Breast Solid Papillary Carcinoma (SPC) is an uncommon malignancy that primarily affects elderly women, with an incidence of 1-2%. It is morphologically characterised by well-defined nodules with low-grade nuclear features, which are associated with fibrovascular cores and neuroendocrine development. Although SPC can be invasive or in-situ, the prognosis is generally quite good. Its morphology can mimic several precancerous conditions, leading to frequent misdiagnosis. Distinguishing this tumour from its various morphological equivalents is more straightforward when an appropriate Immunohistochemistry (IHC) panel is utilised. A 45-year-old woman presented with a retroareolar mass in her right breast. Sono-mammography revealed a firm, incompressible mass with irregular borders, calcifications, cystic changes, and necrosis. A right-side simple mastectomy was performed, and the tumour was diagnosed as triple-negative high-grade SPC of the breast with focal invasion. The tumour’s origin was ruled out, and the diagnosis was confirmed by a prominent histopathologist. This report discusses a case of SPC, emphasising invasion patterns and evaluating previous knowledge.
Immunohistochemistry, Invasive carcinoma, Neuroendocrine differentiation, Triple-negative breast cancer
Case Report
A 45-year-old lady presented with an ulcerative retroareolar lesion measuring 7×7 cm in her right breast. She subsequently experienced pain, blood discharge, and nipple retraction. Sono-mammography suggested a firm, incompressible mass with uneven echotexture and irregular borders, as well as calcifications, cystic changes, and necrosis, all consistent with a Breast Imaging Reporting and Data System (BIRADS) IV lesion. A right simple mastectomy was performed, and the specimen was sent for histopathologic assessment.
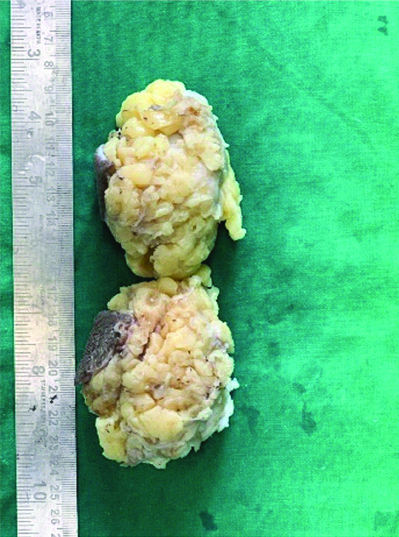
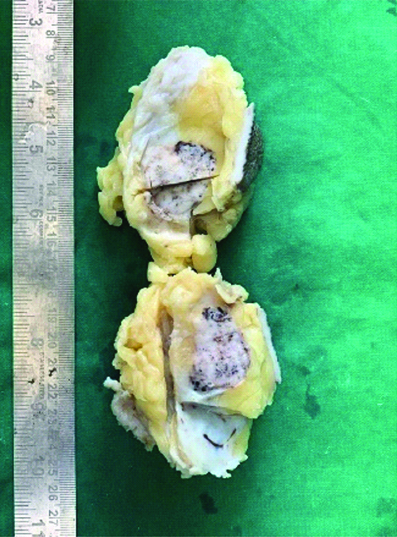
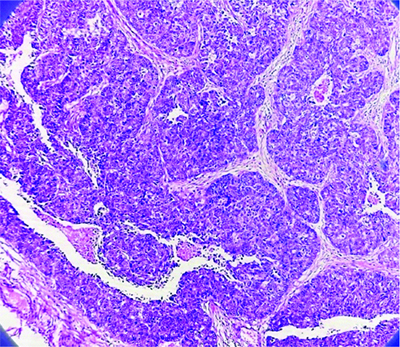
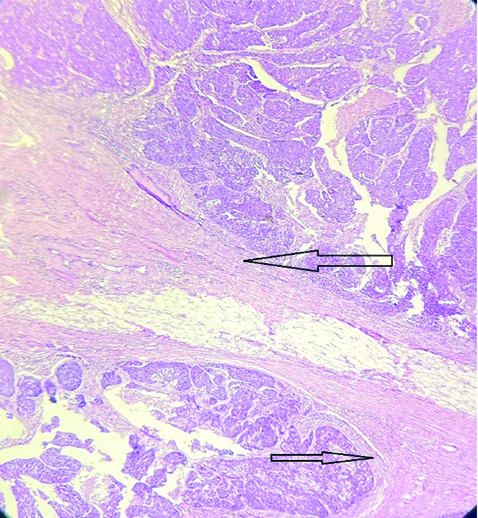
The gross specimen showed a local excision of breast tissue measuring 8×7×4 cm, with a skin flap measuring 6×3 cm [Table/Fig-1]. The cut section revealed a well-circumscribed grey-white to brownish lump with a granular solid cut surface. The adjacent breast tissue displayed patchy grey-white areas. The base margin was 1.5 cm away, while the rest of the resection margins were 1.5, 3, 5, and 3 cm away [Table/Fig-2]. Microscopically, the tumour was well-circumscribed, consisting of a central sclerotic area and solid nodules comprised of sheets of cells separated by fibrovascular septae [Table/Fig-3]. The cells had enlarged, pleomorphic, hyperchromatic, mitotically active nuclei with clumped chromatin and moderate eosinophilic cytoplasm. The adjacent breast tissue showed Lobular Carcinoma in-situ (LCIS) and focal invasion, with no lymphovascular emboli or perineural invasion [Table/Fig-4].
