Acute Pancreatitis (AP) is a prevalent and rapidly progressive inflammatory disorder of the pancreas, often presenting with abdominal pain and elevated serum pancreatic enzymes. The present case report emphasises the crucial need for accurately diagnosing the underlying cause of Hypertriglyceridemic Pancreatitis (HTGP) in patients who present without any pre-existing co-morbidities. A 16-year-old female presented with a three-day history of generalised abdominal pain, vomiting and low-grade fever, with no co-morbidities. Initial clinical assessment revealed signs of dehydration, tachypnoea, tachycardia and diffuse abdominal tenderness. Laboratory workups indicated a significant rise in amylase and lipase, hyperglycaemia, metabolic acidosis and pronounced Hypertriglyceridemia (HTG). Imaging studies corroborated the diagnosis of AP, revealing necrotic fluid collections. The patient was immediately started on plasmapheresis, aggressive intravenous fluid resuscitation, insulin infusion and supportive care, which included electrolyte correction, pain management and vigilant monitoring of vital signs. The autoantibody panel for type 1 diabetes, Antiglutamic Acid Decarboxylase antibodies (GAD) returned positive. Over the following days, the patient exhibited marked clinical improvement, with decreased abdominal tenderness, stabilisation of vital parameters and normalisation of biochemical abnormalities. Subsequent imaging confirmed the resolution of pancreatitis. Upon discharge, the patient was prescribed a regimen of insulin, statins, fenofibrate and saroglitazar for ongoing management of her lipid levels and diabetes. This case underscores that HTG may present as the first clinical manifestation of Type 1 Diabetes Mellitus. It also emphasises the critical importance of prompt and coordinated therapeutic interventions in managing HTGP, where early and effective treatment is crucial for optimising patient outcomes.
Case Report
A 16-year-old female presented to the Emergency Department with a three-day history of generalised abdominal pain, vomiting and low-grade fever. She denied experiencing diarrhoea, loose stools, or dysuria. Her past, family and medication histories were unremarkable. On physical examination, the patient was alert, oriented and appeared dehydrated. She had tachypnoea with a respiratory rate of 27 breaths per minute and a pulse rate of 122 beats per minute. Her temperature was 99.8°F, blood pressure was 90/60 mmHg and oxygen saturation was 97% on room air. The abdominal examination revealed diffuse tenderness with guarding, while other systemic examinations were unremarkable.
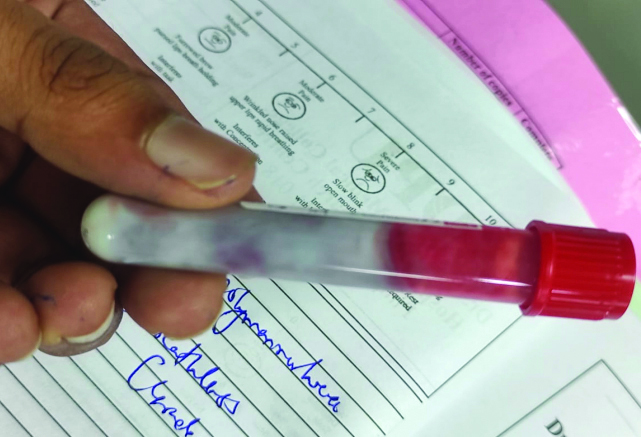
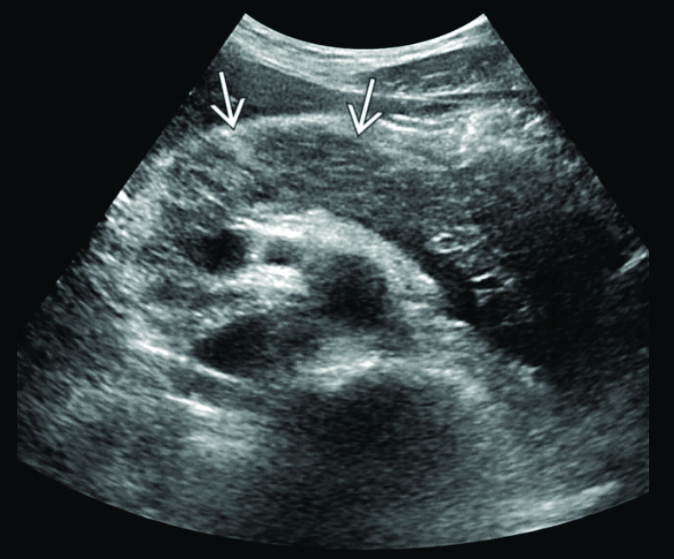
