The Subclinical Hypothyroidism (SCH) is defined as an increased serum Thyroid Stimulating Hormone (TSH) level above the upper limit of the normal range, while serum free thyroxine and tri-iodothyronine levels remain within the population reference range [1]. The incidence of SCH increases with age, leading to a higher prevalence. SCH cases are characterised by mildly elevated TSH levels, typically ranging from 4.0 to 10.0 mU/L [2]. Cardiovascular abnormalities associated with SCH include left ventricular diastolic dysfunction, decreased resting and exertional systolic function, increased vascular resistance, arterial stiffness, endothelial dysfunction, and atherosclerosis [2-4]. The hypothalamus serves as the controlling centre for both the thyroid gland and the Autonomic Nervous System (ANS), establishing a close link between them and their effects on the cardiovascular system [5].
The heart is primarily regulated by the ANS, with an increase in Heart Rate (HR) resulting from the sympathetic division and a decrease in HR caused by the parasympathetic division. Fluctuations in heartbeat intervals arise from a synchronous and complex mechanism involving both branches of the ANS. Good interbeat variation indicates a strong capacity to adapt and respond to internal and external stimuli, reflecting effective neurovegetative control and enhanced cardiovascular functionality [6,7].
However, there are limited studies available that demonstrate cardiac autonomic dysfunction in patients with SCH. Some authors have reported hypofunctional parasympathetic systems by analysing time and frequency domains in HRV [15,16], while others have indicated an increased sympathetic tone and decreased HRV, suggesting a reduction in vagal tone [17]. This scarcity of reports on SCH presents contradictory findings. Therefore, present study was undertaken to evaluate any autonomic function abnormalities in patients with SCH.
Materials and Methods
A cross-sectional analytical study was conducted after obtaining ethical clearance (Memo No. RKC/617) from the Institutional Ethics Committee (IEC) in the Department of Physiology at R.G. Kar Medical College and Hospital, Kolkata, in collaboration with the Department of Endocrinology at R.G. Kar Medical College, Kolkata, West Bengal, India. The study took place from November 2022 to October 2023. The sample population consisted of 50 newly diagnosed patients with SCH cases, who presented with complaints such as weight gain, fatigue, and menstrual abnormalities in the Outpatient Department of the Department of Endocrinology. Additionally, 50 healthy individuals (controls) who accompanied patients of a similar age group and had no apparent complaints or co-morbidities were included.
Subclinical Hypothyroidism (SCH) is defined as an increased serum TSH value within the range of 4.0-10.0 mU/L, with T3 and T4 levels remaining within the population reference range [2]. The HRV test was conducted using a Physiograph Polyrite-D instrument equipped with bioamplifiers, four channels, and accessories (RMS latest software-version 1.2.5).
Sample size calculation: Sample size was calculated using the formula:
N=(Z1-α/2)2×p×q/L2Where, N=sample size
p=Expected prevalence of SCH=41% [18],
q=(100-p),
L=absolute Precision=10%
Z1-α/2=Standard normal deviate of value 1.96 considering 95% confidence interval, ‘q’ thus was taken to be (100-41)% i.e., 59%,
So, sample size N=(1.96×1.96)×41×(100-41)/10×10=92(approx.)
Final sample size of 100 was taken out of which 50 were taken as newly diagnosed SCH patients and fifty were healthy individuals.
Inclusion criteria for cases, both male and female participants aged 18 to 55 years with SCH were included and a healthy control group with no apparent complaints or co-morbidities, who accompanied the patients and were also aged between 18 and 55 years, was included in the study.
Exclusion criteria for cases and controls: Individuals were excluded, if they met any of the following criteria:
The SCH patients on levothyroxine
Patients with overt hypothyroidism
Individuals younger than 18 years or older than 55 years
Known cases of diabetes mellitus
Individuals with any underlying cardiovascular illnesses, such as arrhythmia, rheumatic heart disease, ischaemic heart disease, or cardiac failure
Patients with respiratory diseases, including bronchial asthma, pulmonary tuberculosis, or chronic obstructive pulmonary disease
Individuals with psychological illnesses
Patients with any surgical trauma or recent surgery
Patients taking medications such as anti-arrhythmics, diuretics, beta-blockers, or vasodilators that affect ANS
Severely ill patients who were unable to walk
Pregnant or lactating women
Individuals unwilling to participate or who did not provide consent
The cases were categorised based on the relationship between SCH and HRV evaluation, highlighting the underlying cardiovascular risk among the subjects. The HRV parameters used included:
HF band: 0.15-0.4 Hz [19]
LF band: 0.04-0.15 Hz [20]
An LF/HF ratio less than one indicates a parasympathetic drive and good cardiovascular health, while an LF/HF ratio greater than one indicates sympathetic overdrive [19].
