Recent developments in neurosurgical interventions have increased interest in detailed anatomical knowledge of different craniometric points, such as the pterion, which is an important anatomical bony landmark on the lateral aspect of the skull in the temporal fossa. It is located at the junction of the calvaria, skull base and facial skeleton. Four different cranial bones or bony components-namely, the frontal bone, the greater wing of the sphenoid, the parietal bone and the squamous part of the temporal bone-converge over the pterion. It is also closely related to the anterolateral fontanelles in the foetal skull [1,2]. Based on the type of bony contact, either directly or through a small sutural bone, four different categories of pterion are classified: sphenoparietal, frontotemporal, stellate and epipteric. In these cases, new centers of ossification appear as sutural bone binding other bones [3]. Subsequently, two more subtypes-zygomatico-parietal and zygomatico-temporal-were incorporated as variants of the sphenoparietal and frontotemporal types, respectively [4].
The pterion is considered the weakest part of the skull, being vulnerable to blunt trauma, which can lead to the rupture of the anterior division of the Middle Meningeal Artery (MMA) and result in an extradural haematoma [5]. The shape and localisation of this sutural pattern are highly variable, making it important as a superficial projection of various deep-seated structures of the brain that are pertinent to preoperative planning, operative procedures and postoperative outcomes during neurosurgical interventions in the cranial fossae [6].
Studies have also indicated that the position and type of pterion may change with age and sex, which might serve as a basis for age and sex determination for forensic experts [6-8]. Furthermore, it overlies other clinically significant structures of the brain, such as Broca’s motor speech area, the insula and the course of the Middle Cerebral Artery (MCA). Consequently, interventions on Broca’s area, the repair of MCA aneurysms, or access to the optic canal are often performed through a “pterional” or lateral approach. This approach, with a shorter duration, has been considered better than traditional methods, as it offers the advantages of minimal tissue damage, a lower risk of brain manipulation and better cosmetic outcomes [9,10].
Considering the recent interest, the present study aimed to observe variations in the morphology and position of this sutural confluence, along with its clinicoanatomical association, to contribute anatomical reference data related to skulls in the eastern Indian population.
Materials and Methods
The cross-sectional study was conducted in the Department of Anatomy at Midnapore Medical College, Paschim Medinipur, West Bengal, India from June 2024 to December 2024, after obtaining approval from the Institutional Ethical Committee (IEC/2024/13). A total of 62 adult dry skulls of unknown age and sex were studied bilaterally based on availability and were carefully documented with photographs for morphological and morphometric analysis of the pterion.
Inclusion criteria: Adult dry skulls without any pathology or bony damage were included in the study.
Exclusion criteria: Skulls with decomposition in the areas of interest on both sides were excluded from the study.
Study Procedure
Based on morphological characteristics, the sphenoparietal, frontotemporal, stellate and epipteric types of pterion were defined [3]. Skulls were also categorised based on either symmetrical or asymmetrical distribution of these sutural patterns on either side. For metric analysis, linear distances between the center of the pterion and adjacent standard anatomical bony landmarks were measured using a digital vernier caliper with an accuracy of 0.01 mm. Five linear measurements were taken on either side for pterion [Table/Fig-1], as follows:
Morphometric measurements: P: centre of pterion (P); FZS: frontozygomatic suture; MZ: zygomatic arch; MT: tip of mastoid process; SMS: highest point on suprameatal spine; IN: inion.
