Introduction
Obesity, defined by an excess accumulation of body fat, has reached epidemic levels worldwide, posing significant public health challenges [1]. Traditionally viewed as a metabolic condition linked to lifestyle factors and genetic predispositions, obesity is now understood to have wide-ranging effects beyond physical health, impacting brain function and cognitive well-being. The prevalence of obesity has spurred extensive research into its impacts on the brain, as literature increasingly link excess weight to structural and functional changes in neural pathways [2]. In particular, obesity is associated with alterations in brain regions responsible for cognition, impulse control and emotional regulation, such as the Prefrontal Cortex (PFC) and hippocampus. Neuroinflammation- a chronic, low-grade inflammation triggered by adiposity- can alter neural connectivity, affecting cognitive processes and self-regulation [3].
Obesity
Obesity has emerged as a critical global health crisis, influenced by a myriad of complex factors, including biological, physiological, behavioural, social, economic and environmental dimensions [4]. By the end of the 20th century, obesity had reached epidemic proportions, becoming a major contributor to mortality and morbidity worldwide [5]. This increase has also imposed a significant economic burden on healthcare systems and society. In the 1970s, experts in the United Kingdom and the United States underscored the need to recognise obesity as a substantial public health issue primarily affecting adults, particularly women [6]. Despite this early recognition, effective treatment options for obesity have remained limited. Currently, overweight and obesity account for more deaths globally than underweight, with obesity prevalence exceeding 50% in several nations [7]. The Body Mass Index (BMI) is frequently used to assess excess weight in relation to height and age. The growing incidence of obesity is largely attributed to the availability of high-calorie foods combined with a lack of physical activity, presenting a substantial risk to public health due to its detrimental effects on multiple tissues and organs [8].
Obesity is strongly associated with diabetes, Metabolic Syndrome (MetS) and cardiovascular diseases. Recent studies have also underscored the link between obesity and cognitive impairments [9]. Neuroimaging research has demonstrated alterations in both brain structure and function associated with obesity and related diseases like MetS [10]. A meta-analysis of 16 studies involving over 25,000 participants found that low, overweight and obese BMI in midlife significantly increases the risk of developing Alzheimer’s disease and other types of dementia [11]. Moreover, obesity increasingly exhibits characteristics akin to addiction, influenced by neurobiological mechanisms similar to those seen in drug addiction. Cognitive impairments, especially in learning and memory, are frequently associated with obesity and comorbid conditions like hypertension and type 2 diabetes [12]. Addressing the multifaceted impacts of obesity, including its neurological implications, is crucial for developing effective prevention and treatment strategies.
Neuronal response in fasting: The Sympathetic Nervous System (SNS) plays a crucial role in regulating the body’s balance, including responses to stress and energy expenditure. Fasting reduces SNS activity, whereas eating- especially consuming high amounts of carbohydrates- tends to increase it, affecting how the body manages energy and stress. The SNS governs and impacts the breakdown of fats in adipose tissue [13]. The Parasympathetic Nervous System (PSNS) can contribute to obesity by directly impacting adipose tissue metabolism. Neuroimmune interactions involving the SNS and macrophages are essential for maintaining tissue balance, including in adipose tissue [14]. Reduced lipolysis in fat tissue due to SNS activity can lead to lipid accumulation and weight gain. The vagus nerve connects the brain and gastrointestinal tract [15]. Intra-abdominal fat cells stimulate the SNS, promoting glucose and fatty acid uptake through adrenergic receptors, leading to calorie storage and lipid gain. The cholinergic part of the vagus nerve regulates glucose and insulin, enhancing insulin secretion and lipid synthesis. Ghrelin, for instance, enhances food consumption by blocking vagal signals and releasing insulin [Table/Fig-1] [16].
Brain capillary responsiveness to obesity-associated chronic inflammation and insulin resistance [16].
