The reconstruction of large oncologic defects, especially in the groin region, poses significant surgical challenges due to the exposure of vital structures such as the femoral vessels. A 68-year-old male, previously treated for melanoma of the left foot, presented with a recurrent swelling in the left inguinal region. Fine Needle Aspiration Cytology (FNAC) confirmed malignant melanoma. Imaging studies revealed a lobulated, infiltrative lesion in the left proximal thigh, abutting the femoral triangle but without muscle invasion. The patient underwent radical excision of the lesion along with inguinal lymph node dissection, resulting in a 20×10 cm defect with exposed femoral vessels that required complex reconstruction. A pedicled chimeric left Tensor Fascia Lata (TFL) perforator flap and a vastus lateralis muscle flap were harvested. The TFL flap was raised based on a single dominant perforator, while the vastus lateralis muscle flap was based on the descending branch of the Lateral Circumflex Femoral Artery (LCFA). Both flaps were successfully tunneled under the rectus femoris muscle and inset into the defect. The donor site was partially closed, and the remaining area was planned for delayed secondary skin grafting. The pedicled chimeric TFL and vastus lateralis flaps effectively covered an extensive groin defect with exposed femoral vessels. This approach offers a robust reconstructive option for complex oncologic defects, ensuring functional and aesthetic outcomes. Further studies and case series are warranted to explore the versatility and limitations of this technique.
Case Report
A 68-year-old male patient presented to the facility with complaints of swelling in the upper aspect of the left groin region for the past three years. The patient was apparently well three years ago when he first noticed a swelling in the left inguinal region. The swelling was insidious in onset, and the patient reported a gradual increase in its size over the past few months; it initially measured 2×2 cm and has progressed to its current size of 10×8 cm, accompanied by a sensation of heaviness and discomfort in the affected area.
The swelling was not associated with pain, and there were no aggravating or relieving factors. The patient reported no history of trauma, fever, weight loss, anorexia, or discharge from the swelling, and there were no bowel or bladder complaints. There was also no history of restriction of movement.
His medical history revealed that he had undergone excision of a melanoma mass from the plantar region of his left foot, following which the patient remained asymptomatic for about six months.
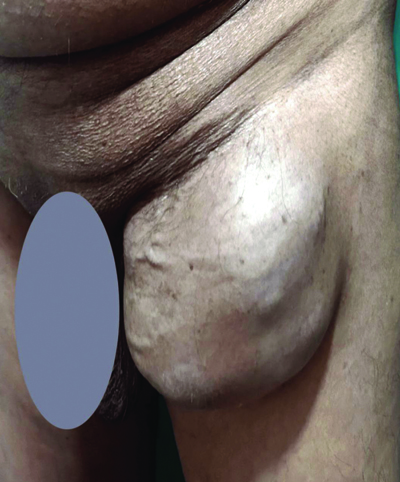
On physical examination, a firm, non tender mass was palpated in the left inguinal region [Table/Fig-1]. Further evaluation with FNAC confirmed the diagnosis of malignant melanoma.
Firm, non tender mass in the left inguinal region.
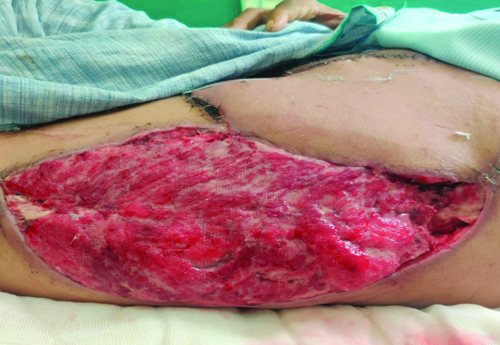
A Contrast-enhanced Computed Tomography (CECT) scan of the abdomen was performed to assess the extent of the disease. The imaging revealed a lobulated, enhancing infiltrating lesion involving the subcutaneous fat of the left proximal thigh. The lesion extended near the femoral triangle, abutting the sartorius and rectus femoris muscles, but without infiltrating these muscles or extending into the intramuscular compartment. The craniocaudal extent of the mass measured approximately 9.7×6.7×12.1 cm, with a satellite nodule adjacent to the adductor magnus muscle measuring 17×15 mm. Multiple enlarged lymph nodes were noted in the left external iliac, common iliac and para-aortic regions. A contrast-enhanced chest CT scan showed no suspicious lung parenchymal infiltrates, indicating no distant metastasis at the time of assessment. Given the size and location of the mass, along with the associated lymphadenopathy, surgical intervention was planned. The patient underwent excision of the left groin swelling along with an inguinal lymph node dissection. Postexcision, a significant defect measuring 20×10 cm was created in the left groin region, with exposed femoral vessels posing a considerable challenge for reconstruction [Table/Fig-2a-c].