Laboratory investigations revealed a random blood glucose level of 360 mg/dL. The patient’s blood sample appeared milky white, suggesting lipemia [Table/Fig-1]. Arterial Blood Gas (ABG) analysis indicated uncompensated metabolic acidosis [Table/Fig-2] [1]. Routine laboratory tests showed elevated levels of amylase, lipase and TGs, along with ketonuria and glucosuria [Table/Fig-3,4] [1,2]. Ultrasonography of the abdomen and pelvis demonstrated a mildly bulky and heterogeneously hypoechoic body and tail of the pancreas with surrounding fat stranding. Ill-defined peripancreatic free fluid collections were also observed, consistent with AP [Table/Fig-5].
Lipemic serum of the patient.
Uncompensated metabolic acidosis on Arterial Blood Gas (ABG) analysis.
| Investigation | Day 1 | Normal value [1] |
|---|
| pH | 7.22 | 7.38-7.44 |
| PCO2 (mmHg) | 28 | 38-42 |
| PO2 (mmHg) | 106 | 75-100 |
| Glucose (mg/dL) | 332 | 70-99 |
| Lactate (mmol/L) | 1.4 | <1.3 |
| Bicarbonate (mmol/L) | 16 | 23-26 |
PCO2: Partial pressure of carbon dioxide; PO2: Partial pressure of oxygen
All routine investigations.
| Investigation | Day 1 | Normal value [1] |
|---|
| Complete blood count [2] |
| Haemoglobin (g/dL) | 13.6 | 12-16 |
| White blood cells (/microliter) | 16300 | 4500-11000 |
| Platelet count (/microliter) | 503000 | 150000-400000 |
| Haematocrit (%) | 33.4 | 36-46 |
| Mean corpuscular volume (FL) | 77.6 | 80-100 |
| Urea (mg/dL) | 60 | 17.12-42.8 |
| Creatinine (mg/dL) | 1.8 | 0.5-1.1 |
| Serum sodium (mmol/L) | 131 | 136-145 |
| Serum potassium (mmol/L) | 3.9 | 3.5-5 |
| Serum chloride (mmol/L) | 105 | 98-106 |
| Serum calcium (mg/dL) | 7.4 | 8.6-10.2 |
| Serum magnesium (mg/dL) | 1.9 | 1.6-2.6 |
| Serum phosphorous (mg/dL) | 1.8 | 3-4.5 |
| Serum amylase (U/L) | 1546 | 25-125 |
| Serum lipase (U/L) | 1875 | 10-140 |
| Investigation | Day 1 | Normal value [15] |
|---|
| HbA1c (%) | 12.9 | 4.0-5.6% |
| Urine routine (Dipstick method) | Protein- 3+, Glucose- 2+, Ketones- 1+ | AbsentAbsentabsent |
| Total cholesterol (mg/dL) | 349 | <200 |
| Triglycerides (TG) (mg/dL) | 2870 | <100 |
| HDL cholesterol (mg/dL) | 10 | <50 |
| LDL cholesterol (mg/dL) | 60 | <100 |
| C-reactive protein (mg/L) | 47.2 | ≤0.8 |
| Serum Lactate Dehydrogenase (LDH) | 450 | 80-225 |
| Erythrocyte sedimentation rate (mm/hour) | 56 | 0-20 |
| Total bilirubin (mg/dL) | 0.83 | 0.3-1 |
| Direct bilirubin (mg/dL) | 0.58 | 0.1-0.3 |
| Indirect bilirubin (mg/dL) | 0.25 | 0.2-0.7 |
| Aspartate aminotransferase (AST) (U/L) | 400 | 10-40 |
| Alanine aminotransferase (ALT) (U/L) | 30 | 10-40 |
| Alkaline phosphatase (U/L) | 81 | 30-120 |
The ultrasound of abdomen and pelvis which shows a mildly bulky and heterogeneously hypoechoic body and tail of the pancreas with surrounding fat stranding marked by the arrows suggestive of Acute Pancreatitis (AP).
