Introduction
Parkinson’s Disease (PD) is a neurodegenerative disorder that affects both motor and non motor brain pathways. It is the world’s second most common neurodegenerative illness, with an increasing incidence. The PD results in slow movement, tremors, involuntary movements, difficulty walking, and imbalance. The focus of physical therapy treatment is to improve balance, postural stability, and walking. The use of walking aids promotes stability, and cues help individuals with PD improve their stride. External cues provide temporal or spatial inputs related to the initiation and cessation of motor activities.
Aim
To evaluate the effectiveness of cueing strategies and walking aids in patients with PD and their impact on gait and balance.
Materials and Methods
The interventional study was carried out at Adarsh Chander Medicare Centre, Rohtak, Haryana, India for six weeks, from March 2023 to May 2023. The participants were divided into two groups, A and B, with 30 participants in each group. Participants in Group A were trained with walking aids, while those in Group B were trained with cueing strategies. Pre- and post-training assessments were conducted using the Unified Parkinson’s Disease Rating Scale (UPDRS), the Berg Balance Scale (BBS), and a measuring tape to evaluate step length, stride length, balance, and gait. Data analysis was performed using Statistical Package for Social Sciences (SPSS) version 20.0. Mean values were compared using a t-test (for two groups). A p-value of less than 0.05 was considered statistically significant.
Results
The mean age of Group A was 67.60±4.80 years, while the mean age of Group B was 69.40±5.63 years. The pretraining UPDRS reading for Group A was 152.43±12.55, while for Group B, it was 153.00±15.14 (p=0.87). The post-training reading for Group A was 144.27±12.43, and for Group B, it was 140.43±15.07 (p=0.28). The pre-training BBS reading for Group A was 28.20±5.68, and for Group B, it was 28.17±4.72 (p=0.98). Post-training, Group A had a reading of 32.63±5.76, and Group B had 38.80±4.35 (p=0.01). There was also a significant improvement in step and stride lengths in Group B compared to Group A.
Conclusion
There was a notable enhancement in balance and gait among individuals who engaged in an exercise program across both groups. However, the group that underwent exercise training with cueing strategies exhibited more favorable outcomes compared to the group that solely used walking aids. Therefore, exercise training incorporating cueing strategies may present a promising treatment approach for individuals with PD.
Auditory cues, Balance, Stability, Tremors, Visual cues
Introduction
The PD is a neurodegenerative disorder that affects both motor and non motor brain pathways [1]. It is a central nervous system degenerative condition in which there is a loss of dopamine-generating cells in the substantia nigra, an area of the midbrain, which causes the motor symptoms of the disease.
The PD is a multi-system neurological illness that primarily affects patients in their later years of life. It is the world’s second most common neurodegenerative illness, with increasing incidence and prevalence as a result of changing demographics [2]. Among developed countries, the prevalence of PD is estimated to be 0.3% of the total population and roughly 1% in those over 60-years-old [3]. The prevalence in both men and women increases with age, with no decline observed as they grow older [4].
The morbidity of patients with PD is quite high and dramatically progresses within the elderly population. Patients with PD experience problems with gait and gait-related activities, such as balance control and postural instability. Poor balance, gait, and other symptoms of Parkinson’s affect the activities of daily life for these patients. The conventional treatment method uses walking aids for gait training related to the disease [5]. Previous research shows that cues are effective for motor and gait training. Rhythmical cues associated with exercises and motor training can facilitate anticipatory and compensatory reactions, improving gait and preventing falls [6]. Auditory cues set at 7.5% and 15% faster than a person’s preferred stepping frequency improve walking speed, while stride length remains unchanged [7]. Floor stripes, as visual cues, help stabilise stride length in people with PD, reducing variability in their walking patterns [8]. Cues help individuals with PD improve their stride. External cues provide temporal or spatial inputs connected with the start and stop of a motor activity [9,10]. Physical therapy focuses on exercises to improve balance, posture, walking, and daily tasks. The program includes relaxation, stretching, mobility training, posture correction, balance routines, gait improvement, and incorporates music, dance, and speech therapy [11].
