Introduction
Navigating through the realm of treating long-standing thyroid swellings demands a meticulous understanding of the nature of the condition. From a clinical perspective, these lesions of long duration may cause significant troubles for the treating surgeons and anaesthesiologist during surgery, and for the pathologist in reaching a correct histological diagnosis. Thyroid nodules may remain non functional or may gain autonomy. The latter is considered as the critical factor in the pathogenesis of toxic nodular goitre. It has been found that the majority of benign toxic adenomas and most cases of toxic nodular goitre harbour mutations in the TSH receptor gene [1,2].
Patients with huge thyromegaly seek clinician’s attention because of cosmetic disfigurement or compromised airway or digestive tract. Chronic thyroid swellings can also cause compressive neuropathies of laryngeal nerves, presenting as voice changes, dyspnoea, or stridor [3]. Long-standing thyroid nodules have been shown to create a favourable environment for the development of cancer. The rate of malignancy in long-standing goitre is around 5-17% [4-6]. Here, the authors would like to report three cases of rare malignancies that we encountered while managing chronic thyroid swellings.
Case Series
Case 1
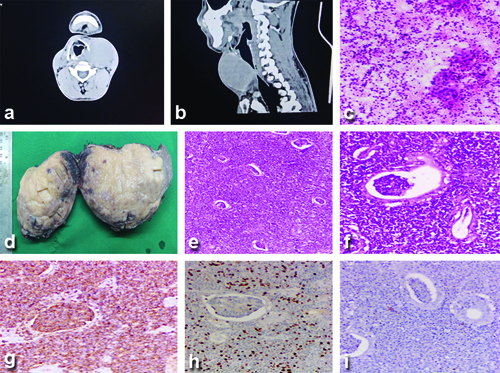
A 42-year-old male with a history of thyroid swelling of 12 years’ duration, hyperthyroidism on oral carbimazole 10 mg thrice daily, and propranolol 20 mg at night, presented with an increase in the size of the swelling over the last two years. Clinically, the patient was euthyroid without any eye signs of thyrotoxicosis. On examination of the swelling, a diffuse anterior neck swelling of size 12×10 cm was noted with varying consistency. Ultrasonography (USG) of the neck reported an enlarged hypervascular right lobe and isthmus of the thyroid gland suggestive of toxic goitre and a large hypoechoic area involving the left lobe of the thyroid gland with moderate vascularity. Contrast-enhanced Computed Tomography (CECT) of the neck showed a well-defined heterogeneous lesion with a smooth margin involving the right lobe, isthmus, and left lobe, measuring 6.7×10.2×8 cm in anteroposterior, transverse, and craniocaudal dimensions. No evidence of calcifications was seen in the nodule in CECT. The lesion was causing displacement of the trachea and neck vessels to the right [Table/Fig-1a,b]. A CECT-guided Fine Needle Aspiration Cytology (FNAC) reported features suspicious for malignancy with co-existing autoimmune thyroiditis and dense lymphoid infiltrate - Bethesda V [7]. The patient, in a euthyroid status, was planned for total thyroidectomy. Intraoperatively, the airway was secured by awake fibre optic bronchoscopy-assisted endotracheal intubation. The left lobe lesion was found to be extending upto the ipsilateral prevertebral area, with the recurrent laryngeal nerve densely adherent to the capsule of the gland. Enlarged paratracheal lymph nodes were noted on both sides and were dissected out. The postoperative period was uneventful, and the patient was discharged on postoperative day 5. The histopathology of the specimen showed features suggestive of Low-grade B-cell Non-Hodgkin’s Lymphoma (NHL) with paratracheal nodes negative for malignant changes. The adjacent thyroid parenchyma showed features of lymphocytic thyroiditis and Hurthle cell changes [Table/Fig-1c-f]. Immunohistochemistry showed neoplastic cells positive for Cluster of Differentiation 20 (CD20) with Ki-67 25 to 30% and negative for CD5, CD10, Bcl2, CD138, and CD23, confirming the diagnosis of low-grade B-cell NHL consistent with MALT Lymphoma [Table/Fig-1g-i]. The patient was referred to the Radiation Oncology Department for further management and is on regular follow-up.