Based on the clinical presentation, laboratory findings and radiological imaging, HTGP secondary to type 1 diabetes was suspected by the end of day 2, as the patient had elevated TG levels along with increased serum amylase and lipase levels. Given that she was a young patient with elevated Glycated Haemoglobin (HbA1c), further evaluation for type 1 diabetes was conducted. The Bedside Index for Severity in AP (BISAP) score is a clinical tool used to predict the severity and mortality risk associated with AP. The BISAP score includes five key parameters assessed within the first 24 hours of admission: B- Blood Urea Nitrogen (BUN) levels exceeding 25 mg/dL, I- Impaired Mental Status (indicated by a Glasgow Coma Scale score of less than 15), S- Systemic Inflammatory Response Syndrome (SIRS), A- Age greater than 60 years, and P- Evidence of pleural effusion identified through imaging studies. Higher scores correlate with an increased risk of severe pancreatitis and mortality [3]. The patient’s BISAP score was 2.
The Ranson score remains a key tool for predicting the severity and prognosis of AP, with scores taken at admission and those assessed within 48 hours. The scoring criteria are based on assessment at admission: Age >55 years, white blood cell count >16,000/mm3, blood glucose >200 mg/dL, serum Aspartate Aminotransferase (AST) >250 IU/L, and serum Lactate Dehydrogenase (LDH) >350 IU/L. Assessment within 48 hours includes: haematocrit drop >10%, BUN increase >5 mg/dL, calcium <8.0 mg/dL, partial pressure of oxygen (PaO2) <60 mmHg, base deficit >4 mEq/L and fluid sequestration >6 L. A higher score indicates a greater risk of mortality [4]. Her Ranson score was 3 at the time of admission, indicating a mortality risk of 15%.
Intravenous fluid therapy, insulin infusion and supportive care were promptly initiated for this patient [Table/Fig-6]. A Foley catheter was placed for urine output monitoring, and a nasogastric tube was inserted to prevent aspiration. The patient was kept Nil Per Oral (NPO). Plasmapheresis was performed, and further cycles were planned to reduce serum lipid levels.
Immediate management given on day 1.
| Treatment modality | Dosage |
|---|
| Intravenous fluid resuscitation | 20 mL/kg/hour bolus of 0.9% saline was given in the first two hours followed by maintenance fluid of 150-250 mL/hour was continued. Choice of fluids (0.45% saline or 5% dextrose) according to blood glucose levels were given |
| Insulin therapy | The patient was started on short-acting regular insulin infusion intravenously 0.1 unit/kg/hour, titrated according to hourly blood sugar levels |
| Analgesia | Round the clock analgesics were administrated. Ketorolac 30 mg i.v. TDS, Acetaminophen 1 gm TDS, Tramadol 50 mg i.v. when required |
| Antiemetics | Ondansetron 4 mg i.v. TDS |
| Electrolyte monitoring and correction | Intravenous potassium chloride correction was given when potassium <3.5 meq/L and potassium was monitored every four hourly |
The patient was closely monitored for any signs of worsening. She remained haemodynamically stable. Intake and output charting was conducted hourly. Potassium levels and urine ketones were assessed every four hours, while ABG analysis was conducted every six hours, with corrections administered, as needed.
