Retention, described as “secondary orthodontic treatment,” is a key step in ensuring the long-term stability of the results obtained [1]. To avoid relapse, almost every person who has undergone orthodontic treatment requires some form of retention, such as fixed or removable retainers. Removable Hawley retainers have the inherent disadvantage of the unaesthetic display of wire on the labial surface of teeth compared to Vacuum-formed Retainers (VFRs), which are superior in terms of durability, aesthetics, comfort, reduced fabrication time, cost and the ability to produce minor tooth movements, resulting in their popularity in recent times [2,3].
The VFRs, introduced in the 1970s by Ponitz RJ, are composed of polypropylene/polyethylene [4]. Bisphenol A (BPA) is added to the polymers to make them clear and tough. BPA release has been detected from a wide variety of orthodontic products, including orthodontic adhesives, elastomerics, thermoformed biocryl acrylic resin materials and clear aligners [1,5].
Within the oral cavity, these materials are exposed to temperature variations, alterations in pH, mechanical wear and enzymatic degradation from bacterial and salivary enzymes, which can lead to BPA leaching [6,7]. Leaching is one of the most important reasons for toxicity. BPA is a ubiquitous toxic chemical that mimics the endogenous endocrine hormone oestrogen, interfering with the endocrine system, with a chemical structure containing diphenyl rings with two hydroxy groups in para positions [1]. Concern over BPA released from dental materials is receiving wide attention as a result of reported BPA effects at the tissue, organ and systemic levels. BPA has been shown to have cytotoxic effects even at low doses [8,9]. It is known to cause early onset of puberty in females, feminisation in males and is also known to have carcinogenic effects [10-13].
With several aligner companies flooding the market and the rising popularity of VFRs, more clarity regarding their composition and biological effects based on in-vivo studies is required. Emerging research indicates that even low doses of BPA can affect and alter cellular signalling and cell functions [14,15]. Until clarity emerges, there is a need to use these materials with caution.
In light of this information, the present study was designed to assess BPA release in the saliva of patients wearing vacuum-formed orthodontic retainers across different time intervals and to evaluate the effects of immersing the VFRs in distilled water for 24 hours at 37°C before delivery on BPA release using HPLC.
Materials and Methods
This was a single-centre, prospective randomised clinical trial conducted over a period of six months in 2021 at Bharati Vidyapeeth Deemed to be University, Dental College and Hospital, Sangli, Maharashtra, India. A total of 28 patients aged between 15 and 25 years who had completed their fixed orthodontic treatment during that year were selected for this in-vivo study. Informed consent was obtained from all participants. The present received clearance from the Institutional Ethical Committee (Ref No: BV(DU)MC&H/Sangli/IEC/D-30/19).
Inclusion and Exclusion criteria: A total of 28 patients aged between 15 and 25 years who had completed their fixed orthodontic treatment during that year were selected for this in-vivo study.
Patients with composite restorations, poor oral hygiene, those requiring fixed bonded retainers and smokers were excluded from the study. This exclusion was necessary because components of commercial resin-based composites and sealants used for restorations or bonding fixed orthodontic retainers contain BPA and BPA methacrylate, which could interfere with the results of the study [10-12].
Sample size calculation: The sample size of 14 was calculated using GPower Software based on data obtained from a previous study conducted by Raghavan AS et al., [1]. An alpha error of 5% and a power of 80% were set, with p<0.05 considered statistically significant.
Study Procedure
For all the patients included in the study, Vacuum-form Retainers (VFRs) were fabricated and delivered using Duran+ Polyethylene Terephthalate Glycol (PET-G) thermoplastic sheets of 1 mm thickness in a Biostar pressure molding machine for both upper and lower arches. A double-blind design was implemented and a randomisation technique was used to assign patients into Group I and Group II using a head and tail coin toss method. Group I (the control group) consisted of 14 patients who received vacuum-formed orthodontic retainers that were not immersed in water before delivery, while Group II (the experimental group) consisted of 14 patients who received vacuum-formed orthodontic retainers that were immersed in distilled water at 37°C for one day prior to delivery.