The findings of a study demonstrated that both freezers and non freezers experienced improvements in lower extremity variability following Stepping in Place (SIP) training. Incorporating audio cues into the training also had moderating effects on the excitability of the cortex [12]. Another study found that people with PD and age-matched healthy adults increased their pedaling rate with auditory cues compared to their baseline. These cues are also known to enhance the self-perception of motion [13]. Additionally, a study showed that subjects could use an attentional strategy to increase step amplitude; they were able to combine a rhythmic auditory cue with an attentional strategy during single and dual tasks [14]. A study involving ten Parkinson’s patients who could walk independently found that using visual cues during walks led to significant changes in their stride and step lengths compared to walks without cues that immediately followed [15]. Acoustic and visual cueing also improved the amplitude of the initial loading and unloading forces [16].
Since, cues and walking aids assist with walking and balance, the study aimed to optimise rehabilitation outcomes by determining whether cueing strategies or walking aids provide superior improvements in gait, balance, and mobility. This can help create personalised treatment plans, potentially reducing healthcare costs while enhancing patient independence and quality of life. Furthermore, it aids in determining which intervention yields better outcomes in terms of enhancing gait performance, stability, and overall mobility in patients with PD. Therefore, the present study aimed to assess the effectiveness of using cueing strategies and walking aids in patients with PD.
Materials and Methods
This was an interventional study conducted from March 2023 to May 2023 at Adarsh Chander Medicare Centre in Rohtak, Haryana, India. Subjects were informed about the study, and consent was obtained. The present study adhered to the tenets of the Declaration of Helsinki.
Sample size: A total of 60 patients were recruited for the study through purposive sampling.
Inclusion criteria: Patients aged between 50 and 80 years were selected using a convenience sampling technique. Participation of the subjects was based on loss of balance, mobility issues, and impaired gait due to PD.
Exclusion criteria: Patients with any untreated hearing or vision impairment, any terminal illness, or any other neurological conditions or co-morbidities were excluded from the study.
Subjects were assessed for balance using the BBS [17] and for the severity and progression of PD using the UPDRS [18]. Step and stride lengths were measured using ink and a measuring tape for gait analysis.
Study Procedure
Subjects were divided into two groups, with 30 participants in each, namely Group A and Group B. Each patient attended a one-hour training session four times a week for six weeks. Pre- and post-training assessments were conducted using a measuring tape for step and stride length, the BBS [17] for balance, and the UPDRS [18] for the severity and progression of PD.
