted by Ishikawa G et al., found that 4.7% of patients had concomitant psoriasis and fibrotic lung disease [10]. In contrast to our study, this was a retrospective study that enrolled patients from an ILD clinic, and their medical records were reviewed. Patients with a concomitant history of psoriasis were further evaluated based on their medical history. In a wide epidemiological study conducted by Peluso R et al., the incidence of ILD was found to be 1% in a total of 387 patients with psoriatic arthropathy [11].
Methotrexate-induced lung toxicity is well documented [11]. In the present study, out of the 10 patients, only 5 (50%) had a history of immunosuppressant use in the form of methotrexate and cyclosporine. The remaining patients developed ILD without prior exposure to immunosuppressive therapy. This ignites the debate to explore various immunological pathways and underlying mediators common to both conditions that may lead to lung fibrosis. In the study by Ishikawa G et al., 63.6% of the cases who had both psoriasis and ILD were not exposed to immunosuppressive therapy, either concomitantly or previously [10].
In the present study, PASI scores had an inverse relationship with the time of development of respiratory symptoms, and this correlation was statistically significant. Patients with high PASI scores took less time to develop respiratory symptoms (r=-0.906, p<0.001). The probable reason may be an overexpression of pro-inflammatory cytokines in patients with high PASI scores, leading to the development of respiratory symptoms and fibrotic lung changes [12,13]. The pathogenesis leading to the development of lung fibrosis in patients with psoriasis may be linked to the upregulation of pro-inflammatory cytokines such as IL-17, Tumour Necrosis Factor α (TNF-α), and Transforming Growth Factor β (TGF-β) [14]. This was a unique observation in our cases, and we could not find any literature in Medline showing a correlation between high PASI scores and the development of lung fibrosis. Psoriasis may lead to immune dysregulation, which may further trigger an abnormal immunological cascade in the lung parenchyma, leading to fibrosis and other interstitial changes [14].
Immunological markers for collagen vascular diseases were negative in all patients. The mean FVC% predicted and DLCO% predicted were 58% and 42%, respectively, at the time of presentation. The mean decline in FVC values was mild (range: 32-85%) compared to DLCO values (23-79%), which showed very severe levels of impairment. This decline in FVC and DLCO was also observed in the study conducted by Ishikawa G et al., [10]. The study established that patients presented with mild impairment in FVC with a median of 69% predicted (range: 38-89%). On the contrary, the DLCO exhibited a more severe reduction, with a median of 47% predicted (range: 17-95%). Remarkably, compared to patients with Idiopathic Pulmonary Fibrosis (IPF), the decline in pulmonary function was relatively mild, as reflected by a subtle annual variation in predicted FVC%, ranging from -5.7% to +18%. We did not perform serial FVC and DLCO to see the performance of pulmonary function, as most of the patients were lost to follow-up. Radiological findings in 7 (70%) cases were consistent with a UIP pattern, NSIP pattern was seen in 2 (20%) cases and 1 (10%) had findings consistent with a CPFE pattern. A high incidence of severe fibrosis in the form of a UIP pattern indicated that the immune dysregulation triggered in psoriasis is severe and is much more than an inflammatory process.
All the patients who developed UIP were current (71.42%; n=5) or former smokers (28.57%; n=2).
Although the prevalence of ILD in our study is below one percent, it cannot be ignored, as the plausible association of psoriasis with a life-threatening entity such as lung fibrosis cannot be overlooked. Smoking history and use of agents such as methotrexate were the biggest confounders in the study, as both can independently perpetuate fibrotic changes in lung parenchyma. Methotrexate appears to be less likely the cause of lung fibrosis in our study, as the duration of methotrexate intake before the development of respiratory symptoms was less than 10 months in all the patients. It is very unlikely for a fibrosis-causing drug to induce fibrotic changes in such a short duration.
Limitation(s)
(a) Relatively small sample size; (b) Study has been performed in a single centre; (c) Study was not conducted among the general population.
Conclusion(s)
Although the proportion of ILD was less than one percent, a confirmatory association between these two entities cannot be established and more of prospective studies with a much larger sample size are needed to establish a direct association between these two entities. The prevalence of ILD in general population needs to be compared to that of the psoriasis cohort to set out the association between these two entities.
CPP: Chroni plaque-type psoriasis
M: Males; F: Females
**Correlation is significant at the 0.01 level (2-tailed)