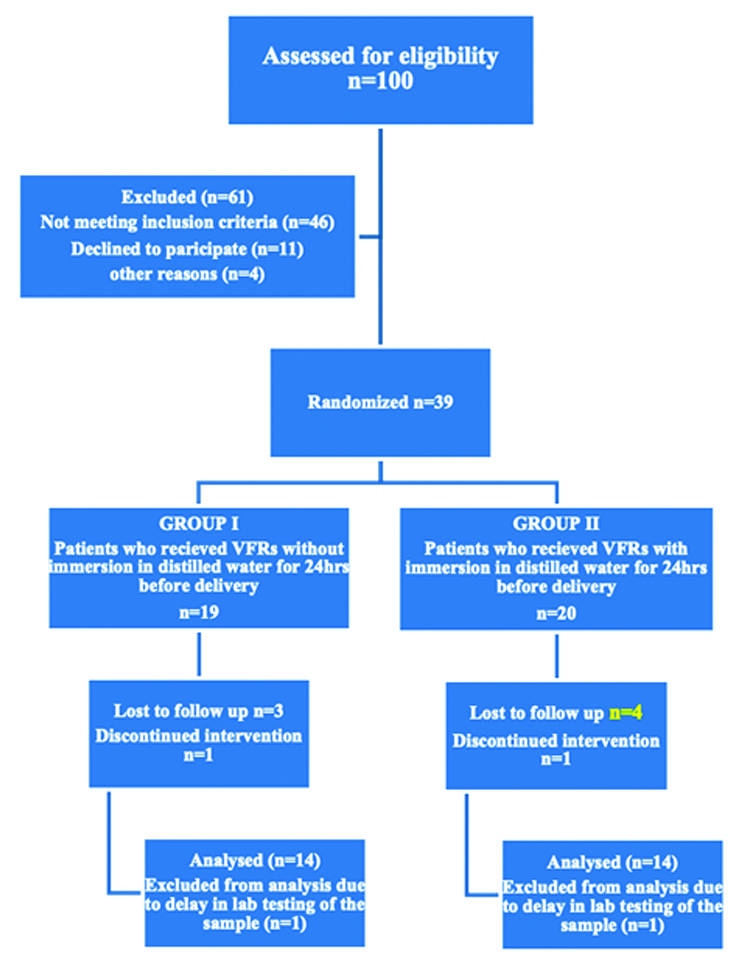
Four samples of 5 mL of unstimulated whole saliva were collected in 10 mL glass vials from each patient using the spit method at four different time intervals: T0-before placement of the retainers, T1-one hour after wearing the retainers, T2-one week after wearing the retainers and T3-three weeks after wearing the retainers. Progress through the phases of the randomised trial for both groups, including enrollment, intervention allocation, follow-up and data analysis, is represented in the form of a flow chart in [Table/Fig-1]. Patients were instructed not to eat or drink anything two hours before saliva sample collection. To minimise diurnal variation in saliva composition, all samples were collected at 11:00 AM. Patients were instructed to wear the retainers for 24 hours a day, except while eating and brushing their teeth. Saliva samples were stored at -80°C and processed using HPLC to evaluate BPA release (in ppm) according to the method described by Eliades T et al., [19]. Briefly, the samples were processed by mixing and vortexing 5 mL of saliva with an equal volume of ethanol. The samples were then centrifuged at 3000 rpm for 15 minutes to settle the particulate matter. From the centrifuged sample, 50 μL was collected and injected into the instrument for analysis via HPLC. These assays were performed in triplicate for each time period and the results were averaged.
Flow diagram of the progress through the phases of randomised trial of the two groups (that is, enrollment, intervention allocation, follow-up and data analysis).
Statistical Analysis
Statistical analysis was conducted using Statistical Package For The Social Sciences (SPSS) 23.0 software program (Chicago, IL, USA). For numerical continuous data following a normal distribution, intergroup comparisons between two groups were performed using the t-test. For numerical continuous data following a normal distribution, intergroup comparisons among more than two groups were conducted using one-way ANOVA followed by Tukey’s post-hoc test. Intragroup comparisons for numerical continuous data following a normal distribution were carried out using the paired t-test. An alpha error of 5% and a power of 80% were maintained, with p<0.05 considered statistically significant.