By day 3, the abdominal examination still revealed diffuse tenderness, but she remained haemodynamically stable. Electrolytes, blood glucose levels and ABG were continuously monitored, and she maintained good urine output with ongoing intravenous hydration. Laboratory investigations were repeated [Table/Fig-7,8]. The Ranson’s score at 48 hours was 2, indicating no worsening of the illness.
Comparison of the laboratory values done on day 3, day 7 and day 15.
| Investigation | Day 3 | Day 7 | Day 15 |
|---|
| Haemoglobin (g/dL) | 13.60 | 12.80 | 12.20 |
| White blood cells (/microlitre) | 12,300 | 10,000 | 6800 |
| Platelet count (/microlitre) | 4,02,000 | 4,00,000 | 329000 |
| Haematocrit (%) | 35.20% | 38 | 40 |
| Mean corpuscular volume (FL) | 77.60 | 82.3 | 85.5 |
| Urea (mg/dL) | 30 | 40 | 32 |
| Creatinine (mg/dL) | 1.2 | 0.7 | 0.6 |
| Serum sodium (mmol/L) | 141 | 140 | 138 |
| Serum potassium (mmol/L) | 4.2 | 4.0 | 3.78 |
| Serum chloride (mmol/L) | 106 | 99 | 102 |
| Serum calcium (mg/dL) | 8.60 | 8.40 | 8.5 |
| Serum magnesium (mg/dL) | 2 | 1.6 | 2 |
| Serum phosphorous (mg/dL) | 3 | 2.2 | 2.8 |
| Serum amylase (U/L) | 912 | 256 | 40 |
| Serum lipase (U/L) | 879 | 423 | 66 |
| Serum Lactate Dehydrogenase (LDH) | 320 | 210 | 98 |
| AST | 289 | 104 | 38 |
| Triglycerides (mg/dL) | 1458 | 800 | 220 |
The Arterial Blood Gas (ABG) analysis comparison of day 3, 7 and 15.
| Variables | ABG on day 3 | ABG on day 7 | ABG on day 15 |
|---|
| pH | 7.38 | 7.42 | 7.4 |
| PCO2 (mmHg) | 34 | 38 | 40 |
| PO2 (mmHg) | 100 | 90 | 88 |
| Glucose (mg/dL) | 178 | 169 | 130 |
| Lactate (mmol/L) | 1.2 | 1.0 | 0.9 |
| Bicarbonate (mmol/L) | 22 | 26 | 24 |
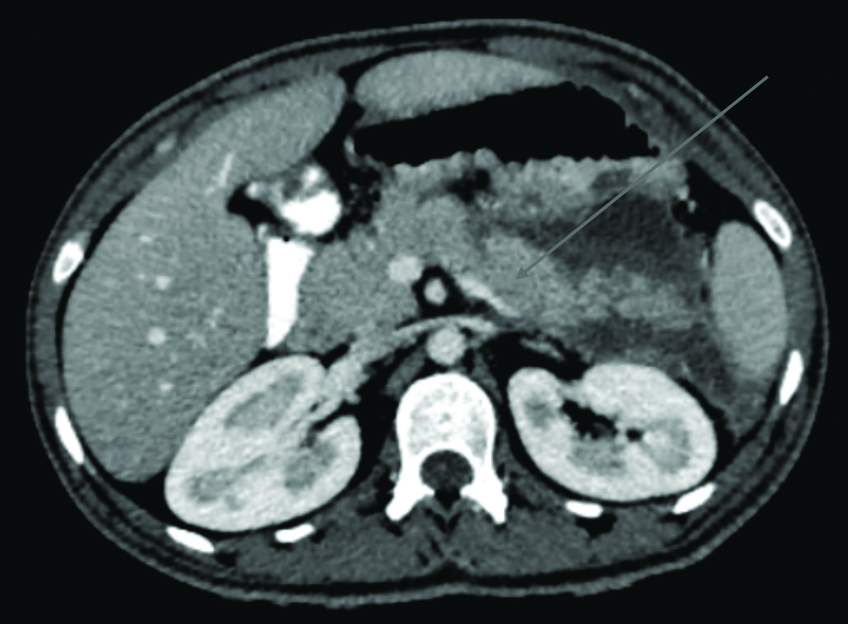
The Contrast-enhanced Computed Tomography (CECT) of the abdomen and pelvis revealed an enlarged liver (18.9 cm) with hypoattenuation, suggestive of diffuse hepatic steatosis. The pancreas was mildly bulky and oedematous with heterogeneous postcontrast enhancement. An ill-defined hypodense fluid collection was noted in the peripancreatic region, extending to the perigastric and splenorenal regions, with areas of heterogeneous enhancement indicative of an acute necrotic collection. Mild reactive wall thickening was observed in adjacent gastrointestinal structures. These findings were consistent with severe acute necrotising pancreatitis, with a modified CT severity score of 8/10 [Table/Fig-9].