An SDNN value <50 ms indicates a high-risk for cardiovascular disease and sympathetic drive [19].
An SDNN value between 50-100 ms indicates a moderate risk for cardiovascular disease [19].
An SDNN value >100 ms indicates normal values and a parasympathetic drive [19].
Study Procedure
The study parameters included age, waist-hip ratio, Body Mass Index (BMI), Systolic Blood Pressure (SBP), Diastolic Blood Pressure (DBP), TSH values, LF/HF ratio, and SDNN.
Time domain: This section involves a statistical analysis of the HRV report, which includes time domain parameters such as SDNN (Standard Deviation of NN intervals) and SDANN (Standard Deviation of the Average NN Intervals). Time domain analysis measures HRV over a specific period or the intervals between each cardiac cycle. A continuous Electrocardiogram (ECG) recording was taken, capturing each QRS complex (which represents ventricular depolarisation). The normal RR intervals, resulting from instantaneous heart rate measurements, were obtained. After eliminating artifacts such as ectopic beats, abnormal beats, omitted heartbeats, and machine noise, the normal sinus rhythm intervals, or NN intervals, were recorded. The standard deviation of RR or NN intervals is typically calculated to assess overnight fluctuations in HRV. SDNN serves as an index of HRV and reflects circadian rhythms as well as long-term components responsible for the variability of heart rate during the ECG recording period.
Frequency domain: This section includes HRV parameters such as LF (Low Frequency), HF (High Frequency), and the LF/HF ratio. It involves a power spectral analysis of the HRV report, signifying periodic oscillations of heart rate that exhibit different frequencies with varying amplitudes from the sinus rhythm. The Fast Fourier Transformation (FFT) is a simple and rapid method used to examine the frequency domain or spectral density of HRV. This analysis can differentiate individual R-R intervals and identify discrete peaks for various frequency components of the heart’s sinus rhythm. These components are transformed into bands with different spectral frequencies, which are stored in a computer. The power spectrum is divided into four bands: HF, LF, Very Low Frequency (VLF), and Ultra-low Frequency (ULF), covering a frequency range between 0-0.5 Hz [19].
Data collection: A detailed history was taken, and blood reports were collected from each subject. After obtaining informed consent from each subject, a general physical examination was conducted, and the details were recorded. Hip and waist sizes were measured in centimeters, and the ratio was calculated. Subjects were instructed to refrain from consuming any drinks containing caffeine for atleast 12 hours before the test. They were also advised to avoid solid food for two hours prior to the test and to discontinue any sedative or central nervous system-affecting drugs beforehand. Additionally, subjects were asked to refrain from smoking for 24 hours before the test. They were instructed to wear loose, comfortable clothing, as tight clothes and metallic objects were not permitted. Each subject was asked to evacuate their bladder before the test.
The test was performed in the morning according to standard guidelines under normal room temperature. Autonomic function tests were conducted using the Physiograph Polyrite-D instrument, equipped with bioamplifiers, four channels, and accessories (RMS latest software - version 3.0.16) in the autonomic function research laboratory of the Department of Physiology at the study Institute. The subjects were made comfortable and familiarised with the testing area. They were instructed to lie down in a supine position for 15 minutes before the study.