Gross specimen of right breast mastectomy. An 8×7×4 cm local excision of the breast can be observed in the material, along with a 6×3 cm skin flap.
The cut surface of the gross specimen. A well-circumscribed grey, whitish to brownish lump with a granular, firm-cut surface was visible in the part that had been sliced. Patchy regions of grey-white breast tissue were visible nearby. The remaining resection margins are spaced 1.5, 3, 5, and 3 centimeters from the base.
Low magnification microscopic picture of Solid Papillary Carcinoma (SPC) breast- A well-circumscribed tumour with solid nodules and a core sclerotic region is observed under a microscope. The cells exhibit larger, pleomorphic hyperchromatic nuclei with clumped chromatin and mild eosinophilic cytoplasm (4x).
Low Magnification (4x) of Solid Papillary Carcinoma (SPC) - A well-circumscribed tumour in breast tissue was seen under a microscope. It had solid nodules without lympho-vascular emboli or perineural invasion, and a central sclerotic region. Arrows are marked at areas of invasion. (Images from left to right)
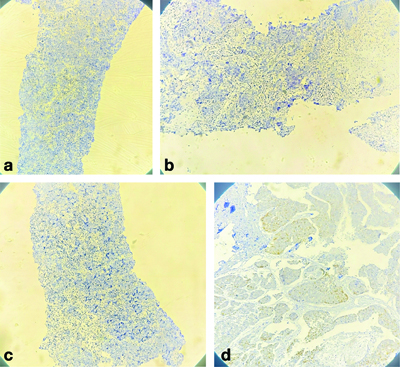
The results of IHC showed that the Oestrogen Receptor (ER), Progesterone Receptor (PR), and Human Epidermal growth factor Receptor 2/neu (HER2/neu) were negative. P63 was also negative at the perimeter of the nodule [Table/Fig-5]. The tumour was negative for Thyroid Transcription Factor-1 (TTF-1), PAX8, and CDX2 on the IHC study, thus ruling out thyroid, ovarian, and gastrointestinal origins. The diagnosis of pure type SPC with invasion was confirmed by negative results for all neuroendocrine markers, including CD56, chromogranin A, and synaptophysin. The tumour was diagnosed as triple-negative high-grade SPC of the breast with focal invasion (T3N0M0), with a Modified Bloom-Richardson score of the invasive components being 9 (3+3+3) and no lymphovascular emboli or perineural invasion [1]. The tumour’s origin was ruled out, and a second review by a senior histopathologist confirmed the diagnosis.
a) IHC Oestrogen Receptor (ER) showing negative for breast lesion (4x); b) IHC Progesterone Recepter (PR) showing negative for breast lesion (4x); c) IHC HER2/neu showing negative for breast lesion (4x); d) p63 negativity suggests that this lesion may be an invasive carcinoma, as myoepithelial cells typically express p63 (4x).