Group A received an exercise program that included stretching exercises for the lower limbs using a hold-and-relax technique. This program incorporated Proprioceptive Neuromuscular Facilitation (PNF) techniques to reduce rigidity. Patients in this group maintained an active lifestyle with active range-of-motion exercises to improve flexibility. Frenkel’s exercises, consisting of three sets of 15 repetitions, were practiced to enhance coordination and balance [Table/Fig-1a]. Gait training was performed with the assistance of walking aids (canes and frames) [Table/Fig-1b,c].
a) Group A performing Frenkel’s exercise; b) Group A walking with cane; and c) Group A walking with frame; d) Group B performing Frenkel’s exercise; e) Group B walking with assistance of auditory cue (by counting 1,2,3 and start/rest); and f) Group B walking with assistance of visual cue (patient was asked to walked within the strips).
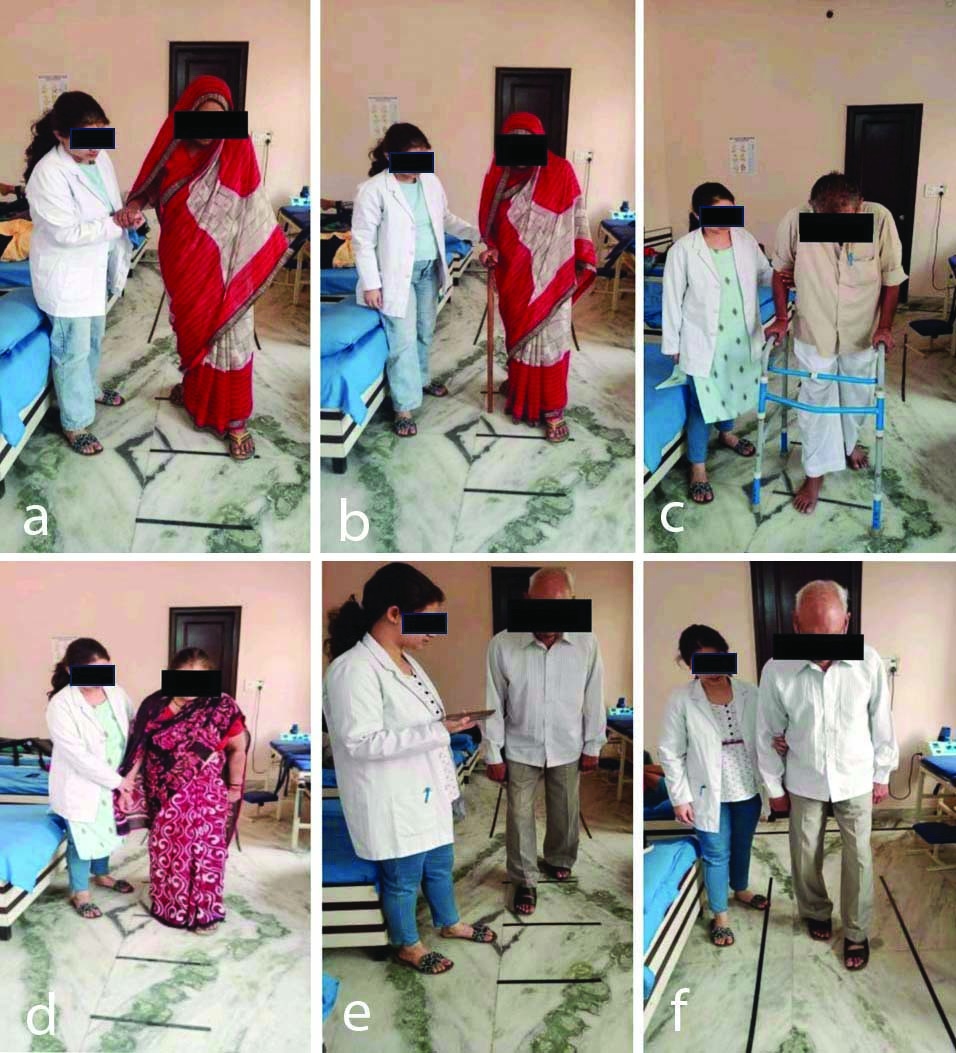
Participants in Group B practiced the same exercise regimen as Group A [Table/Fig-1d]. During balance and gait training, auditory and visual cues were provided to the patients. Auditory cues were delivered verbally (by counting 1, 2, 3, and start/rest) to maintain the rhythm of walking. Visual cues were given using strips on the floor, with the patient being instructed to walk within the strips [Table/Fig-1e,f].
Post-training assessments for step length, stride length, balance, and gait were conducted using a measuring tape, the BBS, and the UPDRS.
Statistical Analysis
The statistical analysis was conducted using IBM’s Statistical Package for the Social Sciences (SPSS) version 20.0. Intergroup comparisons were performed using an unpaired t-test. A p-value of ≤0.05 was considered statistically significant.
Results
The mean age of Group A was 67.60±4.80, while the mean age of Group B was 69.40±5.63, with no statistically significant difference between the groups (p-value 0.18) [Table/Fig-2].
Comparison of age within the groups.
| Group | Mean±SD | p-value |
|---|
| Age (years) | Group A | 67.60±4.80 | 0.18 |
| Group B | 69.40±5.63 |
Comparison of age within the groups
The mean and Standard Deviation (SD) of the UPDRS pretest score were 152.43±12.55 for Group A and 153.00±15.14 for Group B (p-value=0.87), while the post-test score was 144.27±12.43 for Group A and 140.43±15.07 for Group B (p-value=0.28) [Table/Fig-3].
Comparison of pre and post-treatment UPDRS test scores.
| Group | Mean±SD | p-value |
|---|
| UPDRS-pretest score | Group A | 152.43±12.55 | 0.87 |
| Group B | 153.00±15.14 |
| UPDRS-post-test score | Group A | 144.27±12.43 | 0.28 |
| Group B | 140.43±15.07 |
*Significant at p≤0.05 (t-test)
The mean and SD of the BBS pretest score were 28.20±5.68 for Group A and 28.17±4.72 for Group B (p-value 0.98), while the post-test score was 32.63±5.76 for Group A and 38.80±4.35 for Group B, showing a significant difference between the groups (p-value 0.01) [Table/Fig-4].