(Figure credit: corresponding author (Dr. Puspalatha))
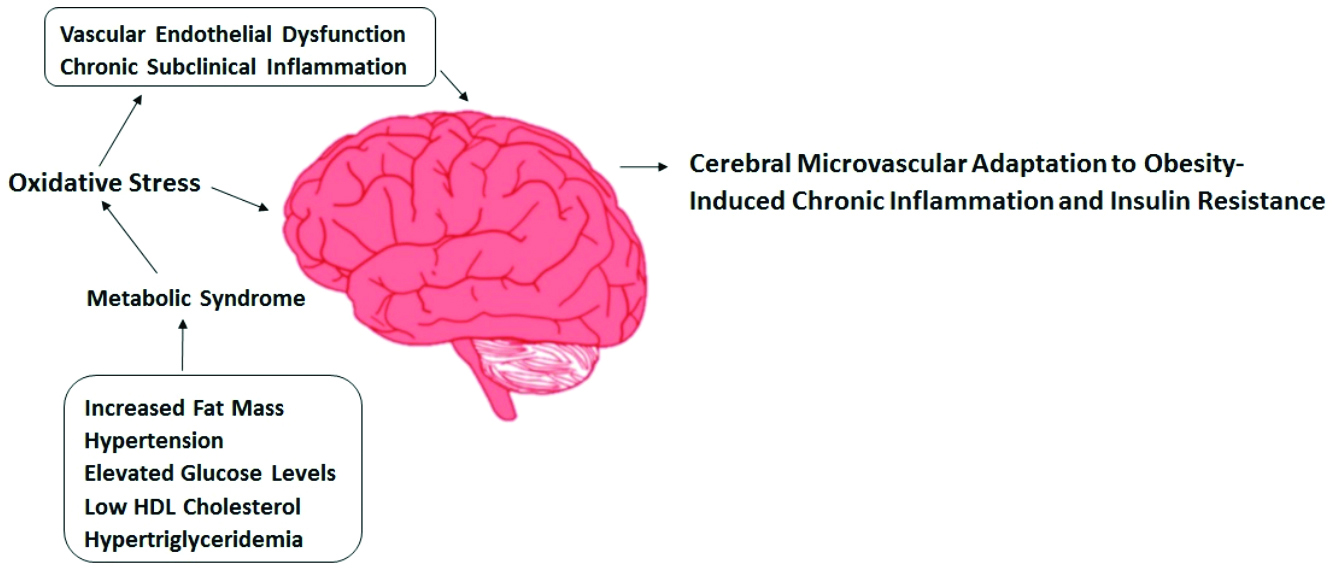
Neuroinflammation in obesity: Obesity-related inflammation may affect different brain areas, such as the hippocampus, cerebral cortex, brainstem and amygdala. Obesity’s low-grade inflammation may cause neuroinflammation by impacting the choroid plexus and disrupting the Blood-brain Barrier (BBB). Obesity-induced peripheral inflammation leads to the development of insulin resistance [17]. While the brain is protected from immune responses, changes in inflammation have been seen between the peripheral and central areas [18]. Adipokines, secreted by adipose tissue, are also present in the Central Nervous System (CNS), where they bind to certain receptors. Adipokines originating from the body’s outer regions can pass across the BBB or modify its function by affecting the cells responsible for the formation of BBB, hence controlling the CNS. Adipokines control neuroinflammation and oxidative damage, a crucial process in neurodegeneration and various chronic neurodegenerative diseases [19].
Age-related BBB damage can result in encephalitis, leading to brain inflammation. Neuroinflammation is likely a significant factor in cognitive impairment and a primary pathogenic process linked to the ageing process have shown regional cerebral atrophy in individuals with high BMI. However, the mechanisms behind these structural brain changes in obese individuals remain unclear. The amount of gray matter in the cerebral parts of the hippocampus, which plays a crucial role in learning and memory, may be affected. Individuals suffering from traumatic brain injury and neurodegenerative illnesses have been shown to have elevated levels of Neuron-specific Enolase (NSE) in their blood [20].
Structural Brain Changes in Obesity (Evidence from Neuroimaging Studies)
Gray matter: Early research consistently showed that patients with obesity had a reduction in gray matter volume compared to control groups. The decrease in gray matter was particularly pronounced in specific brain regions, including the inferior frontal gyri, right insula, precentral gyri, middle frontal gyrus, middle temporal gyrus, amygdala and left cerebellar hemisphere [21]. Azmi S et al., reported that obesity contributes to neurological damage, as reported by reduced corneal nerve fibre density (25.6 vs 32.0 n/mm2, p<0.0001) and length (17.9 vs 29.8 mm/mm2, p<0.0001) [22]. These changes align with reduced gray matter volume and neurological deficits impacting cognition and emotion regulation. Improvements after bariatric surgery, such as increased corneal nerve fibre density (28.2 vs 25.6 n/mm2, p=0.03) and length (20.4 vs 17.9 mm/mm2, p=0.02), implies the potential to mitigate these effects by addressing obesity. The correlation between reduced corneal nerve fibre density and length with decreased gray matter volume highlights the importance of maintaining a healthy weight for overall brain function. The positive changes observed after bariatric surgery offer hope for individuals struggling with obesity-related neurological damage, showing that intervention can lead to improvements in nerve fibre density and potentially cognitive function. These findings highlight obesity contribution to neurological damage, as reflected in reduced corneal nerve fibre density and length, which correspond to gray matter reduction and deficits in cognition and emotion regulation. Improvements after bariatric surgery suggest that addressing obesity can mitigate these effects, enhancing nerve fibre density and potentially restoring cognitive function. The findings regarding gray matter volume in patients with obesity have significant implications for both cognitive function and mental health. The reduction of gray matter in critical regions associated with decision-making, emotion regulation and sensory processing may contribute to challenges in impulse control, emotional well-being and overall cognitive performance in these individuals [23]. Conversely, the observed increases in gray matter in specific areas may indicate compensatory mechanisms or adaptations in response to obesity-related changes.
White matter: Compelling data suggests that persons with greater BMI have a worldwide decrease in white matter volume [24]. A comprehensive investigation of a cohort study has shown the association between high BMI and impaired white matter integrity. Elevated BMI is linked to reduced volume of white matter areas, consisting of the anterior and posterior thalamic radiations, inferior frontal-occipital fascicles, inferior and superior longitudinal fascicles and corpus callosum [25].
Subcortical areas: According to a study of Prunell-Castañé A et al., individuals classified as normal weight but metabolically obese exhibit reduced volumes in the bilateral caudate nucleus, accompanied by increased dimensions of the bilateral thalamus, putamen and globus pallidus. These neuroanatomical alterations may reflect underlying metabolic or structural adaptations associated with obesity-related pathophysiology, despite a normal body weight classification. These structural differences suggest alterations in brain regions involved in reward processing, motivation and cognitive control, which may contribute to the development and maintenance of obesity [26]. Understanding these neurobiological changes is crucial for developing effective interventions to address obesity and its associated health consequences.