Results
The mean age of patients selected for the study was 20±5 years. Out of 28 patients, Group I consisted of eight female and six male patients, while Group II comprised nine female and five male patients. The BPA levels recorded in the salivary samples of patients from Group I and Group II using HPLC at different time intervals are shown in [Table/Fig-2,3].
BPA levels (in ppm) measured in saliva of Group I patients at T0, T1, T2 and T3 using HPLC.
| Time period | T0 | T1 | T2 | T3 |
|---|
| 1 | 0.00001 | 1.9645 | 1.120 | 0.1264 |
| 2 | 0.000001 | 1.5642 | 1.165 | 0.1045 |
| 3 | 0.00001 | 2.3434 | 1.586 | 0.2168 |
| 4 | 0.00001 | 2.1945 | 2.005 | 0.24645 |
| 5 | 0.000008 | 2.1245 | 1.9629 | 0.2348 |
| 6 | 0.000001 | 1.4546 | 1.0999 | 0.046 |
| 7 | 0.00001 | 2.4846 | 1.6521 | 0.22464 |
| 8 | 0.00002 | 1.4648 | 1.0625 | 0.0964 |
| 9 | 0.0001 | 2.299 | 2.246 | 0.1648 |
| 10 | 0.00001 | 1.9434 | 1.8465 | 0.2094 |
| 11 | 0.00005 | 2.1464 | 1.9994 | 0.21648 |
| 12 | 0.00001 | 2.4864 | 1.8513 | 0.01318 |
| 13 | 0.000002 | 2.54318 | 2.3428 | 0.2079 |
| 14 | 0.00002 | 1.7615 | 1.64348 | 0.0245 |
BPA levels (in ppm) measured in saliva of Group II patients at T0, T1, T2 and T3 using HPLC.
| Time period | T0 | T1 | T2 | T3 |
|---|
| 1 | 0.00001 | 0.3218 | 0.72168 | 0.015 |
| 2 | 0.00001 | 0.2359 | 0.71546 | 0.0246 |
| 3 | 0.000002 | 0.2434 | 0.94548 | 0.10054 |
| 4 | 0.00001 | 0.34248 | 0.9134 | 0.09424 |
| 5 | 0.00001 | 0.24648 | 0.84344 | 0.0426 |
| 6 | 0.000003 | 0.32464 | 0.699434 | 0.0215 |
| 7 | 0.000001 | 0.31231 | 0.75465 | 0.0161 |
| 8 | 0.00001 | 0.24649 | 0.893884 | 0.1243 |
| 9 | 0.000004 | 0.23648 | 0.92128 | 0.016434 |
| 10 | 0.00001 | 0.21346 | 0.67546 | 0.009434 |
| 11 | 0.000001 | 0.30615 | 0.754349 | 0.08734 |
| 12 | 0.00001 | 0.43613 | 0.965249 | 0.01946 |
| 13 | 0.00001 | 0.32496 | 0.75434 | 0.04348 |
| 14 | 0.00001 | 0.21046 | 0.546496 | 0.06424 |
The baseline comparison of BPA release values across different time intervals in each group is presented in [Table/Fig-4]. In Group I, there was an increase of (+0.33 ppm) in BPA levels from T0 (0.000019 ppm) to T1 (0.33 ppm), which then decreased (-0.15 ppm) from T1 to T2 (0.177 ppm) and further decreased (-0.167 ppm) from T2 to T3 (0.008 ppm). A similar pattern of BPA release was observed in Group II, where BPA release increased (+0.071 ppm) from T0 (0.000010 ppm) to T1 (0.07 ppm), but then decreased (-0.061 ppm) from T1 to T2 (0.009 ppm) and further decreased (-0.008 ppm) from T2 to T3 (0.001 ppm) [Table/Fig-5].