Comparison of pre and post-treatment BBS test score.
| Group | Mean±SD | p-value |
|---|
| BERG balance pretest score | Group A | 28.20±5.68 | 0.98 |
| Group B | 28.17±4.72 |
| BERG balance post-test score | Group A | 32.63±5.76 | 0.01* |
| Group B | 38.80±4.35 |
*Significant at p≤0.05 (t-test was used); BBS: Berg balance scale
The mean and SD of the step length pretest score were 28.57±6.43 for Group A and 26.80±5.45 for Group B (p-value 0.25), while for the post-test, it was 34.27±6.58 for Group A and 38.00±5.43 for Group B, indicating a significant difference between the groups (p-value 0.02) [Table/Fig-5].
Comparison of pre and post-treatment step length test scores.
| Group | Mean±SD | p-value |
|---|
| Step length pretest score | Group A | 28.57±6.43 | 0.25 |
| Group B | 26.80±5.45 |
| Step length post-test score | Group A | 34.27±6.58 | 0.02* |
| Group B | 38.00±5.43 |
*Significant at p≤0.05 (t-test)
The mean and SD of the stride length pretest score were 30.43±6.02 for Group A and 29.70±5.39 for Group B (p-value 0.62), while the post-test score was 38.30±6.54 for Group A and 41.90±5.61 for Group B (p-value 0.02) [Table/Fig-6].
Comparison of pre and post-treatment stride length test scores.
| Group | Mean±SD | p-value |
|---|
| Stride length-pretest | Group A | 30.43±6.02 | 0.62 |
| Group B | 29.70±5.39 |
| Stride length-post-test score | Group A | 38.30±6.54 | 0.02 |
| Group B | 41.90±5.61 |
*Significant at p≤0.05 (t-test was used)
Discussion
The PD is a neuromuscular disorder that affects neurotransmitter pathways in the brain. This study compares the effectiveness of cueing strategies versus walking aids on balance and gait in individuals with PD. Balance and gait change with the progression of the disease in people with PD. The aim of present study was to find and compare the effects at the end of the treatment session between the two groups, which showed equal benefits with respect to balance, gait, and mobility; however, the findings revealed a noteworthy immediate improvement in balance, gait, and mobility in the group that received exercise training with cueing aids.
The present study was conducted to determine which rehabilitation technique is more effective for various parameters in patients with PD. The study concluded that the group involved in exercise training with cueing strategies showed better improvement in gait and severity of the disease, as measured by the UPDRS, and the findings of the BBS indicated that balance improved noticeably more for participants engaged in exercise training with cueing strategies than for those using only walking aids. The results demonstrated that exercise training with cueing strategies led to better improvements in balance and gait in individuals with Parkinson’s.
A similar study was conducted on balance training with rhythmical cues to improve and maintain balance control in PD. The researchers developed an exercise program to promote balance using auditory cues. The results of present study showed that rhythmical cues associated with exercises and motor training can facilitate anticipatory and compensatory reactions, improving gait and preventing falls [6].
The possible reason for these outcomes may be that cueing strategies provide prompts that guide movement, helping individuals overcome difficulties in initiating and executing actions. Auditory cues assist individuals in regulating their movement timing and synchronising their steps, resulting in smoother and more coordinated movement patterns. Additionally, research by Calabro RS et al., indicated that rhythmic auditory stimulation training offers additional advantages in terms of overall gait quality, balance, and the number and length of strides compared to non rhythmic auditory stimulation [19].
The measuring tape showed that both step length and stride length improved in both groups, but the group that was trained with cueing strategies demonstrated more promising outcomes. Similar results were observed in a study conducted by Baker K et al., [14]. The main findings of present study indicated that the attentional strategy and the combination of a rhythmic auditory cue with an attentional strategy were equally effective and significantly improved walking speed and step amplitude during both single and dual tasks [14]. A possible reason for this could be that external cues can activate different neural pathways than those typically used for spontaneous movement. This activation can help compensate for the impaired internal motor control mechanisms in PD. For instance, rhythmic auditory cues can stimulate the brain’s motor control regions and enhance movement timing and coordination. An identical study on the effect of visual cues on the gait of independently mobile PD patients showed statistically significant changes in relative stride length and step length when walking with visual cues [15]. Similar results were found in a study that showed visual cue training was more effective than conventional training alone in improving gait and walking velocity in chronic stroke patients [20].