Contrast-enhanced Computed Tomography (CECT) showing an ill-defined hypodense fluid collection noted in the peripancreatic region, extending to the perigastric and splenorenal regions, with areas of heterogeneous enhancement indicative of an acute necrotic collection suggestive of severe acute necrotising pancreatitis marked by the arrow with a modified CT severity score of 8/10.
On day 4, the patient remained vitally stable, and the same line of management was continued. On day 5, urine ketones were negative, and the abdomen was soft on examination. With the cessation of vomiting, the patient was started on an oral liquid diet, which she tolerated well; hence, her nasogastric tube was removed. She was then continued on a semisolid diet. Subcutaneous insulin therapy was initiated. Following three cycles of plasmapheresis, oral hypolipidemic medications were started. The autoantibody panel for type 1 diabetes, specifically anti-GAD antibodies, was sent to evaluate the diabetic cause, and the report was positive [Table/Fig-10].
The autoantibodies panel for type 1 diabetes.
| Investigation | Result |
|---|
| C-peptide (ng/mL) | 0.3 |
| Fasting insulin (mIU/mL) | 4 |
| Antiglutamic Acid Decarboxylase (GAD) antibodies | Positive |
| Anti-insulin autoantibodies | Negative |
| Islet cell cytoplasmic autoantibodies | Negative |
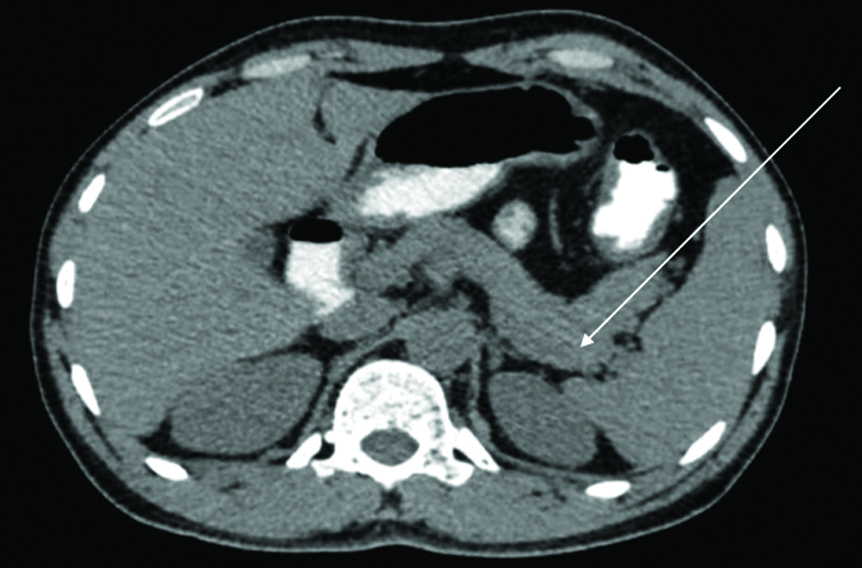
From day 5 until day 14, daily monitoring of electrolytes and blood glucose levels (five times per day) was conducted, and she was treated with subcutaneous insulin therapy according to her blood sugars. Oral hypolipidemic agents, intravenous hydration, along with oral hydration, and a soft diabetic, fat-free diet were continued, and the patient reported no complaints of abdominal discomfort during this period. By day 15, the patient had shown significant improvement. A repeat CECT of the abdomen and pelvis indicated resolving pancreatitis without any signs of complications [Table/Fig-11].