After proper counseling and adequate rest, subjects were asked to relax and remain calm during the examination, avoiding specific activities such as sleeping, vigorous movements, emotionally overwhelming situations, coughing, sneezing, or any other movements during the examination. Both upper limbs were exposed upto the elbows, and the left lower limb was exposed upto the knee. The exposed areas of the limbs were thoroughly cleaned with an alcohol swab. All clamp-type limb electrodes (for the right hand, left hand, and left foot) were attached using electroconductive jelly. Hand electrodes were placed around the wrists, and a foot electrode was positioned just above the ankle of the left foot. All electrodes were connected to the bio-amplifier of the Physiograph Polyrite-D via a transducer mechanism. The resting HR was recorded. Frequency domain (spectral) and time domain analysis of short-term (5 minutes) HRV was conducted. There was no single HRV number; to ensure the reliability of the test, the most common HRV parameters, such as SDNN and the LF/HF ratio, were considered in the study. SDNN is regarded as the gold standard for evaluating cardiac risk, as it reflects the overall activity of the ANS. Parameters like Myocardial Infarction (MI), autonomic dysregulation, and mental conditions such as depression and anxiety are reflected in the SDNN parameter [20]. The LF/HF ratio is included as it is a widely used parameter in the assessment of autonomic function [20].
Statistical Analysis
Relevant data were collected, and analysis was conducted using SPSS version 20.0. An unpaired Student’s t-test was used for continuous variables, while the Chi-square test was used for categorical variables. Pearson’s correlation tests were also performed. A p-value of <0.05 was considered statistically significant.
Results
There was no significant difference in age, height, weight, Body Mass Index (BMI), or Waist-Hip Ratio between the cases and controls. However, the TSH value showed a significant difference between the cases (7.46±1.46 mU/L) and the controls (2.33±0.94 mU/L) [Table/Fig-1].
Comparison of baseline characteristics between the cases and control.
| Variables | Cases | Controls | p-value |
|---|
| Age (years) | 31.82±8.91 | 32.52±8.00 | 0.68 |
| Height (cm) | 158.58±5.9 | 159.28±6.41 | 0.57 |
| Weight (kg) | 63.76±7.94 | 62.43±7.82 | 0.40 |
| BMI (kg/m2) | 25.31±3.02 | 24.57±2.38 | 0.17 |
| Waist (cm) | 88.16±10.77 | 86.26±6.62 | 0.29 |
| WHR (cm) | 0.88±0.06 | 0.87±0.01 | 0.24 |
| TSH (mU/L) | 7.46±1.46 | 2.33±0.94 | 0.0001 |
| Gendern (%) | Male | 12 (24%) | 28 (56%) | 0.0012# |
| Female | 38 (76%) | 22 (44%) |
Student t-test was used. Value presented as Mean±SD
The present study [Table/Fig-2] indicated that HR (80.44±5.03 mmHg), SBP (122.04±5.15 mmHg), and DBP (78.36±4.56 mmHg) in the cases were not significantly different from those in the controls {HR (81.62±9.76 per min), SBP (124.60±12.86 mmHg), DBP (77.30±9.05 mmHg)}.
Comparison of study parameters between the cases and control.
| Variables | Cases (mean±SD) | Control (mean±SD) | p-value |
|---|
| HR (per min) | 80.44±5.03 | 81.62±9.76 | 0.45 |
| SBP (mmHg) | 122.04±5.15 | 124.60±12.86 | 0.19 |
| DBP (mmHg) | 78.36±4.56 | 77.30±9.05 | 0.46 |
| LF power (Hz) | 42.79±18.31 | 30.28±13.01 | 0.0002 |
| HF power (Hz) | 34.88±14.07 | 34.72±13.95 | 0.96 |
| LF/HF | 1.28±0.75 | 0.90±0.32 | 0.002 |
| SDNN (ms) | 40.63±21.89 | 79.86±15.36 | 0.0001 |
Unpaired Student’s t-test was used
The LF/HF ratio was significantly higher (p-value=0.002) in the newly diagnosed SCH cases (1.28±0.75) compared to the controls (0.90±0.32). The SDNN in the cases (40.63±21.89) was also significantly lower (p-value=0.0001) than in the controls (79.86±15.36) [Table/Fig-2].
Approximately 72% of newly diagnosed SCH cases had an LF/HF ratio (2.33±5.45) of more than one, while 28% of cases had an LF/HF ratio (0.81±0.18) of less than 1 [Table/Fig-3].