a) Axial CT section of the neck showing left lobe thyroid lesion causing right-side airway shift; b) Sagittal CT section of the neck showing thyroid lesion compressing the airway; c) Cytology smears showing small to medium-sized lymphoid cells (H&E, 40X); d) Gross section of thyroid showing diffuse greyish and fleshy areas; e) Diffuse sheets of lymphoid cells (H&E, 40X); f) Histopathology section showing MALT ball (H&E, 40X); g) Lymphoid cells showing positive staining with anti-CD-20 antibody (100X); h) Ki 67 showing a medium proliferative index of 25-30% (100X); i) Lymphoid cells showing negative staining with anti-CD-10 antibody (100X).
Case 2
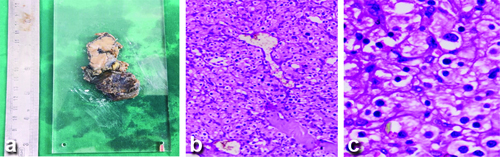
A 43-year-old male with no history of any co-morbidities came to the OPD with complaints of an anterior neck swelling of eight years’ duration. On examination, a firm nodule of size around 3×3 cm was noted in the left lobe of the thyroid gland. USG of the neck showed a well-defined heterogeneous nodule at the isthmus reaching upto the left lobe of the thyroid with prominent vascularity on colour Doppler. CECT showed an intensely homogeneous enhancing mass with an irregular outline involving the left lobe and isthmus. The lesion is noted to extend to the left anterolateral aspect of the trachea and anteriorly infiltrating the strap muscles, with radiological features suggestive of malignancy. The patient was referred to the Surgical Oncology Department. Imaging-guided FNAC smears showed cellular smears suggestive of a follicular neoplasm. The patient underwent total thyroidectomy with central compartment node dissection and partial tracheal resection (first and second ring). Intraoperatively, the airway was secured initially with endotracheal intubation, and after post-tracheal resection, the patient was tracheostomised. Histopathological examination {Haematoxylin and Eosin (H&E)} showed a tumour composed of sheets and nests of polygonal cells with eosinophilic cytoplasm and deep-staining nuclei, with foci showing a Zellballen appearance. A differential diagnosis of parathyroid carcinoma/paraganglioma was suggested with invasion of the tracheal rings and strap muscles. All the level VI nodes were negative for metastasis. In immunohistochemistry, tumour cells were positive for synaptophysin, chromogranin, vimentin, GATA 3, and S100, and negative for Cytokeratin (CK), Thyroid Transcription Factor 1 (TTF1), Calcitonin, CAM 5.2, thyroglobulin, and PAX 8. The Ki proliferation index was 4-5%. A diagnosis of paraganglioma of the thyroid gland was made [Table/Fig-2a-c]. Postoperative whole-body Fluorodeoxyglucose-18 (FDG) Positron Emission Tomography (PET) showed no evidence of metabolically active FDG-avid disease anywhere else, suggestive of no distant metastasis. The patient is on regular follow-up with 75 mcg of oral thyroxine/day supplementation.
a) Gross cut surface specimen of the thyroid paraganglioma; b) Histopathology section showing round, polygonal cells arranged in nests (Zellballen pattern, H&E, 100X); c) Cells showing round to ovoid nuclei with eosinophilic cytoplasm (H&E, 400X).