A repeat CECT of the abdomen and pelvis indicating resolving pancreatitis (marked by arrow) without any signs of complications.
The patient was discharged with a prescription for human Actrapid insulin, 12 units three times daily, and glargine, 10 units at night, administered subcutaneously. Additionally, the patient was prescribed hypolipidemic agents, including statins 40 mg and fenofibrate 160 mg at night, along with saroglitazar 4 mg once daily. The patient was also started on a low dose of prednisolone, 5 mg once daily, for the circulating anti-GAD antibodies.
During her follow-up, fasting and postprandial blood sugars, as well as, fasting TGs and other laboratory investigations, were monitored. Since recurrence is common in type 1 diabetes, it is essential to educate the young patient and family members about the warning signs, such as fever, fatigue, abdominal pain, vomiting, sweating, dizziness, tachycardia, blurred vision and steatorrhea, along with the importance of compliance with the treatment.
Discussion
The AP is an inflammatory disorder of the pancreas marked by abdominal pain and increased levels of pancreatic enzymes in the bloodstream as a result of injury to the pancreatic duct and acinar cells [5].
In a retrospective study conducted at Fujian Provincial Hospital, the incidence of HTG causing AP increased over ten years, rising from 8.4% in April 2012 to March 2013 to 22.3% in April 2020 to March 2021 [6]. According to data from the German Lipid League (DGFF), among 306 patients with severe HTG, 19% developed AP [7]. HTG is recognised as the third most prevalent cause of pancreatitis, with an incidence rate reaching as high as 7% in India [8]. AP due to HTG was observed in just 4% of adults experiencing Diabetic Ketoacidosis (DKA) episodes [9]. The present case study emphasises the importance of diagnosing the underlying cause, which is HTG, to prevent long-term complications.
Clinical examination and image-based diagnosis are used in AP. According to the revised Atlanta classification criteria, AP caused by HTG is diagnosed in the patient, if two out of the following three conditions are met: firstly, if the patient experiences a sudden onset of severe epigastric pain that often radiates to the back, or if there is an elevation in serum lipase or amylase to at least three times the upper limit of normal. Finally, imaging studies such as CECT, Magnetic Resonance Imaging (MRI), or transabdominal ultrasound may reveal features like homogeneous pancreatic parenchymal enhancement, with or without evidence of necrosis, and may be associated with a pseudocyst and other characteristic features of AP [10].
A similar case report describes a 14-year-old girl from Korea with DKA and a 7-year history of type 2 diabetes. The article presents the clinical manifestations, laboratory findings and management of HTG causing AP [11]. Another case reported in 2023 involved a 42-year-old woman with uncontrolled type 2 diabetes who developed DKA and severe HTG-induced pancreatitis [12]. A similar management approach is employed in the acute stages of illness. Unlike both studies, where HTG was due to type 2 diabetes mellitus, type 1 diabetes is associated with a higher incidence of recurrent HTG pancreatitis. The present case study stresses not only the importance of acute crisis management but also the necessity of long-term follow-up to reduce recurrence and morbidity.
The initial management of HTG-induced Pancreatitis (HTGP) involved supportive care and AP treatment, including fluid resuscitation with a 20 mL/kg/hour bolus of 0.9% saline, which was administered in the first two hours. This was followed by maintenance intravenous fluids at a rate of 150-250 mL/hour according to the patient’s requirements, based on blood sugar levels and electrolyte correction. Electrolyte levels were monitored every four hours, focusing on potassium and sodium levels. Pain management was provided with fentanyl. Restoring oral feeds, as soon as, symptoms subsided helped improve the patient’s nourishment and decrease the mortality rate in the first week of illness.