Proportion between the newly diagnosed SCH cases according to LF/HF ratio.
| LF/HF ratio | n (%) | LH/HF (mean±SD) |
|---|
| ≥1 | 36 (72%) | 2.33±5.45 |
| <1 | 14 (28%) | 0.81±0.18 |
About 78% of newly diagnosed SCH cases had an SDNN of less than 50 ms (31.19±10.69), 20% had between 50-100 ms (70.39±15.14), and 2% of cases had more than 100 ms [Table/Fig-4].
Proportion between the newly diagnosed Subclinical Hypothyroidism (SCH) cases according to SDNN value.
| Groups | n (%) | SDNN (mean±SD) |
|---|
| High-risk (<50 ms) | 39 (78%) | 31.19±10.69 |
| Moderate risk (50-100 ms) | 10 (20%) | 70.39±15.14 |
| Normal value (>100 ms) | 1 (%) | 109.681 |
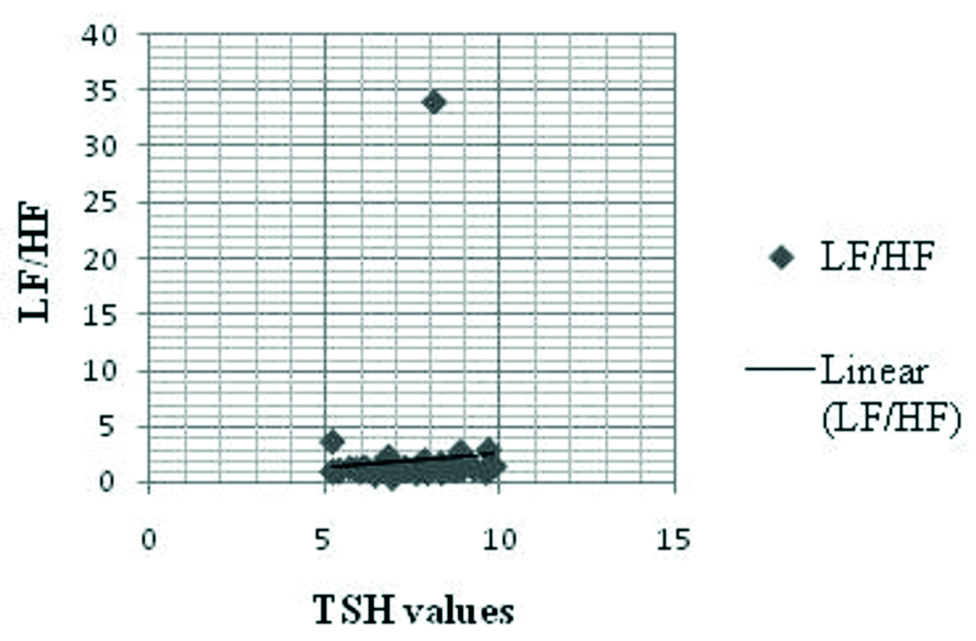
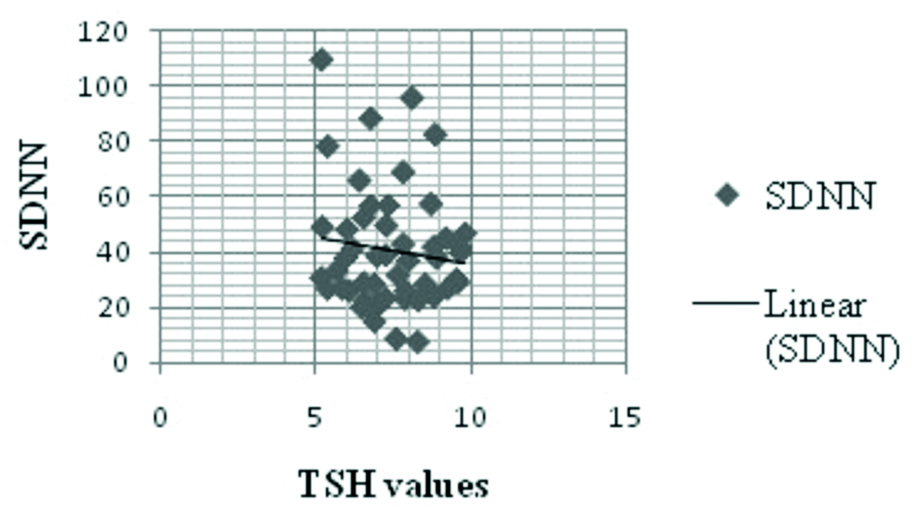
The LF/HF ratio had a positive correlation with TSH (r=0.09, p=0.57) [Table/Fig-5,6]. Conversely, SDNN had a negative association with TSH (r=-0.08, p=0.58) among the cases [Table/Fig-5,7].