Neuronal Changes in Children and Adolescents
Children with obesity often show reduced gray matter volume in critical brain areas, including the right medial temporal gyrus, thalamus, superior parietal gyrus, pre- and postcentral gyri and cerebellum. Each of these areas has distinct roles in cognitive processing, motor control and emotional regulation. The medial temporal gyrus and thalamus are linked with memory and emotional processing. Reduced gray matter here may impair memory formation and retrieval, contribute to difficulties in emotional regulation that are often observed in children with obesity. The parietal and central gyri are essential for spatial reasoning, attention and motor skills. These neural changes may contribute to cognitive and emotional challenges, emphasising the need for early interventions in childhood obesity to mitigate potential impacts on brain health and cognitive development [27]. Chronic inflammation associated with obesity in children and adolescents can harm critical brain regions, such as the hippocampus, which is essential for memory and learning development. During these formative years, obesity-related inflammation may impair cognitive functions and academic performance. Furthermore, obesity disrupts sleep patterns in youth, negatively impacting brain areas involved in mood regulation and emotional well-being while also increasing the risk of developing neuropsychiatric disorders. Long-term obesity during childhood and adolescence can alter neurotransmitter systems, particularly serotonin and dopamine, which are vital for mood stability, reward processing and social behaviour. It can also limit neuroplasticity, a key factor in the brain’s ability to learn and adapt during this critical developmental period and lead to a dysfunctional stress response, further hindering cognitive and emotional growth [28]. These neurological changes emphasise the importance of addressing weight management early in life to prevent potential long-term cognitive, emotional and developmental consequences.
Mechanism of Obesity Induced Neuronal Changes
Adipose tissue, particularly visceral fat, produces proinflammatory cytokines {like Tumour Necrosis Factor (TNF)-alpha and Interleukin (IL)-6} that can cross the Blood-brain Barrier (BBB) and promote neuroinflammation [29]. Microglial activation in response to these cytokines damages neuronal cells and contributes to structural brain changes. The brain depends on insulin for energy regulation and glucose metabolism. Obesity-induced insulin resistance in the brain impairs cognitive functions, reduces synaptic plasticity and is linked to neurodegenerative conditions. Leptin and ghrelin, hormones that regulate hunger and satiety, are often dysregulated in obesity [30]. Leptin resistance can disrupt signalling pathways in the hypothalamus and other brain regions, influencing food intake and energy expenditure. Higher levels of free fatty acids and glucose in obesity lead to oxidative stress, which damages neurons and exacerbates neuroinflammation. Obesity negatively affects neurogenesis (growth of new neurons), particularly in the hippocampus. This impairment is closely associated with memory deficits and reduced cognitive flexibility. Synaptic plasticity is also affected, reducing the brain’s ability to form new connections in response to learning and experience. This may be due to changes in neurotransmitter systems, inflammatory markers and insulin signalling pathways [31].
Metabolic Syndrome (MetS)
Older individuals with higher body fat levels, particularly around vital organs, often experience increased inflammation throughout their bodies. This condition is frequently associated with Metabolic Syndrome (MetS), a cluster of conditions- including high blood pressure, elevated blood sugar levels, excess body fat around the waist and abnormal cholesterol levels- that together increase the risk of heart disease, stroke and diabetes. The presence of MetS has been linked to a significant reduction in gray matter volume in the brain, which is critical for various cognitive functions [32]. As a result, these individuals frequently encounter difficulties when engaging in cognitive activities that require executive function. This decline in cognitive capabilities can manifest in several ways, leading to challenges in memory retention, decision-making, problem-solving and overall cognitive performance. The relationship between obesity, MetS and cognitive decline is multifaceted, involving not only the physical effects of excess body fat but also the complex interactions of inflammation, metabolic processes and brain health [33]. Understanding how obesity and MetS impact brain health is crucial for developing effective interventions aimed at mitigating cognitive decline in ageing populations [Table/Fig-2].
Structural brain changes.
(Figure credit: corresponding author (Dr. Puspalatha))
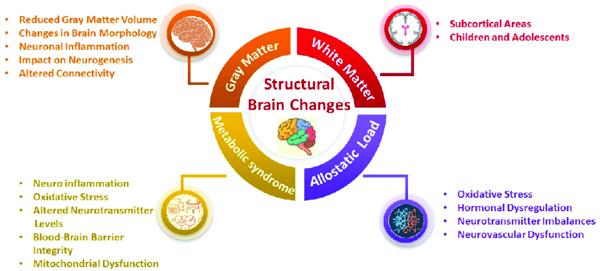
Other Anthropometric Variables
Apart from BMI, there is a relationship between the variables, including Waist-to-Hip Ratio (WHR), waist circumference and cortical thickness. There is a direct correlation between a greater WHR and bigger waist circumference and a decrease in the amount of gray matter in the brain [34]. Childhood obesity is linked to a significant decrease in gray matter, impacting critical cognitive areas of the brain such as the right medial temporal gyrus, right and left thalamus, left superior parietal gyrus, left pre- and postcentral gyri and left cerebellum. During maturity, there is a reduction in the thickness of the outer cortical layer of the brain in the superior regions. The frontal gyrus, seen on both the left and right-sides, is observed in those who are overweight, along with elevated allostatic load, as indicated by biomarkers that include blood pressure, glucose, cholesterol levels and inflammatory markers. Individuals diagnosed with Type 2 Diabetes Mellitus (T2DM) have reduced overall gray matter and hippocampus volume and an increased WHR and BMI [35]. These findings suggest that abdominal obesity may worsen brain shrinkage. Furthermore, WHR, waist circumference and cortical thinning are associated with a decrease in the amount of gray matter.