Discussion
The SPC of the breast is a unique intraductal papillary tumour that often affects women in their 70s [2]. It has a prevalence of 1-2% and distinct clinicopathological characteristics [3]. Cancer in-situ (SPC) is a type of cancer that often behaves indolently until it becomes associated with invasion [4]. The 5th edition of the 2019 World Health Organisation (WHO) classification divides papillary neoplasms into five categories: intraductal papilloma, papillary Ductal Carcinoma in-situ (DCIS), encapsulated papillary carcinoma, SPC (both in-situ and invasive), and invasive papillary carcinoma [5]. The WHO classification recognises SPC for staging purposes, even in cases with unclear or dubious invasive foci. A research by Guo S et al., showed that SPCs lack myoepithelial cells and can lead to local stromal/muscle infiltration, as well as lymph node or distant metastatic spread, which presents a diagnostic challenge for pathologists [6]. SPCs have an excellent prognosis, with few incidences of local recurrence, distant metastasis, and death [7]. According to a literature search on Medline, PubMed, and Scopus, which included 297 reported cases, 90% of cases involve localised lesions in pre- and postmenopausal females, with only 0.4% having upfront metastases [8]. This report discusses the case of a 45-year-old Indian female who have triple-negative SPC with focal invasion.
In 1956, Maluf HM and Koerner FC identified a specific breast lesion called “SPC,” primarily found in elderly women [9]. The WHO defines SPC as closely spaced expansile cellular nodules with delicate fibrovascular core and a solid development pattern. Pseudo-rosettes and nuclear palisading encircling stromal centers are visible in these confined nodules packed with neoplastic cells, which frequently exhibit low to intermediate nuclear grades. These nodules are considered non invasive. They may display low-grade cytological traits, Neuroendocrine Differentiation (NED), or a mucinous invasive component. SPCs appear as circumscribed nodules filled with neoplastic cells, often monomorphic with low to intermediate nuclear grades. Notably, they differ by forming circumscribed nodules that obscure the underlying papillary features and the solid cellular development of cancerous cells, supported by fibrovascular cores [5]. This may bear similarities to other disorders such as DCIS, lobular neoplasia, florid ductal hyperplasia, and intracystic papillary carcinoma [10].
SPC tumours are detected using mammography and ultrasonography, with 50% being occult [11]. Ultrasonography identifies BIRADS-IV or BIRADS-V lesions, while Magnetic Resonance Imaging (MRI) has high sensitivity due to its circumscribed margins, heterogeneous signal intensity, and high apparent diffusion coefficient values [12]. Microscopically, SPCs present as circumscribed nodules filled with neoplastic cells, often monomorphic with low to intermediate nuclear grades, appearing non invasive due to their circumscription, and occasionally displaying pseudo-rosettes and nuclear palisading [13]. SPC is characterised by intracellular mucinous differentiation and extracellular mucin synthesis, but it must be distinguished from invasive mucinous carcinoma. Capella type B mucinous carcinoma can resemble SPC, particularly when mucin synthesis decreases [10]. NED is a crucial IHC characteristic for detecting breast cancer, as it is observed in over half of all reported SPC cases [14]. CK5/6 immunopositivity and NED help differentiate SPC from other common breast neoplasms [15]. Breast biomarkers indicate that SPCs have a luminal phenotype, expressing PR and ER receptors but lacking HER2. They possess a basic genome with minor modifications.
Additional genetic traits include the deletion of chromosome 16q and the gain of chromosomes 1p and 16p. Furthermore, SPCs express the genes DOC7.23, ASCL1, and RET, which are implicated in neuroendocrine development [14]. Among the previously reported 297 cases, only 15 were triple-negative, and only three of these exhibited high-grade cytological characteristics [7]. Due to its triple-negative status, localised invasion, and lack of NED, this particular case is distinct. Invasive Lobular Carcinomas (ILCs) with a solid-papillary growth pattern, similar to solid and encapsulated papillary carcinoma, are characterised by the absence of NED, the presence of in-situ lobular carcinoma, and localised merging of solid-papillary areas with conventional lobular carcinoma. Both solid-papillary and classic lobular components exhibit similar CDH1 mutations and copy number changes. The diagnosis of ILC is validated by the presence of deleted myoepithelium, positive cytoplasmic P120, and negative E-cadherin in IHC [16].
