The OKC was classified as a Keratocystic Odontogenic Tumour (KCOT) in the 2005 classification of World Health Organisation (WHO) head and neck tumours owing to its aggressive behaviour and high recurrence potential [1]. Recently, the lesion has been reclassified within the category of developmental odontogenic cysts [2]. Originally, described in 1876, the term ‘odontogenic keratocyst’ was coined by Phillipsen in 1956 [3]. The cyst is thought to arise from either the epithelial remnants or the basal cells of the oral epithelium [4]. The lesion is typically detected late because it initially grows in an anteroposterior direction at the expense of the medullary cavity [5]. Expansion of the cortices occurs late in the course of progression. Consequently, a comprehensive treatment approach is of utmost importance, requiring complete enucleation with adjuvant peripheral ostectomy, excision of overlying soft tissue, fixation methods and radical treatment in selected cases to prevent recurrence. The recurrence rate of OKC is alarmingly high and has been reported to range from 2.5 to 62.5% [6].
The recurrence potential of OKC has been attributed to several clinical, radiological and histopathological features [7-11]. A recently published systematic review showed that recurrent OKCs had a significantly smaller radiographic diameter and were unassociated with teeth [11]. In contrast, Jung HD et al., found that the size of the lesion was larger in cases showing recurrence compared to non recurrent ones. Additionally, the recurrence potential was more commonly seen in cysts that were multilocular and were managed conservatively [10]. Similar findings were reported by Fidele NB et al., [8]. A few other studies have correlated histopathological features with recurrence in OKC [7,8,11]. Conventionally, satellite cysts and a weak epithelial-connective tissue interface (subepithelial split) are positively associated with recurrence [1,7]. Cottom HE et al., added basal and parabasal mitoses and subepithelial hyalinisation to the list but found no correlation with inflammation in the cyst wall [7]. Although not statistically significant, inflammation was a feature of non recurrent cysts [7]. The histological and clinical data starkly contrast with the findings reported by Pylkkö J et al., [9].
The conflicting data in the literature prompted us to conduct the present study. The study aimed to present a comparison of clinical and pathological features between recurrent and non recurrent OKCs.
Materials and Methods
The present cohort study was conducted in the Department of Oral Pathology and Oral Microbiology at Saveetha Dental College and Hospitals, Chennai, Tamil Nadu, India. It included cases of OKC reported between January 2021 and December 2023, with a maximum follow-up of 36 months. Prior clearance was obtained from the Institutional Human Ethical Committee (IHEC/SDC/UG-2157/24/OPATH/135). During the three years, a total of 2,696 biopsies were received in the department. Detailed data were retrieved from the departmental archival files and the institutional electronic database.
Inclusion criteria: The inclusion criteria for the cases were: a) All cases that were diagnosed and confirmed histologically as OKC; b) Cases that were treated by excision (incisional biopsy specimens were not included); c) Cases with a complete clinical and demographic profile; d) Cases where Formalin Fixed Paraffin Embedded (FFPE) blocks were available for re-evaluation; and e) Cases with complete follow-up data.
Exclusion criteria: Cases where detailed clinical and treatment data were not available, as well as cases that showed malignant transformation or features of epithelial dysplasia, were excluded from the study.
The retrieved cases of OKCs were divided into two groups: Group I - non recurrent OKCs (n=36) and Group II - recurrent OKCs (n=17). Data pertaining to age, gender, site, laterality, radiological findings, specific histological features, treatment and follow-up were retrieved for both groups. Histological criteria for diagnosis included a uniformly thick parakeratinised stratified squamous epithelial lining with tall columnar palisaded basal cells (deeply basophilic nuclei) and surface corrugations. Other specifically tabulated histological parameters included the presence of basal budding (recorded as present/absent), juxtaepithelial hyalinisation (recorded as present/absent), subepithelial split (recorded as present/absent), basal and parabasal mitoses (recorded as present/absent and location), satellite cysts (recorded as present/absent) and the nature and intensity of inflammation in the cyst wall (recorded as none, mild, moderate and intense). In cases where the sections were inadequate for reassessment, either fresh sections were taken or the old slides were restained. All slides were then reassessed independently by two oral pathologists (DP and RPK). A strong inter-rater agreement was noted between the observers (p-value: <0.001), as assessed by Cohen’s kappa statistics. The primary slides of the included recurrent cases were also retrieved and reassessed.