Lowering serum TG levels is crucial to prevent life-threatening complications, including cardiovascular diseases at a younger age; therefore, plasmapheresis along with hypolipidemic agents were utilised. The patient was treated with hypolipidemic agents, including fenofibrate, as fibrates are considered the first line of management. They help decrease Very Low-density Lipoprotein (VLDL) production, increase Fatty Acid (FA) oxidation, and enhance TG catabolism [13]. Additionally, they reduce diabetic microvascular complications [14]. The present case patient was not treated with low molecular weight heparin, as prolonged use raises concerns about rebound HTG and may disrupt the balance between Lipoprotein Lipase (LPL) production and degradation [15]. She was started on saroglitazar 4 mg once daily, as the activation of Peroxisome Proliferator-activated Receptorα (PPARα) by saroglitazar enhances the liver’s oxidation of FA and lowers TG synthesis and secretion [16].
Although there are no established guidelines for managing HTGP, insulin therapy has shown promising results in these patients, as it inhibits lipolysis and reduces TG production in the liver. The present case report underscores the importance of maintaining a high clinical suspicion when diagnosing HTGP, as it can present variably in cases of type 1 diabetes, and emphasises the need for early treatment initiation to decrease mortality in the initial phase, as well as, long-term morbidity.
Conclusion(s)
The HTG pancreatitis presenting as the first clinical manifestation of type 1 diabetes is relatively rare. The present case highlights the critical need for early recognition and management of HTGP in the context of DKA, particularly in young patients. The triad of AP, DKA and severe HTG presents a challenging clinical scenario that demands prompt intervention to prevent life-threatening complications. In the current case, a successful outcome was achieved through timely diagnosis, autoimmune studies, aggressive management with insulin therapy, plasmapheresis and supportive care, along with the use of hypolipidemic agents to control TG levels. The present case underscores the importance of a multidisciplinary approach in managing such complex conditions and emphasises the need for ongoing monitoring and tailored treatment to mitigate the risks of recurrence and long-term complications.
PCO
2: Partial pressure of carbon dioxide; PO
2: Partial pressure of oxygen
[1]. American Board of Internal MedicineABIM laboratory test reference ranges - January 2024 [Internet] 2024 Philadelphia (PA)American Board of Internal MedicineAvailable from: https://www.abim.org/laboratory-test-reference-ranges [Google Scholar]
[2]. Dean L, Blood groups and red cell antigens [Internet] 2005 Bethesda (MD)National Center for Biotechnology Information (US)Table 1, Complete blood count. Available from: https://www.ncbi.nlm.nih.gov/books/NBK2263/table/ch1.T1/ [Google Scholar]
[3]. Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA, Early prediction of mortality in acute pancreatitis: A large population-based studyGut 2008 57(12):1698-703.Epub 2008 Jun 210.1136/gut.2008.15270218519429 [Google Scholar] [CrossRef] [PubMed]
[4]. Basit H, Ruan GJ, Mukherjee S, Ranson criteriaIn: StatPearls [Internet] 2024 Jan Treasure Island (FL)StatPearls PublishingAvailable from: https://www.ncbi.nlm.nih.gov/books/NBK482345/[updated 2022 Sep 26] [Google Scholar]
