Advancements in bonding materials used to attach orthodontic brackets, restorations and other components have caused significant changes in the field of dentistry. Orthodontics, as a branch of dentistry, has dramatically evolved over the past few years to improve treatment quality. Orthodontic treatment is particularly sought after by today’s youth to enhance aesthetics and maintain better oral hygiene. WSL, one of the most common iatrogenic effects of orthodontic therapy, arise due to plaque accumulation, which causes decalcification of the enamel surface adjacent to fixed orthodontic appliances, thereby reducing bond strength [1].
Despite the high aesthetic advantage of conventional adhesives, they have a significant tendency to accumulate plaque and cause enamel demineralisation, leading to prolonged treatment durations. This not only consumes more chairside time but also increases treatment costs, resulting in compromised aesthetics due to enamel chipping caused by decalcification [2]. To address this issue, modifications in the adhesive are necessary to mitigate these side-effects without compromising the bond strength of the material.
Nanotechnology represents one of the most recent advancements in the field of dental materials. The incorporation of nanoparticles into resin composites has enhanced bond strength by increasing the surface area per unit mass, thereby improving the efficiency of the bonding material. Nanoparticles offer several advantages over conventional adhesives currently in use, including improved polymerisation shrinkage, tensile and compressive strength, and reduced marginal leakage. This concept has been applied to dental adhesive materials, where nanoparticles are added as fillers to fill the microgaps between commonly present macro/micro fillers in dental adhesives [3].
Titanium dioxide is a non toxic, biocompatible inorganic filler that possesses mechanical, optical and optimal antibacterial properties, available in various crystalline forms and sizes [4]. TiO2 nanocomposites have demonstrated excellent antimicrobial properties against Streptococcus mutans, the primary organism responsible for WSLs. This advantage, combined with better bond strength, can be utilised to prevent enamel decalcification [5].
While it is essential to prevent WSLs, adhesive bond strength remains paramount for successful clinical practice. Continuous failure of fixed appliances increases treatment costs and prolongs treatment time. Some studies have shown a significant decrease of at least 3 MPa or more in shear bond strength following TiO2 incorporation [6,7]. Previous studies involving other nanoparticles, such as Zirconium Dioxide (ZrO2), have shown improved shear bond strength, and investigations into the use of TiO2 nanoparticles have indicated no significant difference in shear bond strength compared to conventional composites [5,8-10].
Therefore, the purpose of the present study was to evaluate the shear bond strength of orthodontic adhesive incorporated with TiO2 nanoparticles.
Materials and Methods
The present in-vitro study was designed and conducted in the Department of Orthodontics and Dentofacial Orthopaedics, Best Dental Science College, Madurai, Tamil Nadu, India, between July 2022 and December 2023. Ethical clearance was obtained from the Institutional Ethical Committee (BDSC-IEC2022-STU-BrV-AQD-22). Informed consent was obtained from the patients who required upper premolar extraction for orthodontic treatment.
The inclusion and exclusion criteria for the study are as follows:
Inclusion criteria:
Maxillary first premolar teeth extracted for orthodontic therapy.
Teeth without caries and WSLs.
Teeth with all surfaces intact.
Teeth with no cracks or fractures.
Exclusion criteria:
Teeth with fluorosis.
Restored teeth.
Hypoplastic enamel.
Developmental defects.