Since, there is no relationship between the type of surgery and the likelihood of recurrence, local therapy is the best course of treatment for solid papillary breast cancer. However, treatment rates vary significantly, with mastectomy rates ranging from 7-88% [17]. Mastectomy and removal of axillary lymph nodes should be avoided in cases of small, well-defined solid papillary breast cancer. Pure solid papillary breast cancer responds best to adequate local excision, with or without hormone therapy [17]. Present study findings emphasise the need to adopt a broad IHC panel to consistently differentiate solid papillary carcinoma from other breast neoplasms. Given [Table/Fig-6], highlight the key features of differentials that can mimic a solid papillary neoplasm of the breast [10].
Differentiating breast carcinomas mimicking Solid Papillary Carcinomas (SPC).
| Feature | Solid PapillaryCarcinoma (SPC) | Invasive Ductal Carcinoma (IDC) | Encapsulated Papillary Carcinoma (EPC) | Neuroendocrinecarcinoma | Lobularcarcinoma | Tubularcarcinoma |
|---|
| Histological appearance | Predominantly solid growth pattern with delicate fibrovascular cores | Irregular, duct-forming invasive growth | Encapsulated, papillary structures | Neuroendocrine Differentiation (NED), solid nests | Single-file pattern, lack of tubule formation | Well-formed tubules, open lumina |
| Cellular features | Uniform cells, neuroendocrine-like appearance | Variable, pleomorphic cells | Uniform cells, less pleomorphic | Monomorphic cells with granular cytoplasm | Small, uniform cells, often signet-ring | Small, regular cells, angulated nuclei |
| Myoepithelial cells | Absent or very limited at the periphery | Absent in invasive areas | Present within and around the encapsulated lesion | Absent | Absent | Absent |
| Mucin production | Sometimes present | Rarely present | Often present | Common, in Neuroendocrine Differentiation (NED) | Rare | Absent |
| Immunohistochemistry | ER/PR positivity, CK7, chromogranin, synaptophysin | ER/PR positive, HER2 variable | ER/PR positive, CK7, chromogranin | Strong chromogranin, synaptophysin positivity | E-cadherin negative, ER/PR positive | ER/PR positive, HER2 negative, CK7 |
| Stromal reaction | Desmoplastic response | Desmoplastic stroma, varied response | Minimal to absent | Desmoplastic or none | Minimal, often targetoid spread | Minimal to absent |
| Clinical presentation | Often in older women, slow-growing | Variable, can be aggressive | Older women, indolent | Older women, slow-growing | Often bilateral, multicentric | Often in younger women, indolent |
| Prognosis | Generally favourable | Variable, depends on grade and stage | Favourable | Variable, depends on differentiation | Favourable, depends on stage | Excellent |
Conclusion(s)
The SPC is a rare and distinct type of breast cancer characterised by well-defined nodules and low-grade nuclear features. Diagnosis is challenging due to its similarity to other breast conditions. A comprehensive immunohistochemical panel is needed to reliably differentiate SPC from other breast neoplasms. Diagnostic markers include the absence of myoepithelial cells and NED. SPCs have a favourable prognosis, with a low chance of local recurrence or distant metastasis. However, therapy should be adjusted based on the tumour’s invasiveness. Local excision is the best treatment for non invasive SPC, while more aggressive surgical interventions may be necessary for invasive cases. Further research and case reporting are needed to enhance knowledge and management of this specific type of breast cancer.