a-c) Excision of the left groin swelling along with an inguinal lymph node dissection. Postexcision, a significant defect measuring 20×10 cm was created in the left groin region, with exposed femoral vessels.

A pedicled chimeric left TFL perforator flap was harvested to address the large defect and protect the vital structures. The surgical approach involved making an incision 2 cm medial to a line drawn from the Anterior Superior Iliac Spine (ASIS) to the lateral border of the patella. The incision was deepened through the subcutaneous tissue and the deep fascia was incised to identify the rectus femoris muscle. No significant perforators were found between the septum’s rectus femoris and vastus lateralis muscles. A single dominant TFL perforator was visualised approximately 10 cm distal to the ASIS after extending the incision proximally. The entry of the perforator into the skin was confirmed, and the TFL muscle was elevated off the perforator. A skin paddle measuring 20×10 cm was marked around the perforator, and the flap was islanded. The bleeding was assessed and found to be satisfactory, allowing the flap to be tunneled below the rectus femoris muscle into the defect.
In addition to the TFL flap, a 10×7 cm segment of the vastus lateralis muscle was islanded, based on the LCFA’s descending branch, to further cover the exposed femoral vessels. The muscle was tunneled below the rectus femoris into the defect and secured over the femoral vessels using Vicryl sutures. The TFL perforator flap was inset into the defect, providing adequate coverage and protection to the underlying structures. The donor site defect was partially closed, and the remaining area was left open for delayed secondary skin grafting.
The patient’s postoperative course was uneventful. Daily dressing of the donor site was performed, with the flap showing good viability and no signs of infection or dehiscence [Table/Fig-3]. The patient was scheduled for subsequent follow-up and adjuvant therapy as part of the comprehensive management plan for melanoma.
Follow-up image showing donor site with good granulation and healthy flap.
Discussion
Malignant melanoma is one of the most aggressive forms of skin cancer, characterised by its rapid potential for metastasis and poor prognosis in advanced stages [1]. Its incidence has steadily increased worldwide, with significant implications for public health. According to recent global cancer statistics, melanoma accounts for a substantial burden of skin cancer cases, particularly in populations with high levels of ultraviolet radiation exposure and lighter skin types [2]. Despite advances in early detection and surgical management, the survival rate for patients with regional or distant metastases remains low, highlighting the need for comprehensive management strategies that address oncologic and reconstructive challenges [3].
The primary treatment for localised malignant melanoma involves wide local excision of the primary lesion, often coupled with sentinel lymph node biopsy or regional lymphadenectomy in cases of nodal involvement [3]. However, these procedures can result in extensive soft-tissue defects, particularly in the anatomically complex groin region [4]. The groin area presents unique reconstructive challenges due to its proximity to critical vascular structures such as the femoral artery and vein, its high mobility, and exposure to contamination [5]. Inadequate coverage of these defects can lead to complications such as wound dehiscence, infection and lymphoedema, significantly impacting the patient’s quality of life and prognosis [6,7].
The reconstruction of extensive groin defects, especially following oncologic resections, remains a formidable challenge due to the anatomical complexity and the need for durable soft tissue coverage. Defects in this region often involve exposed critical structures, such as the femoral vessels, requiring a reconstructive approach that not only provides bulk and coverage, but also ensures reliable vascularity to support healing and resist infection [8]. Pedicled chimeric flaps, incorporating both the TFL and vastus lateralis muscles, offer a unique and effective solution for addressing these challenges. The TFL flap is a well-established option for lower extremity reconstruction. It is nourished primarily by the LCFA, making it a dependable flap for coverage of large defects due to its robust blood supply and versatile tissue composition [9]. Studies have shown that the TFL flap, when elevated on its perforators, can be used as a pedicled or free flap, providing significant flexibility in reconstructive scenarios [10]. Its application has been particularly beneficial in covering defects following trauma, oncologic resections and chronic infections [11]. In the present case, the TFL flap was effectively utilised to cover a large groin defect with exposed femoral vessels, demonstrating its utility in managing complex defects in this anatomically challenging region.