Correlation of LF/HF ratio and SDNN with TSH values among the cases.
| TSH | Pearson’s coefficient (r) | p-value |
|---|
| LF/HF | 0.09 | 0.57 |
| SDNN | -0.08 | 0.58 |
*p-value of <0.05 is significant
Relationship between LF/HF ratios with TSH value among the cases.
Relationship between SDNN with TSH value among the cases.
Discussion
The newly diagnosed SCH patients in present study were found to have a predisposition to cardiac autonomic neuropathy compared to the healthy control group. In the present study, HR, SBP and DBP among the newly diagnosed SCH cases were found to be within the normal range, and these values were statistically insignificant when compared to the healthy control group. A study by Kalra P et al., found that SBP and DBP were comparable between the cases and controls, which is consistent with the findings of the present study. There was no significant difference in SBP and DBP between the two groups. The BP was recorded to be within the normal physiological range in the SCH group [21]. Another study by Mahajan AS et al., found that SCH patients did not exhibit hypertension, even in those with lower free T4 (fT4) levels. The correlation analysis conducted in their study revealed a significant positive correlation between systolic BP and age in SCH patients, while fT4 levels showed a significant negative correlation with systolic BP [18]. A study by Walsh JP et al., also reported that SCH was not associated with hypertension [22]. Conversely, Manolis AA et al., concluded that SCH is characterised by an increased association with cardiovascular risk factors, including hypertension [23]. According to a study by Brusseau V et al., an increase in systolic BP was associated with lower high-frequency power, and an increase in diastolic BP was associated with lower low-frequency power [24].
In the present study, a significant increase in the LF/HF ratio was found in newly diagnosed SCH patients compared to the healthy control group. This suggests that SCH patients have increased sympathetic activity and lower vagal tone compared to the healthy control group. The sympathetic dysfunction may lead to future cardiac abnormalities, such as autonomic neuropathy, in SCH patients. Galetta F et al., performed an HRV test in SCH patients and reported an increase in the LF/HF ratio, indicating higher sympathetic activity [17]. Brusseau V et al., also concluded that in SCH patients, HRV parameters were markedly decreased. Vagal inhibition was more pronounced than the increase in sympathetic activity, with a more significant decrease in high-frequency power compared to low-frequency power. High-frequency power is the main contributor, accounting for about two-thirds of the decrease, leading to a reduction in vagal control of the heart, while low-frequency and very low-frequency power contribute about one-third. The decrease in HRV parameters is attributed to a substantial decline in cardiac vagal activity [24].
The increased sympathetic activity and decreased parasympathetic activity may have clinical significance, as they are associated with metabolic dysfunction, which can progress to a higher risk of cardiovascular disease. Metabolic abnormalities can occur due to prolonged stress, resulting in high blood pressure, elevated serum triglycerides, high serum glucose, increased waist circumference, and low High-density Lipoprotein (HDL) cholesterol [25]. Hoshi RA et al., concluded that SCH patients presented with higher sympathetic and lower vagal tones while in a supine resting position, with blunted sympathetic autonomic responses to active postural changes [26]. On the contrary, Sahin I et al., concluded that there is no difference in HRV parameters, such as the time and frequency domains, among SCH patients compared to patients with TSH levels less than ten. However, they found a decrease in sympathetic tone among those with TSH levels greater than ten [27]. In the present study, 72% of SCH cases had an LF/HF ratio of more than one, while 28% had an LF/HF ratio of less than one. An LF/HF ratio of more than one indicates sympathetic drive, whereas an LF/HF ratio of less than one indicates parasympathetic drive [19]. Thus, the study showed a predominance of sympathetic drive and a hypofunctional parasympathetic system among SCH cases. Mahajan AS et al., found that sympathetic function abnormalities were more common in SCH, although decreased parasympathetic activity was also present. Their study concluded that sympathetic dysfunction was present in 59% of SCH patients, parasympathetic function abnormalities were present in 13.63% of cases, and 22.72% had both sympathetic and parasympathetic activity abnormalities [18]. In a study by Kalra P et al., the correlation between TSH and the LF/HF ratio in cases was not found to be significant (p=0.75, r=0.04), which corroborates with the present study [21]. In contrast, another study by Galetta F et al., observed that in SCH patients, the LF/HF ratio was significantly positively related to TSH parameters [17].