Obesity and Dyslipidemia
In obese individuals, dyslipidemia occurs due to increased free fatty acids from adipose tissue and Very Low-density Lipoprotein (VLDL) triglycerides from the liver and diet. Excess free fatty acids in the liver increase VLDL production, while a surplus of calories boosts VLDL from chylomicrons [36]. These triglycerides are broken down into lipoprotein lipases that hydrolyse long-chain fatty acids in the vascular endothelium, neurons and glia, burdening the Central Nervous System (CNS) and the Peripheral Nervous System (PNS) with additional free fatty acids. These lipids can build up in blood vessels, obstructing the flow of blood and the delivery of nutrients to tissues. Obesity, characterised by excess body fat, is a complex metabolic condition influenced by genetic, environmental and behavioural factors. Obesity induced excess adipose tissue initiates inflammatory processes and disrupts lipid metabolism, resulting in dyslipidemia. This condition is characterised by elevated levels of Low-density Lipoprotein (LDL) cholesterol and triglycerides, alongside decreased High-density Lipoprotein (HDL) cholesterol, which are frequently observed in individuals with obesity and contribute to heightened cardiovascular and metabolic health risks [37]. Medications like statins and fibrates may also be prescribed to lower lipid levels in obese individuals with dyslipidemia.
Obesity and Peripheral Nerve Damage
Polyneuropathy is a medical condition defined by the progressive loss of sensory experience, starting from the farthest parts of the body and moving towards the center, initially affecting the feet and eventually spreading to the hands. It is often described as a “stocking-glove” pattern of sensation loss, primarily involves severe situations or in the end stage. The sensory axons are impacted, while the motor axons remain unaffected. Prediabetes and type 2 diabetes are the primary reasons for polyneuropathy. However, literature has revealed that treatments aimed at reducing glucose levels had no effectiveness in preventing polyneuropathy in patients with type 2 diabetes [38]. The results indicate that other elements of MetS, which are seen in more than 75% of individuals with type 2 diabetes, could have a role in the progression of polyneuropathy. Obesity, prediabetes and dyslipidemia likely contribute to polyneuropathic pathophysiology through inflammatory processes, oxidative stress and microvascular dysfunction [39]. Altered adipokine secretion, particularly increased leptin and decreased adiponectin in obesity, may also play a role in the development of polyneuropathy. These adipokines can directly affect nerves and contribute to neuropathic pain. Furthermore, obesity-related insulin resistance and hyperinsulinemia may damage nerves through inflammatory pathways and nerve fibrosis [40]. This complex relationship involves metabolic, inflammatory and neuroendocrine pathways, requiring further research to identify potential therapeutic targets in metabolic disorder-associated polyneuropathy.
Neurological Benefits of Targeting Obesity
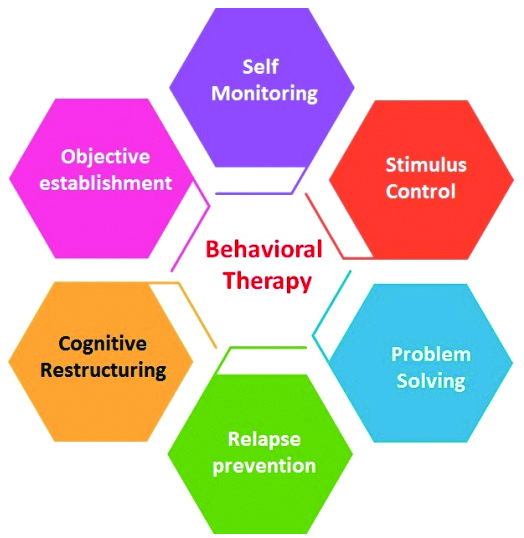
Due to the potential severe neurological effects of obesity and dyslipidemia, the best approach is to directly address obesity through lifestyle changes, involving dietary adjustments along with exercise. This offers a cost-effective and accessible strategy for combating obesity and enhancing neurological function. These interventions yield significant physiological benefits, such as improved metabolic profiles and enhanced cognitive and neural functioning, especially when combined. The modifications include better cellular metabolism, regulated glucose and insulin levels, decreased metabolic inflammation and increased lipid profiles. Although dietary and exercise therapies are the main methods for treating obesity, difficulties with adherence indicate that medication and bariatric surgery may have a role in controlling obesity-related neurological diseases. Research involving the observation of 342 Alzheimer’s disease patients found that lipid-lowering medications (statins or fibrates) were linked to slower cognitive decline [41]. The Fremantle Diabetes Study indicated that these medications might also protect against polyneuropathy in the field trial, demonstrated that fenofibrate effectively decreased non traumatic distal amputations in people with type 2 diabetes [42]. More conclusive clinical studies are necessary and at present, no professional recommendations advocate using statins or fibrates for illnesses affecting the Central Nervous System (CNS). Bariatric operations, such as Roux-en-Y gastric bypass, are very successful in treating individuals with morbid obesity, despite weight regain and frequent medical complications [43].