Comparison of mean BPA release (in ppm) in saliva of patients of Group I and Group II at T1, T2 and T3 time intervals (p-value <0.05).
| Time period | Mean value of BPA release in Group I | Mean value of BPA release in Group II | F value | p-value |
|---|
| T1 | 0.33±0.03 | 0.071±0.02 | 24.040789 | 0.00036* |
| T2 | 0.18±0.02 | 0.009±0.01 | 23.364361 | 0.00043* |
| T3 | 0.008±0.01 | 0.001±0 | 3.760457 | 0.00006* |
Changes in BPA levels in saliva within the groups at T0, T1, T2 and T3.
| Groups | T0- T1 | T1-T2 | T2-T3 |
|---|
| Group I | +0.33 ppm | -0.15 ppm | -0.167 ppm |
| Group II | +0.071 ppm | -0.061 ppm | -0.008 ppm |
The statistical analysis, as shown in [Table/Fig-4], revealed that compared to Group I, the mean values of BPA release in the saliva of patients in Group II were significantly lower at all time intervals of T1, T2 and T3 (p<0.05). To assess intragroup comparisons, Tukey’s post-hoc test was performed. Pair-wise comparisons revealed statistically significant differences in the release of BPA from VFRs across all time intervals in both groups [Table/Fig-6].
Pair-wise comparisons for Group I and Group II BPA levels at time intervals T0, T1, T2 and T3.
| Comparison | Group Ip-value | Group IIp-value |
|---|
| T1−T0 | 0.00036* | 0.00036* |
| T2−T0 | 0.00043* | 0.00043* |
| T3−T0 | 0.00046* | 0.00006* |
| T2−T1 | 0.00043* | 0.00043* |
| T3−T1 | 0.00006* | 0.00006* |
| T3−T2 | 0.00006* | 0.00006* |
(*) statistically significant difference
Discussion
In the current era, clear aligners and transparent vacuum-formed retainers have gained huge popularity amongst orthodontic patients as they are aesthetic appeal and convenience. The growing demand for thermoplastic clear aligners in both active orthodontic treatment and the retention phase necessitates a comprehensive evaluation of this popular and innovative treatment approach.
Vacuum-formed Retainers (VFRs) and aligners must be worn for up to 22 hours per day, except during eating and oral hygiene procedures. They remain in close proximity to the oral environment for extended periods and are continually exposed to fluctuations in pH, temperature, humidity and the enzymatic action of saliva and oral bacteria. These factors can lead to mechanical and chemical transformations, resulting in the leaching of Bisphenol A (BPA) into the oral cavity [20]. Additionally, trauma from oral function and non functional habits, such as bruxism, can cause abrasion at the cusp tips, leading to increased particle release [6].
Based on new evidence and approaches, the safe amount of BPA, also known as the tolerated daily intake, has been established by the European Food Safety Authority as 4 mg/kg body weight (bw)/day [21]. The total external exposure dose of all bisphenols from environmental sources has been reported to be 68.1 mg/kg bw/day. Children are considered more sensitive to the toxicological effects of bisphenols compared to adults due to developmental differences [22].
There is evidence that BPA exhibits non monotonic dose responses, meaning it can show intense reactivity at low levels while exhibiting no response at high levels [23]. This indicates that even conspicuous doses (in the nanomolar range) of BPA are capable of inducing adverse effects. Moreover, the activity level lies within a range that is below the detection limit of most analytical methods.
To date, most of the information available in the literature regarding BPA leaching from aligners and retainers is inconsistent and largely based on in-vitro studies. With the existing evidence, comparisons among studies are challenging due to variations in sample preparation protocols and levels that fall within the detection limits of most analytical methods.
Findings from most of the experimental in-vitro studies support the idea that there is no significant BPA release from thermoplastic materials [3,15,24,25]. However, since these studies have been conducted under simulated conditions that can never perfectly represent the unique oral environment, such findings should be interpreted with caution. Experimental conditions cannot accurately replicate the possible release of bisphenols due to mechanical abrasion from unpredictable masticatory forces, chemical erosion from fluctuating pH and temperature changes and the enzymatic activity of bacteria and saliva in the oral cavity.