Statistical Analysis
The data were entered into a Microsoft Excel spreadsheet 2021 and analysed using IBM SPSS Statistics for Windows, Version 26.0 (Released 2019; IBM Corp., Armonk, New York, United States). Independent t-tests and Pearson Chi-square tests were used to analyse the data. The Kaplan-Meier curve was used to plot disease (recurrence)-free survival, with recurrence considered as the event. Statistical analysis was performed using the Log Rank (Mantel-Cox) test to compare the study groups based on histopathological features. A p-value <0.05 was considered statistically significant.
Results
Clinicodemographic data: Over three and a half years, 53 satisfactory excised cases of OKC were included after screening 2,696 biopsies. These 53 cases were subdivided into non recurrent (n=36) and recurrent cases (n=17). Two cases of non recurrent OKC had multiple lesions; however, both were non syndromic. The overall mean age was 37.08±16.03 years (Median: 37 years). Individually, the mean age of occurrence for Group I was 38.28±17.37 years (Median: 37 years), whereas it was 34.52±12.83 years for Group II (Median: 35 years). No statistically significant difference was noted between the means of age of the two study groups (p-value >0.05). Overall, there were 33 males and 20 females. The body of the mandible was the most commonly affected site (n=26, 49.1%), followed by the ramus (n=9, 17%) and posterior maxilla and anterior mandible (7 cases each, 13.2%). Two cases each were observed in the angle region and anterior maxilla (3.8% each). The left side was slightly more affected than the right side (28 left: 25 right). Radiologically, the maximum number of cases were multilocular (n=37, 69.8%); 24 cases (66.67%) in Group I and 13 cases (76.47%) in Group II. A comparative analysis of age, gender, location, laterality and radiographic features did not showed any significant difference between the two study groups, as entailed in [Table/Fig-1].
Comparison of clinical features between recurrent and non recurrent OKCs included in the present study.
| Parameter | Non-recurrent OKC (n=36) | Recurrent OKC (n=17) | p-value |
|---|
| Age (in years) | 38.28±17.37 | 34.52±12.83 | 0.432# |
| Gender |
| Male | 24 (66.67%) | 9 (52.94%) | 0.254# |
| Female | 12 (33.33%) | 8 (47.06) |
| Location |
| Body of mandible | 18 (50%) | 8 (47.06%) | 0.892# |
| Angle of mandible | 1 (2.78%) | 1 (5.88%) |
| Ramus of mandible | 6 (16.67%) | 3 (17.65%) |
| Posterior maxilla | 4 (11.11%) | 3 (17.65%) |
| Anterior maxilla | 2 (5.55%) | 0 |
| Anterior mandible | 5 (13.89%) | 2 (11.76%) |
| Laterality |
| Left | 18 (50%) | 10 (58.82%) | 0.381# |
| Right | 18 (50%) | 7 (41.18%) |
| Radiological features |
| Multilocular | 24 (66.67%) | 13 (76.47%) | 0.348# |
| Unilocular | 12 (33.33%) | 4 (23.53%) |
#not significant
Comparison of histological features: All 53 cases exhibited characteristic histopathological features of OKC, including uniformly thick parakeratinised stratified squamous epithelium with a corrugated luminal surface and tall columnar basal cells displaying palisaded, deeply basophilic nuclei. In three cases, focal orthokeratinisation was noted, but most areas were lined by parakeratinisation. The characteristic features were lost in areas of inflammation in the adjacent cyst wall, demonstrating metaplastic changes to non keratinised epithelium. As mentioned, there were 36 cases of non recurrent OKC and 17 cases of recurrent OKC.
The two groups were histopathologically analysed and compared for basal budding, subepithelial hyalinisation, epithelial-connective tissue junction split, basal and parabasal mitoses, satellite cysts and the nature and intensity of inflammation in the cyst wall [Table/Fig-2]. Recurrent cases displayed a significantly higher percentage of basal budding in the cyst lining (p-value <0.001) and dense chronic inflammation in the adjacent wall (p-value <0.001). There was no difference in the number and percentage of cases regarding the subepithelial split, juxta-epithelial hyalinisation, satellite cysts and mitoses, as shown in [Table/Fig-3].
