Pierre Robin Sequence (PRS) is characterised by micrognathia, glossoptosis, Cleft Palate (CP) and cardiac defects. The key anaesthetic concerns in managing PRS include the patient’s age, assessment of associated congenital birth defects or syndromes and the potential for a shared airway and anticipated difficult intubation. Intubation should be performed using either a nasal or oral endotracheal tube {Ring-Adair-Elwyn (RAE) or flexometallic} of the appropriate size, with video laryngoscopy or fiberoptic equipment available, if necessary. This toddler, a known case of PRS with a CP, with micrognathia since birth and a history of Dandy-Walker syndrome, presented with complaints of feeding difficulties, growth restriction and recurrent Upper Respiratory Tract Infections (URTIs) since birth. The patient was nebulised and premedicated preoperatively. Induction was achieved with sevoflurane and intubation was performed with a flexometallic tube using fiberoptic guidance due to a Cormack-Lehane grade 3, indicating an anticipated difficult airway. Propofol and the muscle relaxant atracurium were administered and a bilateral maxillary block was provided for postoperative analgesia. The case was managed appropriately throughout induction, intraoperatively and during extubation, with an uneventful postoperative recovery.
Case Report
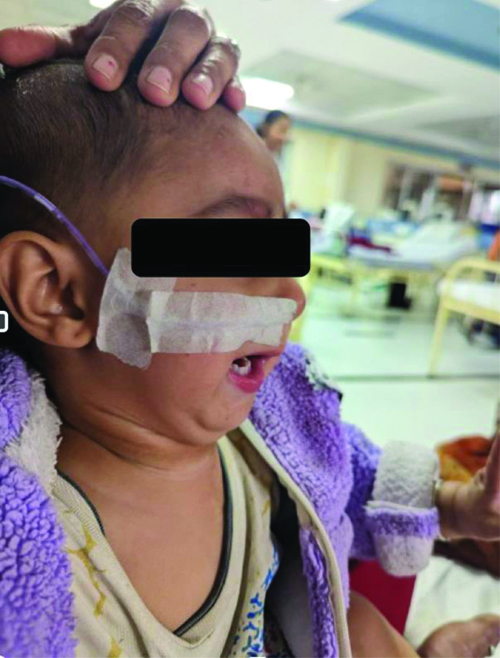
A 1-year-old male toddler, weighing 12.75 kg, with a known case of Pierre Robin syndrome and CP since birth. He had a history of Dandy-Walker syndrome [Table/Fig-1] and presented with complaints of feeding difficulties, growth restriction and recurrent URTIs. He was delivered by caesarean section due to a twin pregnancy, with a birth weight of 1.8 kg and had a seven-day stay in the Neonatal Intensive Care Unit (NICU) requiring oxygen due to heart disease. He underwent Ventricular Septal Defect (VSD) closure and Patent Ductus Arteriosus (PDA) ligation at six months, performed under general anaesthesia with fiberoptic intubation due to difficult airway management. He was placed on a ventilator for 20 days postoperatively in the Intensive Care Unit (ICU). His developmental milestones were achieved and immunisations were up to date. On examination, the patient exhibited a receding jaw, micrognathia and retrognathia, with a feeding tube in place, a bifid uvula, a CP and a Mallampati score of 3.
Toddler with retrognathia, micro mandible.