Integrating the vastus lateralis muscle flap further enhanced the reconstructive outcome by providing additional bulk and a secondary vascularised tissue layer. The vastus lateralis muscle, also vascularised by the descending branch of the LCFA, is a reliable option for muscular coverage, particularly in areas where robust protection of underlying structures is required [12]. Muscle flaps are advantageous in such scenarios because they can fill dead spaces, reduce the risk of haematoma and seroma formation, and decrease the likelihood of wound complications [13]. The use of muscle flaps in reconstructing groin defects has been well-documented, particularly in cases where vascular structures are at risk [7].
Chimeric flaps combine multiple tissue components from the same vascular source and provide an advanced option for complex reconstructions. They offer the benefits of different tissue types such as skin, muscle and fascia, while maintaining a single vascular pedicle, thereby reducing the complexity of the reconstruction [14]. This technique allows for tailored reconstruction, addressing the specific needs of each component of the defect [15]. In the present case, the chimeric TFL and vastus lateralis muscle flaps provided a comprehensive solution by simultaneously addressing skin coverage and muscular support requirements, thus optimising the functional and aesthetic outcomes.
According to the study conducted by Rifaat MA and Abdel Gawad WS, TFL flaps used for covering abdominal wall and groin defects had a good success rate with relatively few complications. Two out of six cases showed wound dehiscence, and one had distal tip necrosis, all of which were treated conservatively and healed spontaneously [16].
Hallock GG highlighted the versatility of chimeric flaps in managing complex defects, noting their ability to provide both functional and aesthetic results in a single-stage procedure [17]. Similarly, the advantages of using perforator-based chimeric flaps for oncologic reconstructions include reduced donor site morbidity and improved flap survival as significant benefits [18]. As demonstrated in the current case, the use of such flaps in the groin region shows promise in minimising complications associated with extensive defect coverage, such as flap necrosis, infection and dehiscence.
Despite the promising outcomes associated with the use of chimeric flaps, it is crucial to consider the potential complications, such as partial flap loss, donor site morbidity, and the technical expertise required for flap harvest and inset [19]. Meticulous surgical planning and intraoperative technique are paramount to ensure flap viability and successful integration into the defect site. Long-term follow-up and additional studies are needed to further evaluate the outcomes and refine the indications for chimeric flap use in complex oncologic reconstructions.
Conclusion(s)
Reconstruction of extensive groin defects following oncologic excision poses significant challenges due to the complex anatomy and the need for durable coverage of vital structures such as the femoral vessels. A pedicled chimeric TFL perforator flap combined with a vastus lateralis muscle flap, offers a reliable and effective solution for such complex reconstructions. This technique provides robust coverage and protection of exposed structures and minimises donor site morbidity by utilising a single vascular pedicle to support multiple tissue components. Given the favourable results and minimal complications, this approach should be considered a viable option for reconstructive surgeons managing similar oncologic cases. Further studies and long-term follow-ups are needed to establish standardised protocols and to evaluate the broader applicability of this innovative reconstructive strategy in diverse clinical settings.