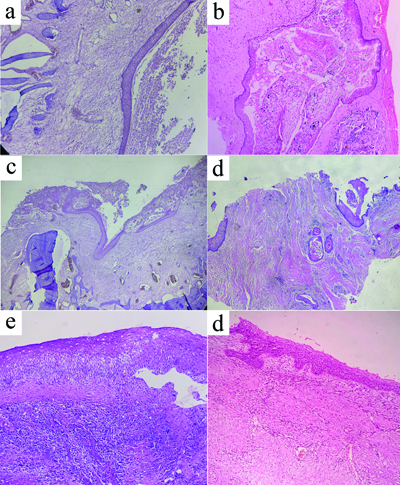
Photomicrographs of H&E stained sections showing a conventional non inflamed OKC with uniformly thick parakeratinised lining and basal palisaded layer (a, 100x); A case of OKC showing metaplastic changes and Rushton bodies and calcifications with focal intense inflammation in the adjacent wall (b, 100X); An inflamed OKC with many areas of non keratinisation (40X) (c); Numerous satellite cysts (d); OKC with intense inflammation in the wall, with loosening of spinous layer and subepithelial split (e) and (f) Basal budding in a case of recurrent OKC with mild inflammatory reaction in the wall.
Comparison of histopathological features between recurrent and non recurrent OKCs included in the present study.
| Parameter | Non-recurrent OKC (n=36) | Recurrent OKC (n=17) | p-value |
|---|
| Juxta-epithelial hyalinisation |
| Present | 25 (69.44%) | 14 (82.35%) | 0.259# |
| Absent | 11 (30.56%) | 3 (17.65%) |
| Basal and parabasal mitoses |
| Present | 5 (13.89%) | 3 (17.65%) | 0.701# |
| Absent | 31 (86.11%) | 14 (82.35%) |
| Inflammation in the wall |
| None | 27 (75%) | 0 | <0.001* |
| Mild | 4 (11.11%) | 1 (5.88%) |
| Moderate | 0 | 0 |
| Intense | 5 (13.89%) | 16 (94.12%) |
| Satellite cysts |
| Present | 25 (69.44%) | 14 (82.35%) | 0.259# |
| Absent | 11 (30.56%) | 3 (17.65%) |
| Basal budding |
| Present | 5 (13.89%) | 16 (94.12%) | <0.001* |
| Absent | 31 (86.11%) | 1 (5.88%) |
| Sub-epithelial split |
| Present | 22 (61.11%) | 12 (70.59%) | 0.362# |
| Absent | 14 (38.89%) | 5 (29.41%) |
#not significant; *statistically significant
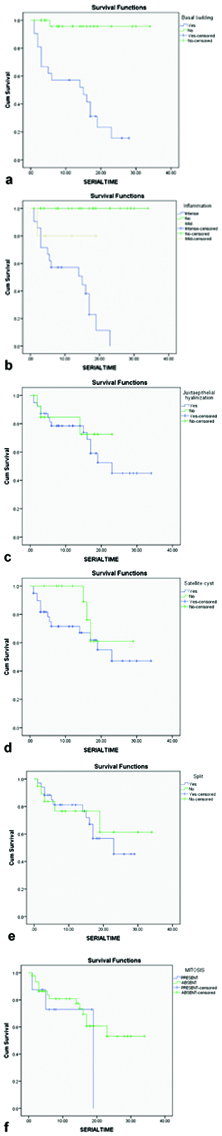
Treatment and follow-up: Most of the recurrent cases (n=14, 82.35%) were conservatively enucleated. Recurrence was considered the event. All cases were followed-up for a maximum of 36 months (Mean: 22.86 months, Median: 23 months, Range: 1-34 months). The disease (recurrence) free survival was compared with individual histological features and the Log-Rank (Mantel-Cox) test indicated that the cases with basal budding and inflammation showed significantly lower DFS than the cases lacking these features (p-value <0.001). Other histological parameters showed no significant difference [Table/Fig-4].
Kaplan–Meier curve demonstrating disease (recurrence) free survival based on the six analysed histopathological features: (a) Basal budding; (b) Inflammatory reaction in the wall; (c) Juxtaepithelial hyalinisation; (d) Satellite cysts, (e) Subepithelial split and (f) Basal and parabasal mitoses.
Discussion
The OKC is one of the most common locally aggressive developmental odontogenic cysts. OKC, considered a cystic neoplasm by many, is one of the components of Gorlin syndrome, which results from mutations in the PTCH1 gene [12]. The same mutation is also noted in about 80% of sporadic cases. A recent systematic review pertaining to the expression of p53 in Dentigerous Cyst (DC), OKC and ameloblastoma found that the expression in OKC was closer to that of ameloblastoma than to that of DC, which led the authors to opine that OKC should be considered a tumour [13]. The latest WHO classification of head and neck tumours accepts the terminology KCOT for OKC [14]. Often discovered incidentally, OKCs show significant bone destruction at the time of diagnosis and display a high recurrence potential. Previous studies have correlated the recurrence of OKC with various clinical, radiological, histopathological and surgical procedures, yielding conflicting results [7-11,15-17]. Thus, the present study was designed in our institution, comprising 53 cases of excised OKC reported in the past three years, with a detailed analysis of clinical, histopathological and follow-up data.