Weight Loss and Cognitive Benefits
Weight reduction has been shown to enhance mental abilities, particularly in individuals with obesity. People with a higher BMI showed gains in at least one cognitive function, while benefits were less evident in those only classified as overweight. Furthermore, recent research corroborates these findings, showing that weight reduction in obese or overweight adults, whether through diet or lifestyle changes, improves executive functions, cognition and language skills over time. These lifestyle changes improve cognitive function by reducing inflammation, enhancing insulin sensitivity and promoting neurogenesis [Table/Fig-3]. Regular exercise boosts Brain-derived Neurotrophic Factor (BDNF), supporting brain plasticity and cognitive health [44]. For individuals with severe obesity, bariatric surgery often leads to significant cognitive improvements, particularly in memory and executive function. Structural brain changes, such as increased gray and white matter, are observed post-surgery, indicating brain remodelling. Bariatric surgery has also been associated with cognitive gains, highlighting improvements in memory and executive functioning postsurgery. This may be due to structural brain changes, like increased gray and white matter, underscoring the brain’s ability to remodel after weight loss. Research suggests that midlife weight management could protect against future cognitive decline [45].
Therapeutic interventions.
(Figure credit: corresponding author (Dr. Puspalatha))
Dopamine Dysregulation in Obesity and Compulsive Eating
Obesity-related dopamine dysregulation plays a crucial role in compulsive eating behaviours, resembling neuroadaptations seen in addiction. This dysregulation includes reduced availability of dopamine D2 receptors in the striatum, which correlates with diminished activation in cortical regions vital for executive control, such as the Prefrontal Cortex (PFC) and Orbitofrontal Cortex (OFC) [46]. Impairment in these areas diminishes an individual’s ability to regulate eating behaviour, intensifying impulsivity and weakening response inhibition. Dopamine’s role in reward and motivation is also influenced by disrupted Dopamine Transporter (DAT) function, which affects dopamine reuptake and altered synaptic release in the mesolimbic pathway [47]. These changes can create an enhanced drive to seek high-calorie, palatable foods, particularly in environments where such cues are prevalent. This imbalance between reward-driven motivation and diminished control is a critical factor in the persistence of compulsive eating, emphasising the similarity between neurobiological pathways in obesity and substance use disorders.
Gender Differences in the Neurological Impacts of Obesity
The neurological impacts of obesity can differ between men and women due to various biological, hormonal and neuroanatomical differences. Research suggests that these gender differences may influence how obesity affects brain function and structure. Hormonal influences in women, specifically oestrogen and progesterone, have significant effects on brain regions such as the hypothalamus, which is involved in appetite regulation and energy balance [48]. Fluctuations in these hormones, especially during different life stages (e.g., pregnancy, menopause), may interact with obesity to impact emotional regulation, memory and reward processing. In men, testosterone influences brain areas like the Prefrontal Cortex (PFC) and hippocampus, which may lead to differences in how men and women respond to the cognitive and emotional effects of obesity. In case of white matter and gray matter differences, women with obesity often shows more pronounced reductions in gray matter volume, particularly in areas related to emotional regulation, such as the PFC and amygdala. In men, obesity-related changes may be more focused on white matter integrity, which could affect the efficiency of neural communication and cognitive processing. These sex-based differences could influence susceptibility to obesity-related cognitive decline or neuropsychiatric disorders. The brain’s reward system may be differently affected by obesity in men and women. Women are often more sensitive to food-related cues, which could lead to greater reward-seeking behaviour and compulsive eating, particularly when experiencing emotional distress. Men, on the other hand, may exhibit different patterns of reward processing, which could make them more vulnerable to binge eating or overeating in response to external food cues. The neuroinflammatory effects of visceral fat could differ in their impact on brain regions associated with memory, emotion and cognition. Women may experience greater neuroplasticity and resilience in response to obesity, especially in terms of memory and emotional regulation [49]. However, chronic obesity can still lead to cognitive decline in both sexes, with differences in the brain areas most affected. Women may show greater impairments in areas related to emotional processing, while men may be more prone to deficits in spatial cognition and motor control [50].
Conclusion(s)
As obesity rates continue to rise worldwide, the neurological impacts of excess weight have become a critical focus for future research and clinical strategies. Emerging evidence reveals that obesity is linked to notable changes in brain structure and function, especially within areas governing reward processing, cognitive control and memory. Future studies are expected to delve deeper into mechanisms such as dopamine signalling disruptions in the brain’s reward system, which may drive increased food intake and inflammation in the hypothalamus, impacting hunger and energy balance. There is a pressing need to integrate neurological health into obesity management, moving beyond traditional focuses on cardiovascular and metabolic issues. Upcoming research may shape multidimensional interventions that address both the physical and neurological aspects of obesity, fostering more holistic treatment approaches.
Authors’ contribution: SB and So B conceptualised the study and collected the data for the present review. MI contributed by conducting the literature search and critically reviewing it. TC was responsible for writing and designing the manuscript. PK compiled the article and played a key role in designing the review. All authors have reviewed and approved the final version of the manuscript.