The present study found that the SDNN parameter was significantly lower in SCH cases compared to the control group, indicating that HRV was markedly reduced in newly diagnosed SCH cases. The lower SDNN values in SCH patients predispose them to an increased risk of cardiovascular disease. Galetta F et al., concluded that SDNN values were lower in SCH patients in reference to healthy controls [17]. Similarly, Brusseau V et al., noted strong evidence that SDNN was significantly lower in hypothyroid patients compared to healthy controls [24]. In the present study, it can be concluded that 78% of cases were at high-risk for cardiovascular disease, 20% were at moderate risk, and 2% were classified as normal. Shaffer F and Ginsberg JP, state that patients with SDNN values below 50 ms are classified as unhealthy, those with values between 50-100 ms have compromised health, and those above 100 ms are considered healthy [19]. In present study, SDNN showed a negative correlation with TSH values among the cases, but this correlation was not statistically significant. Galetta F et al., also observed that in SCH patients, SDNN was negatively related to TSH [17].
In the present study, we found that newly diagnosed SCH patients exhibited increased sympathetic function compared to healthy individuals. Additionally, SCH patients showed a hypofunctional parasympathetic system based on the analysis of HRV. de Miranda EJF et al., mentioned that high TSH levels, and even SCH itself, are associated with an increased risk of sudden cardiac death, particularly when compared to normal TSH levels. Reduced HRV may indicate an early phase of myocardial impairment in patients with SCH. Thus, HRV analysis may serve as a valuable tool for monitoring cardiovascular risk in these patients [14].
The SCH can impact the cardiovascular system by increasing the risk of cardiovascular mortality, coronary artery disease, and arrhythmias. These complications may arise from various factors, including reduced systolic function, diastolic hypertension, atherosclerosis, and sympathovagal imbalance [24]. Patients with decreased vagal tone are at a heightened risk for cardiovascular diseases such as arrhythmias, hypertension, and Myocardial Infarction (MI) [23]. Therefore, HRV analysis can assist in the early diagnosis of cardiovascular disease in SCH patients, allowing for timely intervention and potentially preventing the progression of cardiovascular diseases related to SCH.
The study of cardiac autonomic neuropathy in SCH patients, evaluated through short-term HRV, is limited in Eastern India. The present study is interdisciplinary and contributes to the existing data pool in this area, while also opening avenues for further exploration in the future. Consequently, the evaluation of short-term HRV among SCH patients can be utilised as a routine screening test to diagnose any autonomic dysfunction related to the cardiovascular system and aid in the differential diagnosis of cardiovascular morbidity, thereby facilitating better medical care.
Limitation(s)
The study population in the present study was limited; future research with a larger sample size could provide more informative results. There was also a failure to follow-up with patients after starting treatment with levothyroxine to assess any improvements or changes in the HRV parameters. In present study, there were more female patients compared to males among the newly diagnosed cases of SCH. Future studies should include a larger proportion of males to avoid this bias. Additionally, since, the present was a cross-sectional study, it was not possible to establish a causal relationship.
Conclusion(s)
The present study concludes that there was a significant increase in the LF/HF ratio in newly diagnosed SCH patients, along with reduced SDNN values compared to healthy controls. This indicates that sympathetic hyperactivity is more evident than parasympathetic dysfunction in these patients. Therefore, evaluating short-term HRV among SCH patients can be utilised as a routine screening test to diagnose autonomic dysfunction related to the cardiovascular system and assist in the differential diagnosis of cardiovascular morbidity for improved medical care.
Student t-test was used. Value presented as Mean±SD
Unpaired Student’s t-test was used
*p-value of <0.05 is significant