The present study provides a basis for using visual and auditory cues for training as an additional treatment approach to improve the gait and balance of Parkinson’s patients in clinical settings. Moreover, this method requires only minimal investment, as it relies on devices with basic technology that are easily accessible and inexpensive on the market. Cueing strategies combined with exercise training significantly improved balance and gait in patients with PD who had impaired balance and gait.
Limitation(s)
The sample size was not calculated statistically, and long-term follow-up of patients was not conducted in the present study. Future studies could be conducted with a larger sample size and a longer follow-up period.
Conclusion(s)
There was a significant improvement in balance and gait among people who participated in the exercise program in both groups. Furthermore, cueing strategies showed greater improvement in balance, gait, and other symptoms of PD. Additionally, future studies are necessary to explore the long-term effects and to understand the factors that influence the study outcomes. A longer intervention period should be conducted with a follow-up period to determine whether or not the training’s effects may be long-lasting. By including more modalities or parameters in the exercise training of the study, the significance of the findings can be improved.
Comparison of age within the groups
*Significant at p≤0.05 (t-test)
*Significant at p≤0.05 (t-test was used); BBS: Berg balance scale
*Significant at p≤0.05 (t-test)
*Significant at p≤0.05 (t-test was used)
Author Declaration:
Financial or Other Competing Interests: None
Was Ethics Committee Approval obtained for this study? Yes
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Mar 06, 2024
Manual Googling: Nov 28, 2024
iThenticate Software: Nov 30, 2024 (14%)
[1]. Alexander GE, DeLong MR, Strick PL, Parallel organization of functionally segregated circuits linking basal ganglia and cortexAnnu Rev Neurosci 1986 9:357-81.10.1146/annurev.ne.09.030186.0020413085570 [Google Scholar] [CrossRef] [PubMed]
[2]. Pringsheim T, Jette N, Frolkis A, Steeves TD, The prevalence of Parkinson’s disease: A systematic review and meta-analysisMovement disorders: Official Journal of the Movement Disorder Society 2014 29(13):1583-90.Available from: https://doi.org/10.1002/mds.2594510.1002/mds.2594524976103 [Google Scholar] [CrossRef] [PubMed]
[3]. de Lau LM, Breteler MM, Epidemiology of Parkinson’s diseaseThe Lancet Neurology 2006 5(6):525-35.Available from: https://doi.org/10.1016/S1474-4422(06)70471-910.1016/S1474-4422(06)70471-916713924 [Google Scholar] [CrossRef] [PubMed]
[4]. Simon DK, Tanner CM, Brundin P, Parkinson disease epidemiology, pathology, genetics, and pathophysiologyClinics in Geriatric Medicine 2020 36(1):01-12.Available from: https://doi.org/10.1016/j.cger.2019.08.00210.1016/j.cger.2019.08.00231733690PMC6905381 [Google Scholar] [CrossRef] [PubMed]
[5]. Ellis TD, Colón-Semenza C, DeAngelis TR, Thomas CA, Hilaire MS, Earhart GM, Evidence for early and regular physical therapy and exercise in Parkinson’s diseaseSeminars in Neurology 2021 41(2):189-205.Available from: https://doi.org/10.1055/s-0041-172513310.1055/s-0041-172513333742432PMC8678920 [Google Scholar] [CrossRef] [PubMed]
[6]. Capato TT, Tornai J, Ávila P, Barbosa ER, Piemonte ME, Randomized controlled trial protocol: Balance training with rhythmical cues to improve and maintain balance control in Parkinson’s diseaseBMC Neurology 2015 15:162Available from: https://doi.org/10.1186/s12883-015-0418-x10.1186/s12883-015-0418-x26347052PMC4561447 [Google Scholar] [CrossRef] [PubMed]
[7]. Howe T, Lovgreen B, Cody F, Ashlon V, Oldham J, Auditory cues can modify the gait of persons with early- stage Parkinson’s disease: A method for enhancing Parkinsonian walking performanceClin Rehabil 2003 17:363-67.10.1191/0269215503cr621oa12785243 [Google Scholar] [CrossRef] [PubMed]