A notable advantage of the present study was that the concentrations of BPA were recorded in saliva samples collected from patients. Blood and urine analyses were not considered because previous literature showed that BPA release from composite resins exhibited no correlation between salivary and urinary levels and BPA was undetectable in blood [26]. In a study conducted by Kotyk MW and Wiltshire WA, the BPA release from thermoformed and pre-thermoformed Biocryl Acrylic Essix, Biocryl Acrylic Retainer, Essix A+ and both used and unused Invisalign materials was evaluated over two weeks in artificial saliva using Gas Chromatography/Mass Spectroscopy (GC-MS) [6]. No traceable BPA was found in all other materials except for the Biocryl Acrylic Retainer. Afraz Walele SP et al., tested thermoplastic sheets from five manufacturers [27]. Each sample was powdered and elution was performed with a solution of 75% ethanol and 25% water in a shaker incubator at 35.5°C for seven days, followed by High-Performance Liquid Chromatography (HPLC). All tested products leached BPA, but the amount varied from one product to another. Kopperud HM et al., investigated leachable monomers, additives and degradation products from polymer-based heat-cured, self-cured orthodontic base-plate material and thermoplastic materials (Biocryl C, Essix A+ and Essix Embrace) [28]. Elution was performed in water at 37°C for 10 days. Minimal leaching was found from the thermoplastic materials, while leaching of methacrylates and formaldehyde was observed from other tested materials. Schuster S et al., tested aged and retrieved Invisalign appliances in-vitro for two weeks and found no traceable amounts of BPA leaching in ethanol solution [29]. Eliades T et al., evaluated BPA release from Invisalign by investigating the proliferation of human breast adenocarcinoma cells, based on the notion that BPA mimics estrogenic activity and that BPA release can result in the proliferation of oestrogen-sensitive cells; however, it could not be detected [30]. Alhendi A et al., conducted an in-vitro study to investigate leaching from Invisalign, Eon, Suresmile and Clarity aligner systems in different concentrations of ethanol and water solutions for 2 weeks and found no detectable amount of BPA [31]. Another study by El Idrissi I et al., assessed BPA release from new and used clear aligners for up to eight weeks using an LC-MS device and revealed no traces of BPA leaching [32].
Katras S et al., conducted a comparative evaluation of the amount of BPA released from Invisalign, Smile Direct Club and Essix Ace aligners in artificial gastric fluid, ethanol and artificial saliva over a period of 20 days. The authors concluded that a higher BPA leaching was observed with Invisalign, although the differences were not statistically significant based on the media and time intervals. However, the maximum amount of BPA release occurred during the first 24 hours after incubation and decreased over time [17]. The findings of the present study are consistent with this study.
Another interesting clinical study supporting the present findings was conducted by Raghavan AS et al., They compared BPA levels in the saliva of patients wearing VFRs and Hawley retainers at different time intervals and reported high BPA release occurring within one hour of using Essix Ace retainers, with values sharply decreasing in saliva after one week [1]. The present study further revealed a significant reduction in BPA release at all tested time intervals in patients who received VFRs that had been immersed in water for 24 hours prior to delivery.
The possible explanation for the reduced BPA release in the experimental group could be that the maximum BPA release occurred in distilled water within the first 24 hours before the retainers were delivered to the patients. To date, no other study has evaluated the effect of immersing VFRs in distilled water before delivery on BPA release.
Limitation(s)
The present study had a few limitations, such as the inability to monitor the number of hours of retainer wear by the patients, which was considered homogenous. Any disparity in the duration of wear, if present, could have influenced the obtained values. Furthermore, the BPA release was evaluated at only a few extended time intervals. Future studies should consider assessing BPA release on an hourly basis, as this may help determine the time of peak release, which could have the most significant impact on the body. Additionally, researchers should evaluate the effects of BPA in a dose-dependent manner. Collaborative research with manufacturers of thermoplastic sheets may also help determine the effects of various manufacturing parameters on the final BPA release from VFRs.
Conclusion(s)
Based on the results obtained, it can be concluded that BPA release occurs from VFRs, with maximum release occurring within the first hour of retainer wear. Immersing the VFRs in distilled water at 37°C for 24 hours before delivery can significantly reduce the amount of BPA leaching into the oral cavity of patients. Therefore, this can be adopted as a simple and effective measure to minimise BPA leaching in saliva, thereby reducing its adverse effects. With the growing abundance of knowledge and awareness, the door to further research has opened, allowing for the establishment of correct protocols that provide efficient healthcare services.
(*) statistically significant difference