[5]. Kong MT, Nunes MP, Leong KF, Diabetic ketoacidosis with acute severe hypertriglyceridaemia-induced pancreatitis as first presentation of type 2 diabetesBMJ Case Rep 2021 14(4):e23972710.1136/bcr-2020-23972733875498 [Google Scholar] [CrossRef] [PubMed]
[6]. Lin XY, Zeng Y, Zhang ZC, Lin ZH, Chen LC, Ye ZS, Incidence and clinical characteristics of hypertriglyceridemic acute pancreatitis: A retrospective single-center studyWorld J Gastroenterol 2022 28(29):3946-59.PMCID: PMC936723010.3748/wjg.v28.i29.394636157550 [Google Scholar] [CrossRef] [PubMed]
[7]. Valdivielso P, Ramírez-Bueno A, Ewald N, Current knowledge of hypertriglyceridemic pancreatitisEur J Intern Med 2014 25(8):689-94.Epub 2014 Sep 2710.1016/j.ejim.2014.08.00825269432 [Google Scholar] [CrossRef] [PubMed]
[8]. Rawla P, Sunkara T, Thandra KC, Gaduputi V, Hypertriglyceridemia-induced pancreatitis: Updated review of current treatment and preventive strategiesClin J Gastroenterol 2018 11(6):441-48.Epub 2018 Jun 1910.1007/s12328-018-0881-129923163 [Google Scholar] [CrossRef] [PubMed]
[9]. Wang Y, Attar BM, Bedrose S, Trick W, Rivas-Chicas O, Simons-Linares CR, Diabetic ketoacidosis with hypertriglyceridemia-induced acute pancreatitis as first presentation of diabetes mellitus: Report of three casesAACE Clin Case Rep 2017 3(3):e195-e199.10.4158/EP161389.CR [Google Scholar] [CrossRef]
[10]. Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Classification of acute pancreatitis--2012: Revision of the Atlanta classification and definitions by international consensusGut 2013 62(1):102-11.10.1136/gutjnl-2012-30277923100216 [Google Scholar] [CrossRef] [PubMed]
[11]. Park H, Kim MS, Kim J, Lee SM, Cho SY, Yoo EG, Hypertriglyceridemia with acute pancreatitis in a 14-year-old girl with diabetic ketoacidosisAnn Pediatr Endocrinol Metab 2022 27(1):73-77.Epub 2021 May 14. PMCID: PMC898475310.6065/apem.2040250.12534015902 [Google Scholar] [CrossRef] [PubMed]
[12]. Oshikoya AF, Kumari N, Bai M, Suman F, Haseeb M, Acute pancreatitis, hypertriglyceridemia, and diabetic ketoacidosis: A life-threatening triadCureus 2023 15(9):e45631PMCID: PMC1058897610.7759/cureus.4563137868435 [Google Scholar] [CrossRef] [PubMed]
[13]. Berglund L, Brunzell JD, Goldberg AC, Goldberg IJ, Sacks F, Murad MH, Evaluation and treatment of hypertriglyceridemia: An Endocrine Society clinical practice guidelineJ Clin Endocrinol Metab 2012 97(9):2969-89.10.1210/jc.2011-321322962670 [Google Scholar] [CrossRef] [PubMed]
[14]. Ruiz-García A, Arranz-Martínez E, Morón-Merchante I, Pascual-Fuster V, Tamarit JJ, Trias-Villagut F, Consensus document of the Spanish Society of Arteriosclerosis (SEA) for the prevention and treatment of cardiovascular disease in type 2 diabetes mellitusClin Investig Arterioscler 2018 30(Suppl 1):01-19.10.1016/j.arteri.2018.06.00630053980 [Google Scholar] [CrossRef] [PubMed]
[15]. Tolento Cortes L, Trinh J, Le M, Papayanis P, Tudtud-Hans L, Hong L, Safety and efficacy of insulin and heparin in the management of hypertriglyceridemia-induced pancreatitis in a patient without diabetes: A case reportCase Rep Endocrinol 2022 2022(1):790555210.1155/2022/790555236203643 [Google Scholar] [CrossRef] [PubMed]
[16]. Sosale A, Saboo B, Sosale B, Saroglitazar for the treatment of hypertriglyceridemia in patients with type 2 diabetes: Current evidenceDiabetes Metab Syndr Obes 2015 8:189-96.10.2147/DMSO.S4959225926748 [Google Scholar] [CrossRef] [PubMed]