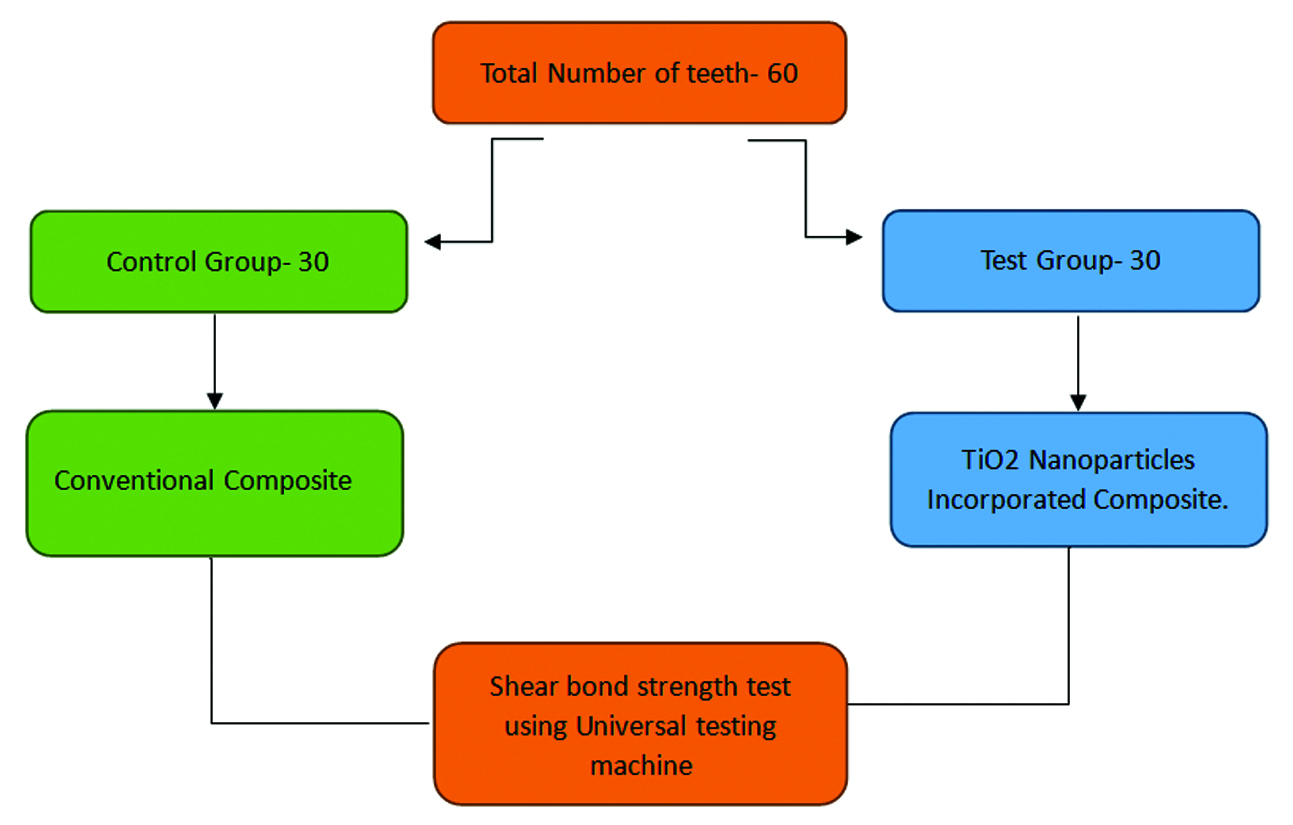
Sample size calculation: The required sample size was estimated from a previous study [5] using G*Power software version 3.1 (power=0.80), resulting in a total of 60 teeth used in the study [Table/Fig-1]. Based on the inclusion and exclusion criteria, a sample of 60 non carious, non fluorosed extracted maxillary first premolar teeth was collected from the study Institute.
Sample size distribution.
Study Procedure
All the teeth were cleaned with pumice for five seconds, rinsed in water for ten seconds, and then air-dried. The buccal surface of the teeth was etched with 37% orthophosphoric acid gel for thirty seconds, rinsed with water for ten seconds, and air-dried until a white, chalky surface appeared. The primer (Transbond TM XT, 3M Unitek, Monrovia, California) was applied to the etched surface in a gingival-occlusal direction and cured for fifteen seconds. MBT stainless steel brackets with a slot size of 0.022×0.028 inches were used, and each tooth was bonded with the composite and cured for 40 seconds [Table/Fig-2,3,4 and 5].
Sample size distribution.

Application of 37% orthophosphoric acid on the buccal surface of the tooth.
Application of primer on the buccal surface of the tooth.
Light curing of the bracket for 40 seconds.
Based on the type of adhesive used, the samples were divided into two groups:
Group 1: Conventional composite group (Control).
Group 2: 1% TiO2 nanocomposite group (Test).