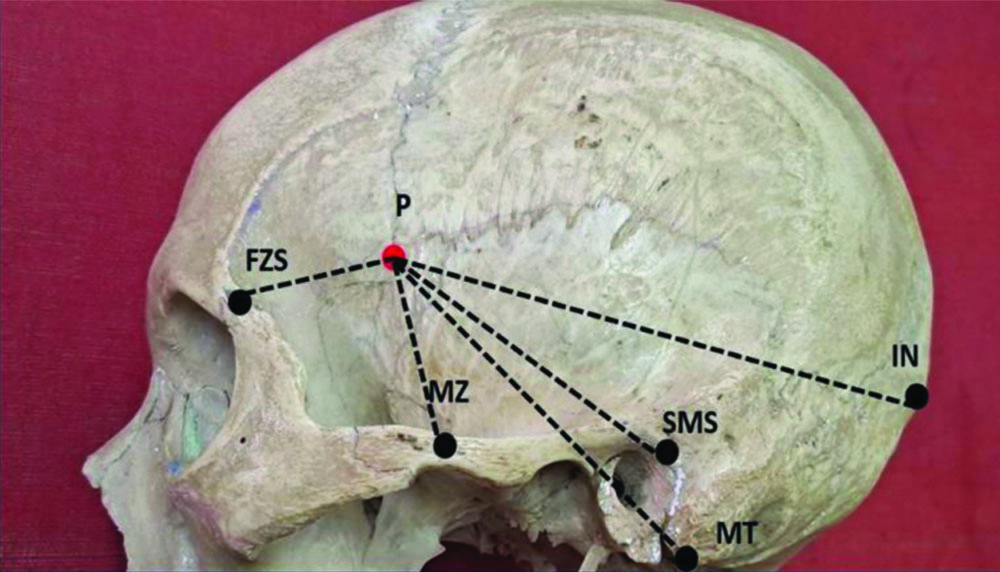
i) The distance between the center of the pterion (P) and the mid-point of the zygomatic arch (MZ) is labelled as P-MZ;
ii) The distance between the center of the pterion (P) and the center of the Frontozygomatic suture (FZS) is labelled as P-FZS;
iii) The distance between the center of the pterion (P) and the inion (IN) is labelled as P-IN;
iv) The distance between the center of the pterion (P) and the tip of the mastoid process (MT) is labelled as P-MT;
v) The distance between the center of the pterion (P) and the highest point on the Suprameatal spine (SMS) is labelled as P-SMS.
All morphological and morphometric parameters were compared based on the side of origin of the skulls.
Statistical Analysis
Results were statistically analysed using Statistical Package for the Social Sciences (SPSS) software version 25.0 with the Chi-square test and Independent samples t-test for categorical (morphological parameters) and numerical (morphometric parameters) data, respectively. A p-value <0.05 was considered statistically significant.
Results
In the present study, 62 skulls were studied bilaterally and the following observations were made:
Morphological analysis: Four types of pterion were identified [Table/Fig-2]. The most common variant was the sphenoparietal type of pterion, which was observed in 35.48% on the right-side and 31.45% on the left-side among the total examined skulls [Table/Fig-3]. No statistically significant difference was observed when comparing these morphological variants between the sides (Chi-square value=1.23, df=3, p-value=0.74).
Morphological variations of pterion: SP-Sphenoparietal; ST- Stellate; EP-Epipteric; FT-Frontotemporal; S-Sphenoid; P-Parietal; T-Temporal; F-Frontal; *sutural bone.