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Jul 08, 2024
Manual Googling: Jul 26, 2024
iThenticate Software: Aug 16, 2024 (17%)
[1]. Meyer JS, Alvarez C, Milikowski C, Olson N, Russo I, Russo J, Breast carcinoma malignancy grading by Bloom-Richardson system vs proliferation index: Reproducibility of grade and advantages of proliferation indexMod Pathol 2005 18(8):1067-78. [Google Scholar]
[2]. Kuroda N, Fujishima N, Inoue K, Ohara M, Mizuno K, Lee GH, Solid papillary carcinoma of the breast: Imprint cytological and histological findingsMed Mol Morphol 2010 43(1):48-52. [Google Scholar]
[3]. Nassar H, Qureshi H, Adsay NV, Visscher D, Clinicopathologic analysis of solid papillary carcinoma of the breast and associated invasive carcinomasAm J Surg Pathol 2006 30(4):501-07. [Google Scholar]
[4]. Nicolas MM, Wu Y, Middleton LP, Gilcrease MZ, Loss of myoepithelium is variable in solid papillary carcinoma of the breastHistopathology 2007 51(5):657-65. [Google Scholar]
[5]. Tan PH, Ellis I, Allison K, Brogi E, Fox SB, Lakhani S, The 2019 World Health Organization classification of tumours of the breastHistopathology 2020 77(2):181-85. [Google Scholar]
[6]. Guo S, Wang Y, Rohr J, Fan C, Li Q, Li X, Solid papillary carcinoma of the breast: A special entity needs to be distinguished from conventional invasive carcinoma avoiding over-treatmentBreast 2016 26:67-72. [Google Scholar]
[7]. Otsuki Y, Yamada M, Shimizu S, Suwa K, Yoshida M, Tanioka F, Solid-papillary carcinoma of the breast: Clinicopathological study of 20 casesPathol Int 2007 57(7):421-29. [Google Scholar]
[8]. Jadhav T, Prasad SS, Guleria B, Tevatia MS, Guleria P, Solid papillary carcinoma of the breastAutops Case Rep 2022 12:e2021352 [Google Scholar]
[9]. Maluf HM, Koerner FC, Solid papillary carcinoma of the breast. A form of intraductal carcinoma with endocrine differentiation frequently associated with mucinous carcinomaAm J Surg Pathol 1995 19(11):1237-44. [Google Scholar]
[10]. Rakha EA, Ellis IO, Diagnostic challenges in papillary lesions of the breastPathology 2018 50(1):100-10. [Google Scholar]
[11]. Joseph B, Shah BA, Shi D, Solid papillary carcinoma of the breast: Mammographic and ultrasound appearance with histopathologic correlationAnn Rev Res 2019 5(3):555662Available from: https://juniperpublishers.com/arr/ARR.MS.ID.555662.php [Google Scholar]
[12]. You C, Peng W, Shen X, Zhi W, Yang W, Gu Y, Solid papillary carcinoma of the breast: Magnetic resonance mammography, digital mammography, and ultrasound findingsJ Comput Assist Tomogr 2018 42(5):771-75. [Google Scholar]
[13]. Clement Z, Jones M, Solid papillary carcinoma of the breast: A reviewInt J Surg Med 2017 3(1):57-59. [Google Scholar]
[14]. Bogina G, Munari E, Brunelli M, Bortesi L, Marconi M, Sommaggio M, Neuroendocrine differentiation in breast carcinoma: Clinicopathological features and outcomeHistopathology 2016 68(3):422-32. [Google Scholar]
[15]. Li X, Lin M, Xu J, Pang Y, Chen H, Sun P, New variant of breast-invasive lobular carcinoma with solid and encapsulated papillary carcinoma growth patternBreast Cancer 2021 28(6):1383-88. [Google Scholar]
[16]. Rakha EA, Gandhi N, Climent F, van Deurzen CH, Haider SA, Dunk L, Encapsulated papillary carcinoma of the breast: An invasive tumour with excellent prognosisAm J Surg Pathol 2011 35(8):1093-103. [Google Scholar]
[17]. Tsang WY, Chan JK, Endocrine ductal carcinoma in situ (E-DCIS) of the breast: A form of low-grade DCIS with distinctive clinicopathologic and biologic characteristicsAm J Surg Pathol 1996 20(8):921-43. [Google Scholar]