[1]. Heistein JB, Acharya U, Mukkamalla SKR, Malignant melanoma 2024 Treasure Island (FL)StatPearls Publishing [Google Scholar]
[2]. Arnold M, Singh D, Laversanne M, Global burden of cutaneous melanoma in 2020 and projections to 2040JAMA Dermatol 2022 158:495-503.10.1001/jamadermatol.2022.016035353115PMC8968696 [Google Scholar] [CrossRef] [PubMed]
[3]. Pisani P, Airoldi M, Allais A, Metastatic disease in head & neck oncologyActa Otorhinolaryngol Ital 2020 40:01-86.10.14639/0392-100X-suppl.1-40-202032469009PMC7263073 [Google Scholar] [CrossRef] [PubMed]
[4]. Joyce KM, Surgical management of melanoma. Ward WH, Farma JM (ed)Brisbane (AU)Codon Publications [Google Scholar]
[5]. Sörelius K, Schiraldi L, Giordano S, Oranges CM, Raffoul W, Di Summa PG, Reconstructive surgery of inguinal defects: A systematic literature review of surgical etiology and reconstructive techniqueIn Vivo 2019 33:01-10.10.21873/invivo.1143130587595PMC6364081 [Google Scholar] [CrossRef] [PubMed]
[6]. Rosen RD, Manna B, Wound dehiscence 2024 Treasure IslandStatPearls Publishing [Google Scholar]
[7]. Price A, Contractor U, White R, Williams I, The use of vascularised muscle flaps for treatment or prevention of wound complications following arterial surgery in the groinInt Wound J 2020 17:1669-77.10.1111/iwj.1344932744430PMC7949366 [Google Scholar] [CrossRef] [PubMed]
[8]. Miyamoto S, Fujiki M, Nakatani F, Kobayashi E, Sakisaka M, Sakuraba M, Reconstruction of complex groin defects after sarcoma resectionAnn Plast Surg 2017 78:443-47.10.1097/SAP.000000000000090227740954 [Google Scholar] [CrossRef] [PubMed]
[9]. Wang L, Ma C, Shen Y, Haugen TW, He Y, Sun J, Application of tensor fascia lata perforator flap in head and neck reconstructionInt J Oral Maxillofac Surg 2024 53:551-57.10.1016/j.ijom.2024.01.00438228464 [Google Scholar] [CrossRef] [PubMed]
[10]. Kwon H, Lee S, Kim S, Reconstruction of complex knee wounds with a distally based gracilis flap and gastrocnemius myocutaneous flap: A case reportFront Surg 2023 10:110993610.3389/fsurg.2023.110993636843998PMC9952235 [Google Scholar] [CrossRef] [PubMed]
[11]. Wei FC, Celik N, Perforator flap entityClin Plast Surg 2003 30:325-29.10.1016/s0094-1298(03)00033-612916589 [Google Scholar] [CrossRef] [PubMed]
[12]. Alkon JD, Smith A, Losee JE, Illig KA, Green RM, Serletti JM, Management of complex groin wounds: Preferred use of the rectus femoris muscle flapPlast Reconstr Surg 2005 115:776-83.10.1097/01.prs.0000152436.50604.0415731678 [Google Scholar] [CrossRef] [PubMed]
[13]. Deramo P, Rose J, Flaps: Muscle and Musculocutaneous 2024 Treasure Island (FL)StatPearls Publishing [Google Scholar]
[14]. Hallock GG, The chimeric propeller flapSemin Plast Surg 2020 34:207-09.10.1055/s-0040-171429033041692PMC7542215 [Google Scholar] [CrossRef] [PubMed]
[15]. Kim JT, Kim SW, Perforator flap versus conventional flapJ Korean Med Sci 2015 30:51410.3346/jkms.2015.30.5.51425931780PMC4414633 [Google Scholar] [CrossRef] [PubMed]
[16]. Rifaat MA, Abdel Gawad WS, The use of tensor fascia lata pedicled flap in reconstructing full thickness abdominal wall defects and groin defects following tumor ablationJ Egypt Natl Canc Inst 2005 17(3):139-48. [Google Scholar]
[17]. Hallock GG, Metachronous flaps from the same subscapular axisPlast Reconstr Surg 2002 109:2424-30.10.1097/00006534-200206000-0003712045570 [Google Scholar] [CrossRef] [PubMed]
[18]. Low OW, Sebastin SJ, Cheah AEJ, A review of pedicled perforator flaps for reconstruction of the soft tissue defects of the leg and footIndian J Plast Surg 2019 52(1):26-36.10.1055/s-0039-168810331456610PMC6664841 [Google Scholar] [CrossRef] [PubMed]
[19]. Yu J, Luo Z, Wu P, Tang J, Novel design of the chimeric deep inferior epigastric artery perforator flap that provides for three-dimensional reconstruction of composite tissue defects of the heel in childrenOrthop Surg 2021 13:216-24.10.1111/os.1288733448701PMC7862167 [Google Scholar] [CrossRef] [PubMed]