Preparation of nanocomposite: To create a mixture of 1% TiO2 nanocomposite, the amount of composite required for each tooth was estimated based on clinical experience. A conventional composite (Transbond TM XT; 3M Unitek, Monrovia, California, USA) weighing 245 mg (approximately 2.5 mg of composite required per tooth×10 teeth) was manually mixed with 5 mg of 1% TiO2 nanoparticle powder (Ultrananotech TiO2 nanoparticles, Purity-99.9%, APS-30-50 nm, Bengaluru, India) on a glass slab in a semi-dark room, away from direct light, to achieve a homogeneous mixture of 250 mg (245 mg of Transbond TM XT+5 mg of TiO2 powder=250 mg TiO2 nanocomposite) for a sample of 10 teeth. This amount was then multiplied by 3 to obtain 750 mg of nanocomposite for a total sample of 30 teeth [Table/Fig-6a,b] [11,12].
a,b) Weighing of TiO2 powder using electronic balance machine and mixed with the conventional composite resin.

Debonding procedure: The Instron 3000 universal testing machine was used to analyse the debonding force of the bracket, in which the crosshead of the machine was moved at a uniform speed of 1 mm/minute. An acrylic block was prepared, and the tooth was mounted in a vertical direction with the help of a jig. The acrylic was covered up to the cementoenamel junction of the tooth to ensure equal distribution of force. The tooth was positioned in the lower crosshead of the Instron machine with the tooth crown facing upwards. The necessary force to debond the bracket was applied in an occlusal-gingival direction parallel to the bracket base. The shear bond strength was evaluated using the unit of Megapascals (MPa) [Table/Fig-7,8].
Mounting of tooth in acrylic blocks.

Debonding of the brackets using Instron 3000 universal testing machine.

After bracket debonding, the ARI score [7] was calculated under a 10× microscope according to the following criteria:
Score 0: Absence of composite on the enamel surface.
Score 1: Less than 50% of composite remains on the enamel surface.
Score 2: More than 50% of composite remains on the enamel surface.
Score 3: The entire composite remains on the enamel surface, with the impression of the bracket base on the remaining composite.
According to Artun J and Bergland S (1984), a score of 3 was considered indicative of better bond strength because the fracture occurs at the bracket-adhesive interface. However, there is a difficulty in removing the adhesive, which might result in some enamel being removed from the surface, potentially mistaken for composite due to its transparent nature [13]. Therefore, a score of either 1 or 2 is considered safe for removal without damaging the enamel surface.
Statistical Analysis
The obtained data values were entered into Microsoft Excel 2013 and statistically analysed using Statistical Package for Social Sciences (SPSS) software version 22.0, Chicago, Illinois. A Student’s t-test was conducted to estimate the difference in the mean compressive stress at the maximum debonding force required for the two groups, and a Chi-square test was performed to evaluate the ARI scores between the two groups. The significance level was set at a p-value of less than 0.05.
Results
The highest bond strength was observed in the TiO2 nanocomposite (Mean±SD: 14.3±1.21 MPa) compared to the conventional composite (Mean±SD: 12.9±1.05 MPa), with a mean difference of 1.4 MPa that was statistically significant (p-value=0.045) [Table/Fig-9].
Comparison of shear bond strength between conventional and TiO2 nanocomposite after bonding.
| Composite | Shear bond strength (Mpa) |
|---|
| Conventional composite (mean±SD) | 12.9±1.05 |
| TiO2 nanocomposite (mean±SD) | 14.3±1.21 |
| p-value | 0.045 |
The results from the Chi-square test indicated that ARI score 1 (<50% of adhesive remnant on the tooth) was more prevalent in the conventional composite group, suggesting a higher occurrence of adhesive bond failure with less adhesive remaining on the enamel. In contrast, score 2 (>50% of the adhesive remnant on the tooth) was greater for the nanocomposite group, indicating a cohesive bond failure that left adhesive remnants on both the enamel surface and the bracket. The difference in the ARI scores between the two groups was statistically not significant (p-value=0.682) [Table/Fig-10].
Frequency of ARI scores after debonding of brackets.
| Adhesive Remnant Index (ARI) | p-value |
|---|
| ARI score | 1 | 2 | 3 |
|---|
| Conventional composite, n (%) | 16 (53.3) | 9 (30) | 5 (16.6) | χ2=0.764
0.682, NS |
| TiO2 nanocomposite, n (%) | 6 (20) | 17 (56.6) | 7 (23.3) |
NS: Non significant
Discussion
The present study assessed the shear bond strength of an orthodontic adhesive incorporated with TiO2 nanoparticles. The results revealed a significant increase in the bond strength of the TiO2 nanocomposite compared to that of the conventional composite.