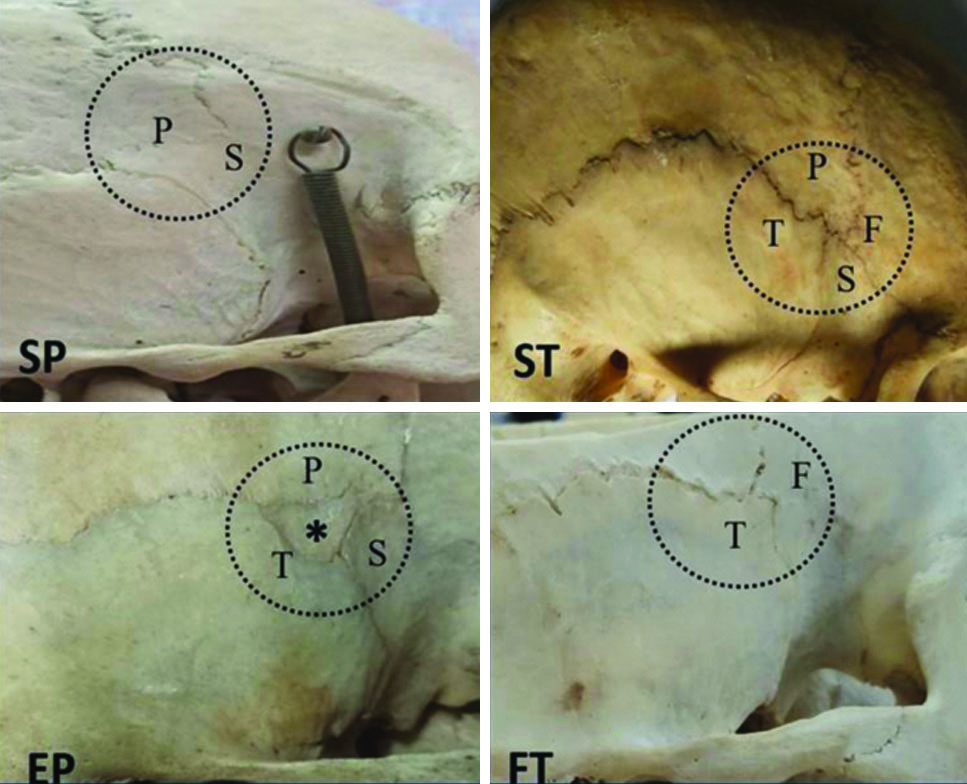
Frequency distribution of different types of pterion.
| Morphological variants | Right-side
N (%) | Left-side
N (%) | Total
N (%) |
|---|
| Sphenoparietal | 44 (35.48) | 39 (31.45) | 83 (66.93) |
| Stellate | 7 (5.64) | 10 (8.06) | 17 (13.71) |
| Epipteric | 7 (5.64) | 7 (5.64) | 14 (11.29) |
| Frontotemporal | 4 (3.22) | 6 (4.84) | 10 (8.06) |
| Total sides | 62 | 62 | 124 |
Based on the different types of pterion present on either side, the skulls were categorised as Type I to Type V in cases of asymmetrical (unilateral) distribution and Type 1 to Type 3 in cases of symmetrical (bilateral) distribution. The most common type among the asymmetrical distribution was the sphenoparietal variety. The bilateral presence of the sphenoparietal type was also the most prevalent, observed in 46.77% of skulls [Table/Fig-4].
Morphological classification of skulls based on types of pterion.
| Side of skull | Types of skulls | Types of pterion | Number of skulls (%) |
|---|
| Unilateral (Right-Left) | Type-I | Sphenoparietal-Stellate/Stellate-Sphenoparietal | 9 (14.52) |
| Type-II | Sphenoparietal -Frontotemporal/Frontotemporal-Sphenoparietal | 9 (14.52) |
| Type-III | Sphenoparietal -Epipteric/Epipteric-Sphenoparietal | 7 (11.29) |
| Type-IV | Frontotemporal-Stellate/Stellate-Frontotemporal | 1 (1.61) |
| Type V | Epipteric-Stellate/Stellate-Epipteric | 1 (1.61) |
| Bilateral (Right-Left) | Type 1 | Sphenoparietal | 29 (46.77) |
| Type 2 | Stellate | 3 (4.84) |
| Type 3 | Epipteric | 3 (4.84) |
Morphometric analysis: Statistical analysis revealed no significant difference between the right and left-sides in all measurements, with p-value >0.05 [Table/Fig-5].