[8]. Lewis G, Byblow W, Walt S, Stride length regulation in Parkinson’s disease: The use of extrinsic, visual cuesBrain 2000 123(10):2077-90.10.1093/brain/123.10.207711004125 [Google Scholar] [CrossRef] [PubMed]
[9]. Morris ME, Iansek R, Matyas T, Summers JL, Stride length regulation in Parkinson’s disease. Normalization strategies and underlying mechanismsBrain 1996 119:551-68.10.1093/brain/119.2.5518800948 [Google Scholar] [CrossRef] [PubMed]
[10]. Rochester L, Hetherington V, Jones D, Nieuwboer A, Willems A, Kwakkel G, Attending to the task: Interference effects of functional tasks on walking in Parkinson’s disease and the roles of cognition, depression, fatigue and balanceArch Phys Med Rehabil 2004 85:1578-85.10.1016/j.apmr.2004.01.02515468014 [Google Scholar] [CrossRef] [PubMed]
[11]. Schenck CH, Boeve BF, Mahowald MW, Delayed emergence of a parkinsonian disorder or dementia in 81% of older men initially diagnosed with idiopathic rapid eye movement sleep behavior disorder: A 16-year update on a previously reported seriesSleep Med 2013 14:744-48.10.1016/j.sleep.2012.10.00923347909 [Google Scholar] [CrossRef] [PubMed]
[12]. Chang HY, Lee YY, Wu RM, Yang YR, Luh JJ, Effects of rhythmic auditory cueing on stepping in place in patients with Parkinson’s diseaseMedicine 2019 98(45):e17874Available from: https://doi.org/10.1097/MD.000000000001787410.1097/MD.000000000001787431702655PMC6855520 [Google Scholar] [CrossRef] [PubMed]
[13]. Gallagher R, Damodaran H, Werner WG, Powell W, Deutsch JE, Auditory and visual cueing modulate cycling speed of older adults and persons with Parkinson’s disease in a Virtual Cycling (V-Cycle) systemJournal of Neuroengineering and Rehabilitation 2016 13(1):77Available from: https://doi.org/10.1186/s12984-016-0184-z10.1186/s12984-016-0184-z27543195PMC4992269 [Google Scholar] [CrossRef] [PubMed]
[14]. Baker K, Rochester L, Nieuwboer A, The immediate effect of attentional, auditory, and a combined cue strategy on gait during single and dual tasks in Parkinson’s diseaseArch Phys Med Rehabil 2007 88(12):1593-600.Available from: https://doi.org/10.1016/j.apmr.2007.07.02610.1016/j.apmr.2007.07.02618047873 [Google Scholar] [CrossRef] [PubMed]
[15]. Bagley S, Kelly B, Tunnicliffe N, Turnbull GI, Walker JM, The effect of visual cues on the gait of independently mobile Parkinson’s disease patientsPhysiotherapy 1991 77:415-20.10.1016/S0031-9406(10)62035-410.1016/S0031-9406(10)62035-4 [Google Scholar] [CrossRef] [CrossRef]
[16]. Lu C, Amundsen Huffmaster SL, Tuite PJ, Vachon JM, MacKinnon CD, Effect of cue timing and modality on gait initiation in Parkinson disease with freezing of gaitArch Phys Med Rehabil 2017 98(7):1291-99.e1.Available from: https://doi.org/10.1016/j.apmr.2017.01.00910.1016/j.apmr.2017.01.00928167093PMC5483187 [Google Scholar] [CrossRef] [PubMed]
[17]. Miranda-Cantellops N, Tiu TK, Berg Balance TestingIn StatPearlsStatPearls Publishing [Google Scholar]
[18]. Movement disorder society task force on rating Scales for Parkinson’s DiseaseThe Unified Parkinson’s Disease Rating Scale (UPDRS): Status and recommendationsMovement disorders: Official Journal of the Movement Disorder Society 2003 18(7):738-50.Available from: https://doi.org/10.1002/mds.1047310.1002/mds.1047312815652 [Google Scholar] [CrossRef] [PubMed]
[19]. Calabrò RS, Naro A, Filoni S, Pullia M, Billeri L, Tomasello P, Walking to your right music: A randomized controlled trial on the novel use of treadmill plus music in Parkinson’s diseaseJ Neuroeng Rehabil 2019 16(1):68Available from: https://doi.org/10.1186/s12984-019-0533-910.1186/s12984-019-0533-931174570PMC6555981 [Google Scholar] [CrossRef] [PubMed]
[20]. Upadhyay S, Verma N, Effect of visual cue training on gait and walking velocity in chronic stroke patients-a quasi-experimental studyJ Clin Diagn Res 2022 16(4):YC06-YC10.10.7860/JCDR/2022/50349.16227 [Google Scholar] [CrossRef]