[1]. Lin X, Li H, Obesity: Epidemiology, pathophysiology, and therapeuticsFront Endocrinol 2021 12:70697810.3389/fendo.2021.70697834552557PMC8450866 [Google Scholar] [CrossRef] [PubMed]
[2]. Hinney A, Körner A, Fischer-Posovszky P, The promise of new anti-obesity therapies arising from knowledge of genetic obesity traitsNat Rev Endocrinol 2022 18(10):623-37.10.1038/s41574-022-00716-035902734PMC9330928 [Google Scholar] [CrossRef] [PubMed]
[3]. Iceta S, Dadar M, Daoust J, Scovronec A, Leblanc V, Pelletier M, Association between visceral adiposity index, binge eating behaviour, and grey matter density in caudal anterior cingulate cortex in severe obesityBrain Sci 2021 11(9):115810.3390/brainsci1109115834573180PMC8468041 [Google Scholar] [CrossRef] [PubMed]
[4]. Kunyahamu MS, Daud A, Jusoh N, Obesity among health-care workers: Which occupations are at higher risk of being obese?Int J Environ Res Public Health 2021 18(8):438110.3390/ijerph1808438133924182PMC8074354 [Google Scholar] [CrossRef] [PubMed]
[5]. Muscogiuri G, Barrea L, Bettini S, El Ghoch M, Katsiki N, Tolvanen L, European Association for the Study of Obesity (EASO) Position Statement on medical nutrition therapy for the management of individuals with overweight or obesity and cancerObes Facts 2024 :01-20.10.1159/00054215539433024 [Google Scholar] [CrossRef] [PubMed]
[6]. Segarra M, Aburto MR, Acker-Palmer A, Blood–brain barrier dynamics to maintain brain homeostasisTrends Neurosci 2021 44(5):393-405.10.1016/j.tins.2020.12.00233423792 [Google Scholar] [CrossRef] [PubMed]
[7]. Shekar M, Popkin B, Obesity: Health and economic consequences of an impending global challenge 2020 Feb 13 World Bank Publications10.1596/978-1-4648-1491-4 [Google Scholar] [CrossRef]
[8]. Safiri S, Kolahi AA, Smith E, Hill C, Bettampadi D, Mansournia MA, Global, regional and national burden of osteoarthritis 1990-2017: A systematic analysis of the Global Burden of Disease Study 2017Ann Rheum Dis 2020 79(6):819-28.10.1136/annrheumdis-2019-21651532398285 [Google Scholar] [CrossRef] [PubMed]
[9]. Montégut L, Lopez-Otin C, Magnan C, Kroemer G, Old paradoxes and new opportunities for appetite control in obesityTrends Endocrinol Metab 2021 32(5):264-94.10.1016/j.tem.2021.02.00533707095 [Google Scholar] [CrossRef] [PubMed]
[10]. Le Thuc O, García-Cáceres C, Obesity-induced inflammation: Connecting the periphery to the brainNat Metab 2024 6(7):1237-52.10.1038/s42255-024-01079-838997442 [Google Scholar] [CrossRef] [PubMed]
[11]. Colizzi C, The protective effects of polyphenols on Alzheimer’s disease: A systematic reviewAlzheimer’s & Dementia: Transl Res Clin Interv 2019 5:184-96.10.1016/j.trci.2018.09.00231194101 [Google Scholar] [CrossRef] [PubMed]
[12]. Ren J, Wu NN, Wang S, Sowers JR, Zhang Y, Obesity cardiomyopathy: Evidence, mechanisms, and therapeutic implicationsPhysiol Rev 2021 101(4):1745-807.10.1152/physrev.00030.202033949876 [Google Scholar] [CrossRef] [PubMed]
[13]. Zhuang QS, Meng L, Wang Z, Shen L, Ji HF, Associations between obesity and Alzheimer’s disease: Multiple bioinformatic analysesJ Alzheimers Dis 2021 80(1):271-81.10.3233/JAD-20123533523009 [Google Scholar] [CrossRef] [PubMed]
[14]. Daoust J, Schaffer J, Zeighami Y, Dagher A, García-García I, Michaud A, White matter integrity differences in obesity: A meta-analysis of diffusion tensor imaging studiesNeurosci Biobehav Rev 2021 129:133-41.10.1016/j.neubiorev.2021.07.02034284063 [Google Scholar] [CrossRef] [PubMed]
[15]. Fernández-Andújar M, Morales-García E, García-Casares N, Obesity and gray matter volume assessed by neuroimaging: A systematic reviewBrain Sci 2021 11(8):99910.3390/brainsci1108099934439618PMC8391982 [Google Scholar] [CrossRef] [PubMed]
[16]. Huang KP, Goodson ML, Vang W, Li H, Page AJ, Raybould HE, Leptin signaling in vagal afferent neurons supports the absorption and storage of nutrients from high-fat dietInt J Obes 2021 45(2):348-57.10.1038/s41366-020-00678-132917985PMC7854885 [Google Scholar] [CrossRef] [PubMed]
[17]. Gómez-Apo E, Mondragón-Maya A, Ferrari-Díaz M, Silva-Pereyra J, Structural brain changes associated with overweight and obesityJ Obes 2021 2021(1):661338510.1155/2021/661338534327017PMC8302366 [Google Scholar] [CrossRef] [PubMed]
[18]. Stranahan AM, Visceral adiposity, inflammation, and hippocampal function in obesityNeuropharmacology 2022 205:10892010.1016/j.neuropharm.2021.10892034902347PMC8958382 [Google Scholar] [CrossRef] [PubMed]