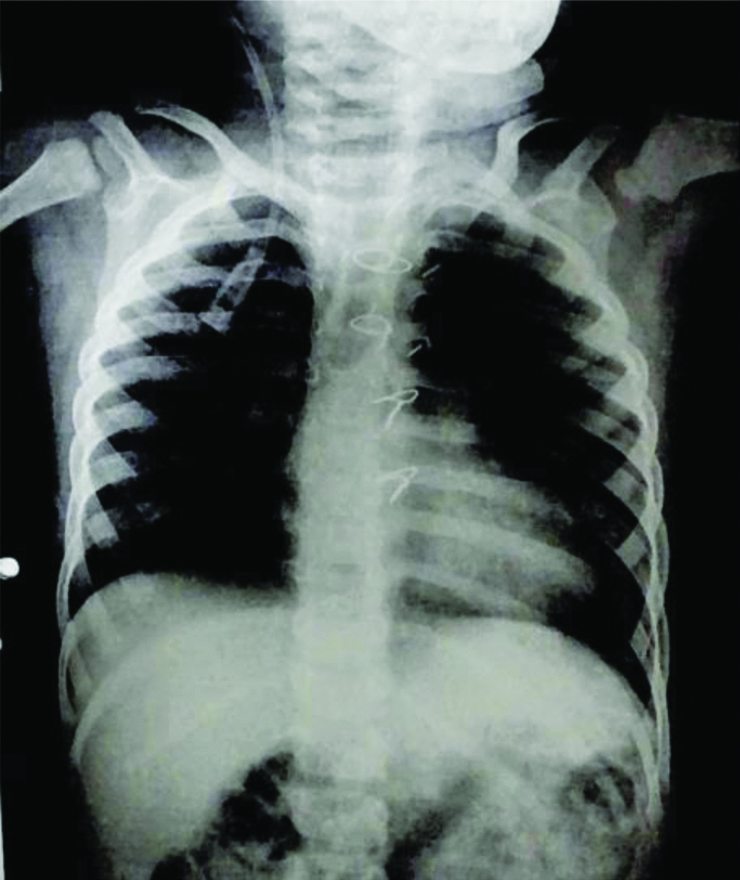
The chest X-ray revealed normal bilateral lung fields and a boot-shaped heart [Table/Fig-2]. The ECG showed no abnormalities and the 2D echocardiogram confirmed the post-VSD closure status, with the VSD patch intact, no signs of Pulmonary Artery Hypertension (PAH) and a Left Ventricular Ejection Fraction (LVEF) of 67%. Laboratory results included a haemoglobin level of 11.7 g/dL, White Blood Cell (WBC) count of 8700/μL, platelet count of 140,000/μL, Prothrombin Time (PT)/International Normalised Ratio (INR) at 12.6/1.1 seconds, urea at 31 mg/dL and creatinine at 0.37 mg/dL. Both liver function tests and serum electrolytes were within normal limits. Written and informed consent was obtained from the parents after a clear explanation of the anaesthesia procedure and the associated risks, including the potential challenges related to a difficult airway. The patient was classified as ASA grade 2 under the American Society of Anaesthesiologists.
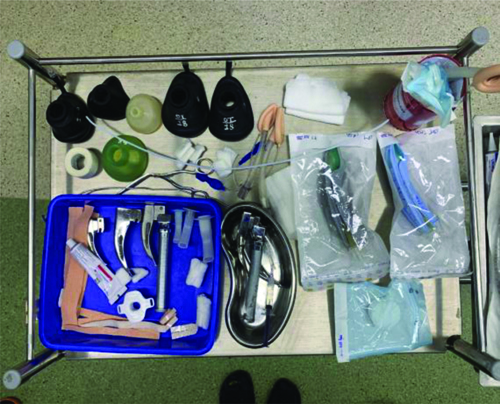
The patient was nebulised with Budecort 250 mcg on the morning of surgery. A 22-gauge i.v. cannula was secured in both feet to maintain hydration during Nothing By Mouth (NBM) hours. In the preoperative phase, nasal midazolam (0.2 mg/kg) and ketamine (3 mg/kg) were administered. Monitoring devices, including Electrocardiogram (ECG), non invasive blood pressure monitoring, EtCO2, pulse oximetry and a temperature probe, were attached. A difficult airway cart and all necessary emergency drugs were kept ready in preparation for any airway management challenges or complications [Table/Fig-3].
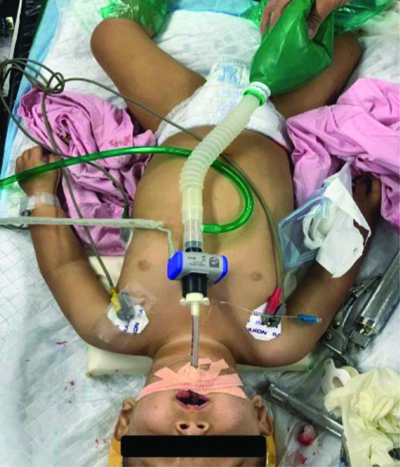
Spontaneous inhalation was induced using a 50% mixture of oxygen and air along with sevoflurane (1%). A 3.5 mm endotracheal tube was inserted into one nostril after adequate lubrication, attached to a Jackson-Rees (JR) circuit, with ventilation confirmed by bag movements and EtCO2 readings. A 2.8 mm fiberoptic scope was passed through the other nostril and once the vocal cords were visualised, lignocaine was sprayed. Oral intubation was performed using a size 4 flexometallic endotracheal tube [Table/Fig-4]. Bilateral air entry was confirmed on auscultation, the EtCO2 graph was displayed on the monitor and the tube was secured with throat packing completed. Intubation was performed with a flexometallic tube using fiberoptic guidance due to a Cormack-Lehane grade 3, indicating an anticipated difficult airway.