Morphometric evaluation of pterion.
| Morphometric variables | Right (mm) Mean±SD (Min-Max) | Left (mm) Mean±SD (Min-Max) | Total (mm) Mean±SD (Min-Max) | p-value* |
|---|
| P-MZ | 38.96±3.80
(30.44-45.92) | 39.05±3.24
(32.75-45.62) | 39.00±3.52
(30.44-45.92) | 0.88 |
| P-FZS | 32.16±5.21
(20.36-42.23) | 31.47±5.26
(21.95-39.72) | 31.82±5.23
(20.36-42.23) | 0.46 |
| P-IN | 131.65±5.16
(119.76-141.7) | 133.07±6.95
(119.61-145.77) | 132.36±6.14
(119.61-145.77) | 0.20 |
| P-MT | 83.06±5.55
(70.45-92.47) | 81.82±5.78
(68.88-92.03) | 82.44±5.67
(68.88-92.47) | 0.23 |
| P-SMS | 59.81±4.31
(50.69-69.53) | 60.14±4.44
(50.38-68.16) | 59.97±4.36
(50.38-69.53) | 0.70 |
*Independent sample t-test
Discussion
Variation is the rule of God’s creation. Bony surface landmarks on dry skulls are no exception to this law, as their morphology and relative locations are not constant [1]. Morphogenetic studies have demonstrated the development of such sutural patterns in relation to skull growth adaptation. The roles of ethnicity, race, sex and environmental factors have also been documented as determinants of the variability in the type and location of these landmarks, with the MSX2 gene reported to be involved in the process of articulation of cranial bony segments leading to the formation of sutures [11].
The pterion is used in treating various anterior and middle cranial base lesions and is recognised as a safe surgical landmark in various neurosurgical procedures, even in neonates [10]. A recent large-scale systematic review encompassing 18,694 skull sides highlighted significant variability in the pterion, which could improve patient outcomes following neurosurgical interventions involving the anterior and middle cranial fossa by enhancing precision and safety during procedures [11]. Morphological variations of the pterion were reported, with the sphenoparietal type being the most common in previous studies, a finding that was also confirmed in the present study, where this type was observed in 66.93% of cases [Table/Fig-6] [3,5,6,10,12-18]. Although the sphenoparietal type of pterion is universally accepted as the most common variant, considerable variations in its incidence have been observed across different populations [19]. The increased incidence of the sphenoparietal type aligns with phylogenetic theory, which illustrates the changing pattern of the pterion from the frontotemporal type in other primates to the sphenoparietal type in humans [5].
Comparison of different types of pterion between present and previous studies [3,5,6,10,12-18].
| Author | Country | Types of pterion (%) |
|---|
| Sphenoparietal | Stellate | Epipteric | Frontotemporal |
|---|
| Murphy T [3] | Australia | 73.2 | 0.7 | 18.4 | 7.7 |
| Seema and Mahajan A [5] | North India | 89 | 4 | 12 | 7 |
| Hossein JM et al., [6] | Iran | 84.2 | 3.5 | 2.4 | 8.1 |
| Natsis K et al., [10] | Greece | 58.4 | 25 | 15.5 | 1.1 |
| Mwachaka PM et al., [12] | Kenya | 66 | 7 | 12 | 15 |
| Saxena RC et al., [13] | North India | 84.72 | 5.17 | - | 10.01 |
| Zalawadia A et al., [14] | West India | 91.7 | 1.2 | 4.8 | 2.4 |
| Hussain SS et al., [15] | India | 69.25 | 9.7 | 3.7 | 17.35 |
| Ukoha U et al., [16] | Nigeria | 75.3 | 1.7 | 3.6 | 19.5 |
| Ersoy M et al., [17] | Turkey | 96 | 0.2 | 9 | 3.8 |
| Oguz O et al., [18] | Turkey | 88 | - | 2 | 10 |
| Present study | Eastern India | 66.93 | 13.71 | 11.29 | 8.06 |
Among other categories of pterion, variable incidences have been described previously for the frontotemporal, stellate and epipteric types [12,17,20,21]. The presence of sutural bones in the epipteric type is usually quiescent in nature and due to lack of awareness, it may lead to complications during burr hole surgeries or even extend to cause orbital penetrations [9]. These small accessory bones have also been found in greater numbers in skulls with hydrocephalus, which causes rapid cranial expansion [5]. Present study observed these types in 8.06%, 13.71% and 11.29% of sides, respectively. Previous studies mentioned skulls with a missing frontotemporal type [9,18,21], stellate type [21,22], or epipteric type [8]; however, present study observed the presence of all four types mentioned above, with variable incidences. While some authors reported gender variations [6,8], another study [10] found that age and gender had no significant impact on the types of pterion.