[19]. Salsinha AS, Rodríguez-Alcalá LM, Relvas JB, Pintado ME, Fatty acids role on obesity-induced hypothalamus inflammation: From problem to solution–A reviewTrends Food Sci Technol 2021 112:592-607.10.1016/j.tifs.2021.03.042 [Google Scholar] [CrossRef]
[20]. Karczewski J, Zielińska A, Staszewski R, Eder P, Dobrowolska A, Souto EB, Obesity and the brainInt J Mol Sci 2022 23(11):614510.3390/ijms2311614535682824PMC9181303 [Google Scholar] [CrossRef] [PubMed]
[21]. Jiang F, Li G, Ji W, Zhang Y, Wu F, Hu Y, Obesity is associated with decreased gray matter volume in children: A longitudinal studyCereb Cortex 2023 33(7):3674-82.10.1093/cercor/bhac30035989308PMC10068275 [Google Scholar] [CrossRef] [PubMed]
[22]. Azmi S, Ferdousi M, Liu Y, Adam S, Iqbal Z, Dhage S, Bariatric surgery leads to an improvement in small nerve fibre damage in subjects with obesityInt J Obes 2021 45(3):631-38.10.1038/s41366-020-00727-933504933 [Google Scholar] [CrossRef] [PubMed]
[23]. Park HR, Quidé Y, Schofield PR, Williams LM, Gatt JM, Grey matter covariation and the role of emotion reappraisal in mental wellbeing and resilience after early life stress exposureTransl Psychiatry 2022 12(1):8510.1038/s41398-022-01849-635220403PMC8882193 [Google Scholar] [CrossRef] [PubMed]
[24]. Chiu S, Krauss RM, Obesity and lipoprotein metabolismIn Handbook of Obesity, Two-Volume Set 2024 Jan 9 (pp. Vol1-481)CRC Press10.1201/9781003437734-55 [Google Scholar] [CrossRef]
[25]. Nota MH, Vreeken D, Wiesmann M, Aarts EO, Hazebroek EJ, Kiliaan AJ, Obesity affects brain structure and function-rescue by bariatric surgery?Neuroscience & Biobehavioral Reviews 2020 108:646-57.10.1016/j.neubiorev.2019.11.02531794778 [Google Scholar] [CrossRef] [PubMed]
[26]. Prunell-Castañé A, Garolera M, Ottino-González J, Jurado MÁ, Allostatic load, adverse childhood experiences, executive functions, and BMI status in adolescents and young adultsAm J Hum Biol 2024 36(9):e2408910.1002/ajhb.2408938665069 [Google Scholar] [CrossRef] [PubMed]
[27]. Sadler JR, Thapaliya G, Ranganath K, Gabay A, Chen L, Smith KR, Paediatric obesity and metabolic syndrome associations with cognition and the brain in youth: Current evidence and future directionsPediatr Obes 2023 18(8):e1304210.1111/ijpo.1304237202148PMC10826337 [Google Scholar] [CrossRef] [PubMed]
[28]. Lenart-Bugla M, Szcześniak D, Bugla B, Kowalski K, Niwa S, Rymaszewska J, The association between allostatic load and brain: A systematic reviewPsychoneuroendocrinology 2022 145:10591710.1016/j.psyneuen.2022.10591736113380 [Google Scholar] [CrossRef] [PubMed]
[29]. Ashford JW, Neuroplasticity: The critical issue for Alzheimer’s disease and links to obesity and depressionAm J Geriatr Psychiatry 2023 31(10):867-75.10.1016/j.jagp.2023.07.00737481402 [Google Scholar] [CrossRef] [PubMed]
[30]. Guo DH, Yamamoto M, Hernandez CM, Khodadadi H, Baban B, Stranahan AM, Visceral adipose NLRP3 impairs cognition in obesity via IL-1R1 on CX3CR1+ cellsThe Journal of Clinical Investigation 2020 130(4):1961-76.10.1172/JCI12607831935195PMC7108893 [Google Scholar] [CrossRef] [PubMed]
[31]. Yeung AY, Tadi P, Physiology, Obesity Neurohormonal Appetite and Satiety Control. 2023 Jan 3In: StatPearls [Internet] 2025 Jan Treasure Island (FL)StatPearls Publishing32310366 [Google Scholar] [PubMed]
[32]. Prehn K, Profitlich T, Rangus I, Heßler S, Witte AV, Grittner U, Bariatric surgery and brain health—a longitudinal observational study investigating the effect of surgery on cognitive function and gray matter volumeNutrients 2020 12(1):12710.3390/nu1201012731906475PMC7019777 [Google Scholar] [CrossRef] [PubMed]
[33]. Morys F, Dadar M, Dagher A, Association between midlife obesity and its metabolic consequences, cerebrovascular disease, and cognitive declineJ Clin Endocrinol Metab 2021 106(10):e4260-e4274.10.1210/clinem/dgab13533677592PMC8475210 [Google Scholar] [CrossRef] [PubMed]
[34]. Forte N, Boccella S, Tunisi L, Fernández-Rilo AC, Imperatore R, Iannotti FA, Orexin-A and endocannabinoids are involved in obesity-associated alteration of hippocampal neurogenesis, plasticity, and episodic memory in miceNat Commun 2021 12(1):613710.1038/s41467-021-26388-434675233PMC8531398 [Google Scholar] [CrossRef] [PubMed]
[35]. Carbine KA, Duraccio KM, Hedges-Muncy A, Barnett KA, Kirwan CB, Jensen CD, White matter integrity disparities between normal-weight and overweight/obese adolescents: An automated fiber quantification tractography studyBrain Imaging Behav 2020 14:308-19.10.1007/s11682-019-00036-430719618 [Google Scholar] [CrossRef] [PubMed]