The introduction of the etching technique by Buonocore and Bisphenol A-glycidyl Methacrylate (Bis-GMA) by Bowen has played a major role in the successful bonding of orthodontic fixed appliances [14]. Despite the use of nanotechnology in other scientific fields, it has recently become popular in the field of orthodontics for efficient bonding. Nanoparticles have been proven to increase the strength of materials by increasing the surface area; this concept has been used to enhance the properties of adhesives [15]. TiO2 is highly attractive for its light-scattering properties and very high refractive index, allowing low levels of the pigment to achieve a white, opaque coating with good mechanical properties [4]. Effective bonding of orthodontic brackets to the enamel surface of the teeth is necessary to save chairside time and improve operator efficiency, which requires excellent materials that provide superior bonding properties. According to the literature, the ideal bond strength of the material is considered to be between 6 Mpa and 10 to resist functional occlusal forces [14].
The present study showed that adding TiO2 as a nanofiller enhanced the bond strength of the nanocomposite compared to the conventional composite (p-value <0.05). The results of the present study contrast with those of Poosti M et al., which indicated that there was no significant difference observed between the nanocomposite and conventional composite (p-value=0.58) [5]. Sodagar A et al., concluded in their study that a nanocomposite used with TiO2 nanoparticles exhibited better bond strength when used at a 1% concentration [12]. However, Elsaka SE et al., concluded in their study that TiO2 nanocomposites provided better mechanical and antibacterial properties, thus enhancing bond strength [16].
The differences observed may be due to small sample sizes, methodological variations, different bonding materials and the standardisation of particle sizes, as sizes greater than the threshold value can affect the adherence of the composite to the tooth material, potentially impacting the outcomes across various studies. Therefore, a larger sample size was used in the present study. Another reason for the differences in bond strength could arise from variations in filler content. A higher filler concentration leads to less polymerisation shrinkage and a higher degree of cure [8]. Transbond TM XT was used in the present study since it is the most widely used composite for comparing different bonding materials for bond strength values. Similar studies from the literature have been tabulated in [Table/Fig-11] [5-7,9,10,12,17].
Literature review of Shear Bond Strength (SBS) using nanoparticles [5-7,9,10,12,17].
| Author’s name | Place of study | Sample size | Materials compared | Parameters assessed | Conclusion |
|---|
| Present study | South India | 60 extracted human maxillary 1st premolars | Transbond TM XT and 1% concentration of TiO2 nanocomposite | Shear bond strength and ARI | TiO2 nancomposite enhanced the bond strength. |
| Ekhlassi S et al., 2011 [17] | USA | 180 bovine central incisors | Transbond TM plus self-etching primer, Grengloo and Transbond TM XT | Shear bond strength and ARI scores | Transbond TM XT had highest bond strength of 14.7 MPa and ARI score of 3. |
| Poosti M et al., 2013 [5] | Iran | 30 extracted human premolars | Trandbond XT and 1% (w/w) of TiO2 nanocomposite particle size-21±5 nm. Purity 99.5% | SBS and ARI scores, antibacterial property | No significant difference in SBS and ARI between the two groups. TiO2 has significant antibacterial property. |
| Reddy AK et al., 2016 [6] | India | 120 extracted human premolars | Transbond TM XT and 1% Ag, 1% TiO2 and 1% ZnO nanocomposites | SBS | Transbond TM XT had better bond strength than all the nanocomposites. |
| Sodagar A et al., 2017 [12] | Iran | 48 bovine central incisors composite disks | Transbond TM XT containing 0%, 1%, 5% and 10% (w/w) of TiO2 nanocomposite preparation+Transbond TM XT | Shear bond strength and antimicrobial test for streptococcus mutans, Streptococcus sanguinis and Lactobacillus | Shear bond of composite containing 1% and 5% NPs are in acceptable range. TiO2 has antimicrobial properties. |
| Behnaz M et al., 2018 [7] | Iran | 120 extracted human premolars | Trandbond XT resilience and Anatase 0.1 wt% concentration of TiO2 nanocomposite | SBS and ARI scores | Adhesive without TiO2 had a significantly higher mean SBS than composites containing TiO2 but bond strength was in acceptable range. No significant difference in the ARI scores. |