Morphometric evaluation of the pterion is also essential, as it can serve as a guide for various neurosurgical procedures, such as incising tumours in the inferior aspect of the frontal lobe, including olfactory meningiomas [5]. Radiological studies have further confirmed a significant relationship between the anterior division of the MMA and the center of the pterion [9]. Although rare, surgeons must be aware of the possibility of altered positioning of the pterion, particularly when there is a variant origin of the ophthalmic artery from a frontal branch of the MMA, which can lead to complications such as pterional craniotomy-induced blindness [10]. A higher position of the pterion in male skulls, attributed to the robust character of the male zygoma, has been reported, making the zygoma an important landmark for localising the pterion [5,9,23].
The literature indicates that the distance between the pterion and the mid-point of the zygoma is 40 mm in Turkish and Nigerian skulls, 38 mm in Thai, Anatolian and Kenyan skulls and 36 mm in the Indian ethnic group [24]. The position of the pterion was mentioned by Seema and Mahajan A in their study, reporting a distance from the margin of the frontozygomatic suture of 3.1±0.44 cm and 3.4±0.4 cm on the right and left-sides, respectively [5]. In contrast, Muche A studied Ethiopian skulls and observed the same distance as 2.92±0.05 cm on the right and 2.75±0.05 cm on the left-side, respectively [9]. Another study by Gupta V et al., noted that the pterion was located 2.83 cm behind the frontozygomatic suture [25]. In the present study, the pterion was found to be 39±3.52 mm superior to the mid-point of the zygomatic arch and 31.82±5.23 mm posterior to the frontozygomatic suture, which nearly corresponds to the findings of other studies [2,5,8,9,24,26].
Present study observed a statistically insignificant bilateral variation in the position of the pterion, similar to previous studies conducted by Adejuwon SA et al., and Kamath V et al., [8,24]. Studies have also been conducted among different populations using other craniometric reference points to define the location of the pterion, such as the tip of the mastoid, inion and suprameatal spine, revealing a wide range of variations [9,19,26,27]. Present study noted that the pterion was more distally situated from the suprameatal spine than reported in previous studies [9]. Present study observations also matched previous research regarding the position of the pterion in relation to the inion and tip of the mastoid process [26,27], whereas other literature indicated that the pterion was found to be closer to these reference points [9]. Thus, the considerable variation in the shape and location of the pterion observed across different populations may indicate the influence of genetic and environmental factors on the growth of various bony parts of the cranium.
Limitation(s)
As the sample size in the present study depended on the availability of skulls in the department, proper sampling methods could not be employed during the selection process. Thus, the maximum number of skulls available during the study period was considered. The present study did not have any bias regarding age and gender-related changes. Additionally, authors did not obtain data related to morphometry based on internal craniometric reference points due to a lack of an adequate number of open skulls. Therefore, further large-scale multicentric studies can be conducted in the future to obtain population-specific data on the pterion.
Conclusion(s)
Alterations in the type and position of the pterion in relation to different anatomical bony landmarks are not infrequent. Therefore, the morphological and morphometric assessment of the pterion, along with its clinical associations in the present study, will undoubtedly contribute to a database in the field of neurosurgery that can be evaluated preoperatively for safer and more effective surgical outcomes.
*Independent sample t-test