In the present study, stringent inclusion criteria yielded 17 cases of recurrent OKCs and these cases were compared with conventional non recurrent OKCs. Overall, the mean age was 37.08 years, which was slightly higher in non recurrent cases (38.28±17.37 years) than in the cases that recurred subsequently (34.52 years). However, the difference in the mean was not statistically significant. In contrast to the available literature, where most cases of OKC are reported in the second and third decades of life, irrespective of recurrence status, present study reported a peak incidence in the fourth decade [8,18,19]. Similar data have been reported from other states in India and a few other countries [10,20,21]. A possible reason, apart from late cortical expansion, could be a lack of awareness on the part of patients and limited availability of healthcare facilities, particularly in rural areas.
Similarly, no differences were noted in site, laterality, radiographic features and gender between the two groups. In general, a male preponderance was observed, as noted in other studies [8,22]. A reverse trend has been reported in a few studies from Brazil, with more cases reported in females [16,23]. Ubiquitously, the posterior mandible was the most commonly affected area globally. The recurrent cases were reported to be larger and multilocular radiographically compared to the non recurrent cases [8,10,17,24]. Jung HD et al., thus warranted long-term follow-up for any case that is larger than six crowns and displays multilocularity [10]. In contrast, present study did not found any significant difference between any clinical feature and the recurrence potential; in fact, most cases in our series were multilocular.
Out of the six histological features studied and compared, the recurrent cases showed significantly more inflammation in the cyst wall and basal budding of the lining epithelium. The data linking the association of inflammation with recurrence yielded contrasting results. Akin to the present study, a positive correlation was reported by Pylkkö J al., de França GM et al., and Kaczmarzyk T et al., while another group of researchers found no similar association [9,16,17]. Present study noted that cases with inflammation showed a higher prevalence of metaplastic epithelium, as also reported previously [15]. Recurrent cases have been shown to exhibit more inflammation compared to non recurrent cases [9,16,17]. Concomitantly, a higher proliferative activity is noted in recurrent cases [17]. Inflammation plays a significant role in metaplasia, dysplastic changes and carcinogenesis [15,25], which forms the basis of the ‘inflammation-metaplasia-carcinoma’ sequence [26], suggesting the role of immunological factors in the process.
While most studies shown that the presence of satellite cysts is correlated with recurrence, present study could not establish any such association [9,16]. Authors believe that the presence of satellite cysts, along with subepithelial split, could be an inherent biological feature more closely associated with local aggressiveness and expansion [27]. Basal budding was another parameter seen more commonly in recurrent OKCs. This may be considered analogous to tumour budding seen in most epithelial malignancies, which is further related to poorer patient outcomes [28-31].
Cottom HE et al., showed a higher prevalence of juxtaepithelial hyalinisation and basal/parabasal mitoses in recurrent cases [7]. No such association was found in the current study. Juxtaepithelial hyalinisation is not an exclusive feature seen in OKCs and has been reported in other odontogenic cysts and unicystic ameloblastoma [32]. Such changes may merely represent a failed attempt by the odontogenic epithelium to lay down enamel in the absence of ectomesenchymal signals. All the features were plotted on the Kaplan-Meier curve and similar results were found, with recurrence considered as the event. The results were akin to those reported by de França GM et al., [16]. Complete excision of the lesion with peripheral ostectomy and wide surgical excision, in selected cases, appears to be the most appropriate treatment for the management of OKC. As the lesion shows a high chance of dysplastic changes and malignancy, long-term follow-up is essential [1,33].
Limitation(s)
Although a detailed comparison of various clinical and histological features was made between recurrent and non recurrent OKCs, there are a few limitations to the current study. Firstly, the number of recurrent cases was comparatively smaller than the number of non recurrent OKC cases included. Secondly, as the cases were retrieved over a long period, there may be confounding factors related to the type of treatment administered, differences in the operating surgeons and variability in the method of cauterisation. Furthermore, the data was retrieved from a single institution, highlighting the need for more multicentric studies to validate the results. Long-term follow-up is mandatory.
Conclusion(s)
In conclusion, recurrent OKCs showed no differences in age, gender, location and radiographic features compared to non recurrent cases. They are most commonly found in males, with a high predilection for the posterior parts of the mandible. Chronic inflammation and basal budding appear to be independently associated with recurrence in OKC; thus, they must be prioritised in incisional biopsies as prognosticators in OKCs.
#not significant
#not significant; *statistically significant