| Al-Saleh S et al., 2021 [9] | Saudi Arabia | 75 extracted maxillary premolars | Experimental adhesive (containing Bis-GMA, TEGDMA, HEMA camphroquinone, benzoate), 5% TiO2 and ZrO2 nanocomposite | SBS and resin dentin interface failures | 5% TiO2 showed highest bond strength (25.35±1.5 MPa) than ZrO2 (23.1±2.2 MPa) and experimental adhesive (21.03±2.4 MPa). TiO2 had good resin tag formation. |
| Prasad T et al., 2024 [10] | India | 40 extracted human premolar teeth | TransbondTM XT and 1% resveratrol, 1% Zno, 1% Ag nanocomposite | SBS | TransbondTM XT had the highest bond strength (8.6 Mpa) with other nanocomposites having acceptable bond strength. |
TEGDMA: Triethylene glycol dimethacrylate; HEMA: 2-Hydroxyethyl methacrylate; SBS: Shear bond strength
Debonding the bracket carries a risk of chipping the enamel surface, which may lead to enamel loss. Therefore, it is considered clinically safe to remove the remnant composite from the enamel surface after debonding to prevent enamel loss [7]. Consequently, the ARI score is regarded as a reliable method for assessing the adhesive-tooth and adhesive-bracket interfaces. ARI scores, compared using the Chi-square test, showed no significant difference between the groups (p-value >0.05).
In the case of the conventional composite, score 1 was higher, indicating more adhesive bond failure with less adhesive on the enamel. In contrast, in the TiO2 nanocomposite, score 2 was higher, suggesting a cohesive bond failure that left adhesive remnants on both sides, i.e., the enamel surface and the bracket. A study by Uysal T et al., showed no significant results in ARI scores between Transbond TM XT, Filtek Supreme Plus nanocomposite and KetacTM N100 nano-ionomer, all of which contain fluoroaminosilicate glass and nanofillers known to release fluoride for caries inhibition and improve mechanical properties [3]. According to Nagar N and Vaz AC, there were no significant differences in the ARI scores between Ceram·X® mono and Transbond TM XT [18].
The incorporation of TiO2 nanoparticles into orthodontic adhesives has substantial clinical relevance. Enhanced bond strength reduces the frequency of bracket failures, thereby minimising the need for rebonding and associated interruptions in treatment. This not only optimises clinical efficiency by reducing chairside time but also improves the patient experience by decreasing the overall treatment duration. Moreover, stronger bonding facilitates better control of orthodontic forces, contributing to more precise and reliable tooth movement. This benefit of improved bond strength enhances both the aesthetic and functional outcomes of orthodontic care, ensuring higher long-term success rates. The forces generated in the present study may not accurately reflect the forces produced by jaw movements, which typically fall within the range of 81-100 mm/sec. This range is comparatively lower than that measured by universal testing machines [19].
Limitation(s)
Though the results of the present study showed adequate bond strength between the two composites, there are certain limitations. For instance, the study was conducted in-vitro, where the bond strength of the material cannot be generalised to clinical situations. The strength of the material may differ among various brands of composites that are commercially available and routinely used in clinical cases. Additionally, the forces generated in the study may not accurately reflect the forces produced by jaw movements.
Conclusion(s)
The addition of TiO2 nanocomposites has improved bond strength compared to conventional composites. This innovation not only contributes to more durable orthodontic bonding but may also reduce clinical failures, chair time and overall treatment duration. Therefore, TiO2 nanoparticles can be used as filler particles to increase the efficiency of bonding. Research on nanocomposite materials is quite extensive in dentistry to enhance bond strength. However, studies regarding nanocomposites are still in the initial stages and remain controversial, as they have been shown to possess antimicrobial properties alongside varying bond strengths. Therefore, clinical trials using appropriate nanoparticle concentrations are required to prove the efficacy and efficiency of the material’s strength in clinical conditions.
NS: Non significant
TEGDMA: Triethylene glycol dimethacrylate; HEMA: 2-Hydroxyethyl methacrylate; SBS: Shear bond strength