[36]. Cao C, Koh HC, Van Vliet S, Patterson BW, Reeds DN, Laforest R, Increased plasma fatty acid clearance, not fatty acid concentration, is associated with muscle insulin resistance in people with obesityMetabolism 2022 132:15521610.1016/j.metabol.2022.15521635577100PMC10424797 [Google Scholar] [CrossRef] [PubMed]
[37]. Chang KC, Pai YW, Lin CH, Lee IT, Chang MH, The association between hyperlipidemia, lipid-lowering drugs and diabetic peripheral neuropathy in patients with type 2 diabetes mellitusPlos one 2023 18(6):e028737310.1371/journal.pone.028737337319238PMC10270586 [Google Scholar] [CrossRef] [PubMed]
[38]. Martin M, Condori AI, Davico B, Rosso LG, Gaete L, Tetzlaff W, Impaired reverse cholesterol transport is associated with changes in fatty acid profile in children and adolescents with abdominal obesityJ Nutr 2024 154(1):12-25.10.1016/j.tjnut.2023.08.03737716606 [Google Scholar] [CrossRef] [PubMed]
[39]. Bonomo R, Kramer S, Aubert VM, Obesity-associated neuropathy: Recent preclinical studies and proposed mechanismsAntioxid Redox Signal 2022 37(7-9):597-612.10.1089/ars.2021.027835152780PMC9527047 [Google Scholar] [CrossRef] [PubMed]
[40]. Huber K, Szerenos E, Lewandowski D, Toczylowski K, Sulik A, The role of adipokines in the pathologies of the central nervous systemInt J Mol Sci 2023 24(19):1468410.3390/ijms24191468437834128PMC10572192 [Google Scholar] [CrossRef] [PubMed]
[41]. Mahajan R, Stokes M, Elliott A, Munawar DA, Khokhar KB, Thiyagarajah A, Complex interaction of obesity, intentional weight loss and heart failure: A systematic review and meta-analysisHeart 2020 106(1):58-68.10.1136/heartjnl-2019-31477031530572 [Google Scholar] [CrossRef] [PubMed]
[42]. Li CM, Song JR, Zhao J, Wang CF, Zhang CS, Wang HD, The effects of bariatric surgery on cognition in patients with obesity: A systematic review and meta-analysisSurg Obes Relat Dis 2022 18(11):1323-38.10.1016/j.soard.2022.07.00736058832 [Google Scholar] [CrossRef] [PubMed]
[43]. Gardner CD, Vadiveloo MK, Petersen KS, Anderson CA, Springfield S, Van Horn L, American Heart Association Council on Lifestyle and Cardiometabolic HealthPopular dietary patterns: Alignment with American Heart Association 2021 dietary guidance: A scientific statement from the American Heart AssociationCirculation 2023 147(22):1715-30.10.1161/CIR.000000000000114637128940 [Google Scholar] [CrossRef] [PubMed]
[44]. Wadden TA, Tronieri JS, Butryn ML, Lifestyle modification approaches for the treatment of obesity in adultsAm Psychol 2020 75(2):23510.1037/amp000051732052997PMC7027681 [Google Scholar] [CrossRef] [PubMed]
[45]. Liang J, Wang H, Zeng Y, Qu Y, Liu Q, Zhao F, Physical exercise promotes brain remodeling by regulating epigenetics, neuroplasticity and neurotrophinsRev Neurosci 2021 32(6):615-29.10.1515/revneuro-2020-009933583156 [Google Scholar] [CrossRef] [PubMed]
[46]. Alhurani RE, Vassilaki M, Aakre JA, Mielke MM, Kremers WK, Machulda MM, Decline in weight and incident mild cognitive impairment: Mayo clinic study of agingJAMA Neurol 2016 73(4):439-46.10.1001/jamaneurol.2015.475626831542PMC4828256 [Google Scholar] [CrossRef] [PubMed]
[47]. Tavares G, Marques D, Barra C, Rosendo-Silva D, Costa A, Rodrigues T, Dopamine D2 receptor agonist, bromocriptine, remodels adipose tissue dopaminergic signalling and upregulates catabolic pathways, improving metabolic profile in type 2 diabetesMol Metab 2021 51:10124110.1016/j.molmet.2021.10124133933677PMC8164040 [Google Scholar] [CrossRef] [PubMed]
[48]. Ebrahimi MN, Banazadeh M, Alitaneh Z, Suha AJ, Esmaeili A, Hasannejad-Asl B, The distribution of neurotransmitters in the brain circuitry: Mesolimbic pathway and addictionPhysiol Behav 2024 284:11463910.1016/j.physbeh.2024.11463939004195 [Google Scholar] [CrossRef] [PubMed]
[49]. DeBenedictis JN, Nymo S, Ollestad KH, Boyesen GA, Rehfeld JF, Holst JJ, Changes in the homeostatic appetite system after weight loss reflect a normalization toward a lower body weightJ Clin Endocrinol Metab 2020 105(7):e2538-e2546.10.1210/clinem/dgaa20232301981PMC7250208 [Google Scholar] [CrossRef] [PubMed]
[50]. Ganguli M, Beer JC, Zmuda JM, Ryan CM, Sullivan KJ, Chang CC, Aging, diabetes, obesity, and cognitive decline: A population-based studyJ Am Geriatr Soc 2020 68(5):991-98.10.1111/jgs.1632132020605PMC8597580 [Google Scholar] [CrossRef] [PubMed]