Propofol (2 mg/kg) and fentanyl (1 mcg/kg) were administered, followed by atracurium (0.5 mg/kg) for maintenance. Inhalational sevoflurane (1%) and a mixture of oxygen and air (1:1) were maintained. Hydrocortisone (2 mg/kg) and dexamethasone (0.1 mg/kg) were given. The patient was positioned for the procedure and peak airway pressure was checked and confirmed. An infusion of dexmedetomidine (0.3 mcg/kg) was started and adjusted as needed. Throughout the procedure, intraoperative monitoring of blood pressure, heart rate, EtCO2 and SpO2 was performed. For analgesia, paracetamol (15 mg/kg) and a bilateral maxillary block using 0.125% bupivacaine (2 cc on each side) were administered. During the intraoperative period, the patient received 300 mL of Ringer’s lactate and the estimated blood loss was 80 cc.
After the procedure, haemostasis was confirmed, throat pack was removed and a reversal injection of neostigmine 0.05 mg/kg+injection glycopyrrolate 0.008 mg/kg was given after the baby was awake with adequate respiratory attempts. Then the patient was extubated and turned to lateral position and shifted to Paediatric Intensive Care Unit (PICU) for observation.
Discussion
Patients with PRS who undergo palatoplasty often face a high incidence of airway complications. These complications, including airway obstruction and difficulties with ventilation and intubation, are frequently related to the anatomical features of PRS, such as micrognathia, glossoptosis and a CP, that make airway management challenging. Careful preoperative assessment, vigilant monitoring and having a specialised difficult airway management plan are critical to managing these risks effectively and ensuring safer postoperative outcomes [1].
In this case, the patient presented with micrognathia and a severe form of glossoptosis, leading to significant airway obstruction and feeding difficulties since birth. A history of difficult airway and fiberoptic intubation during a previous surgery was noted. As the patient aged, the severity of airway obstruction decreased. The use of fiberoptic intubation, combined with a well-planned, stepwise approach, facilitated successful case management. Additionally, the maxillary block contributed to a smooth postoperative recovery. More than 3/4 of patients with PRS demonstrate CP, which is typically wide and U-shaped. It has been hypothesised that micrognathia hinders the downward transposition of the tongue and subsequently blocks the elevation and fusion of palatal shelves, resulting in defects wider than Isolated Cleft Palate (ICP). The wider defect of the cleft, along with limited mouth opening due to micrognathia, makes surgical repair and the subsequent wound healing and speech outcomes more challenging [2]. The PRS is believed to affect an estimated 1 in 8500-14000 individuals [3]. Assessing and evaluating any related congenital birth defects or syndromes, such as shared airways and the possibility of challenging intubation. Obtaining detailed information about antenatal care, birth history, developmental milestones, vaccination, congenital heart disease, breathing and feeding patterns is crucial. Performing thorough physical examinations, routine laboratory and radiological investigations is a must. Preparing for a case requires attention to mask ventilation and intubation. Multiple levels of airway obstruction can occur and patients under anaesthesia may experience difficulty in ventilating, oxygenating and intubating. Anaesthesia requires thorough preparation, including checking and preparing the necessary equipment, including the difficult airway and making it ready to use [4].
Micrognathia and glossoptosis can significantly impact both visualisation and access during intubation, especially when endoscopic assistance is not available. Multiple intubation attempts raise the risk of laryngeal oedema. Additionally, patients with PRS may have abnormalities such as underdeveloped laryngeal cartilage or tracheal stenosis, which further complicate intubation and intraoperative airway management [5].
Difficult airway trolley with bag masks, facemasks, oropharyngeal airways, nasopharyngeal airways, supraglottic devices (laryngeal mask airway, I-gel) endotracheal tubes, bougies, alternative laryngoscopes, video laryngoscopes, fiberoptics and cricothyroidotomy set with ventilation equipment should be located at close proximity [6].
The airway in patients with PRS can be secured using various methods, including laryngeal mask airway, fiberoptic scope and retrograde wire [7]. The gold standard for paediatric difficult airways is fiberoptic intubation. The anatomy includes a larynx located in the upper neck, a fragile epiglottis and a tongue that is large compared to the oropharyngeal space. By using fiberoptic intubation, the larynx can be accessed more easily and these features can be compensated for [8]. Dexmedetomidine is a centrally acting alpha 2 receptor agonist decreases central sympathetic outflow by preserving respiratory drive with association of low rate of occurrence of apnea, respiratory depression and airway obstruction and decreases requirement of opioids. These features are highly favourable in paediatric patients with a risk of airway collapse or respiratory depression [9].
Polysomnography (PSG) can be a valuable preoperative tool for patients scheduled to undergo palatoplasty, particularly to identify potential risk factors for complications such as prolonged ICU stay, repeated hospital visits, or airway compromise postoperatively. A preoperative PSG can help assess the presence and severity of OSA or other sleep-related breathing disorders, providing insights that can guide anaesthesia management and postoperative monitoring. Using PSG as part of the preoperative workup for palatoplasty candidates is thus an important step in assessing and mitigating potential risks associated with airway compromise after surgery [10].
Conclusion(s)
This case highlights the critical importance of meticulous planning and execution in managing patients with PRS undergoing surgical interventions like palatoplasty. The presence of complex anatomical abnormalities, such as micrognathia and glossoptosis, necessitates a well-coordinated anaesthetic approach, including fiberoptic intubation and tailored intraoperative care. Patients with PRS often face varying levels of airway obstruction, feeding challenges and frequent upper and lower respiratory infections. A comprehensive preoperative assessment, including ruling out other congenital abnormalities, is crucial to prevent intraoperative and postoperative complications. Having a well-equipped airway cart, ready access to emergency medications and a strategy for managing intubation and postoperative pain are vital. Proactive anticipation of potential issues, swift response to any incidents and consistent follow-up care are essential for optimal outcomes in these patients.
[1]. Amarillo IE, Dipple KM, Quintero-Rivera F, Familial microdeletion of 17q24.3 upstream of SOX9 is associated with isolated Pierre Robin sequence due to position effectAm J Med Genet A 2013 161A(5):1167-72.Epub [2013 Mar 26]. Available from: https://pubmed.ncbi.nlm.nih.gov/23532965/doi:10.1002/ajmg.a.3584710.1002/ajmg.a.3584723532965 [Google Scholar] [CrossRef] [PubMed]
[2]. Cote A, Fanous A, Almajed A, Lacroix Y, Pierre Robin sequence: Review of diagnostic and treatment challengesInt J Paediatic Otorhinolaryngol 2015 79(4):451-64.Epub [2015 Feb 7]. Available from: https://pubmed.ncbi.nlm.nih.gov/25704848/doi:10.1016/j.ijporl.2015.01.03510.1016/j.ijporl.2015.01.03525704848 [Google Scholar] [CrossRef] [PubMed]
[3]. Benko S, Fantes JA, Amiel J, Kleinjan DJ, Thomas S, Ramsay J, Highly conserved non-codingelements on either side of SOX9 associated with Pierre Robin sequenceNat Genet 2009 41(3):359-64.Epub [2009 Feb 22]. Available from: https://pubmed.ncbi.nlm.nih.gov/19234473/doi:10.1038/ng.32910.1038/ng.32919234473 [Google Scholar] [CrossRef] [PubMed]
[4]. Wang C, Shi B, Li J, Management of cleft palate among patients with Pierre Robin sequenceBr J Oral Maxillofa Surg 2023 61(7):475-81.Available from https://www.clinicalkey.com/#!/content/playContent/1-s2.0-S0266435623001626?returnurl=null&referrer=null10.1016/j.bjoms.2023.06.00337453893 [Google Scholar] [CrossRef] [PubMed]
[5]. Association of Anaesthetists of Great Britain and IrelandChecking Anaesthetic EquipmentAnaesthesia 2012 67:660-68.10.1111/j.1365-2044.2012.07163.x22563957 [Google Scholar] [CrossRef] [PubMed]
[6]. Greenland KB, Irwin MG, Airway management- ‘spinning silk from cocoons’Br J Anaesth 2014 69:296-300.10.1111/anae.1262124641634 [Google Scholar] [CrossRef] [PubMed]
[7]. Cladis F, Kumar A, Grunwaldt L, Otteson T, Ford M, Losee JE, Pierre Robin Sequence: A perioperative reviewAnesth Analg 2014 119(2):400-12.10.1213/ANE.000000000000030125046788 [Google Scholar] [CrossRef] [PubMed]
[8]. Jagannathan N, Sequera-Ramos L, Sohn L, Huang A, Sawardekar A, Wasson N, Randomized comparison of experts and trainees with nasal and oral fibreoptic intubation in children less than 2 years of ageBr J Anaesth 2015 114:290-96.10.1093/bja/aeu37025377166 [Google Scholar] [CrossRef] [PubMed]
[9]. Jung SM, Drug selection for sedation and general anesthesia in children undergoing ambulatory magnetic resonance imagingYeungnam Univ J Med 2020 37(3):159-68.10.12701/yujm.2020.0017132299181PMC7384916 [Google Scholar] [CrossRef] [PubMed]
[10]. Costa MA, Murage KP, Tholpady SS, Flores RL, Airway compromise following palatoplasty in Robin sequence: Improving safety and predictabilityPlast Reconstr Surg 2014 134(6):937e-945e.10.1097/PRS.000000000000072925415116 [Google Scholar] [CrossRef] [PubMed]