Case 3
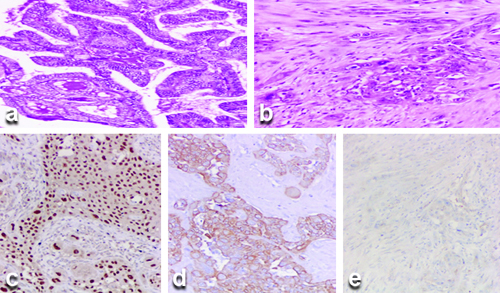
A 72-year-old female patient, who underwent a left hemithyroidectomy 10 years ago at a different Institute, presented to the Ear Nose and Throat (ENT) Department with complaints of difficulty in breathing and change in voice for the past four weeks. Upon detailed questioning, the patient also reported difficulty in swallowing for the past two weeks, particularly with solids, with no symptoms suggestive of aspiration. The details of the left hemithyroidectomy performed previously were not available. Clinical examination revealed a well-defined nodule with varying cystic and firm consistency in the right lobe of the thyroid, along with an enlarged lymph node in the right level II cervical region. An ultrasound of the neck showed post left hemithyroidectomy status with two cystic nodules in the right lobe, including microcalcifications, and enlarged lymph nodes in the right jugular region. Magnetic Resonance Imaging (MRI) of the neck with T2-weighted imaging revealed a heterogeneously enhancing solid-cystic lesion in the right lobe of the thyroid measuring 9×6.5×6 cm, extending into the anterosuperior mediastinum. Fine Needle Aspiration Cytology (FNAC) guided by imaging reported Bethesda VI with features consistent with papillary thyroid carcinoma. High-resolution Computed Tomography (HRCT) of the thorax was also performed and showed no evidence of metastasis in the lung fields. The patient was scheduled for a complete thyroidectomy with right modified radical neck dissection, including central compartment dissection. Intraoperatively, multiple enlarged lymph nodes were observed at levels II, III, IV, and VI, with the largest measuring 4×5 cm at level III. The tumour was found invading the right sternocleidomastoid, the sympathetic ganglion and vagus nerve. A modified radical neck dissection type II with resection of the vagus nerve was performed. The patient underwent tracheostomy. The left lobe was not visualised intraoperatively. The histopathological examination report revealed classic variant papillary carcinoma in the right lobe, with 7 out of 35 lymph nodes showing metastatic carcinoma involving level VI lymph nodes on both the right and left-sides, staging the patient as PT3aN1a. The right neck dissection specimen showed a mass measuring 4×3×1 cm that histologically revealed a poorly differentiated malignant neoplasm. The differential diagnosis included dedifferentiated papillary carcinoma with squamoid features, metastatic squamous cell carcinoma, or poorly differentiated sarcoma [Table/Fig-3a,b]. Tissue was collected for immunohistochemistry, which showed that tumour cells of papillary carcinoma in the thyroid were positive for TTF1 and Thyroglobulin, while tumour cells of the poorly differentiated malignant mass showed CK5/6, p63, and weak p40 positivity [Table/Fig-3c-e]. The possible differential diagnosis based on these findings were: 1) Anaplastic thyroid carcinoma with a squamous cell carcinoma pattern; 2) Primary squamous cell carcinoma or metastasis from another site. Since, no other primary tumour was detected in the whole-body PET scan, a final diagnosis of anaplastic carcinoma with a squamous cell carcinoma pattern was made.
a) Histopathology section showing classical papillary thyroid carcinoma areas (H&E stain, 100x magnification); b) Histopathology section showing squamoid cell areas (H&E stain, 100x magnification); c) Tumour cells showing positive staining with p63 antibody (100x magnification); d) Tumour cells showing positive staining with CK-5 antibody (100x magnification); e) Squamoid areas showing negative staining with TTF-1 antibody (100x magnification).
Details of all three cases are summarised in [Table/Fig-4]. Patient consent was obtained for the inclusion of relevant images in the publication, and the study was conducted in accordance with the ethical standards outlined in the Declaration of Helsinki (2013 amendment).
| Case no. | Duration of swellings | Past history | Histopathological diagnosis | Follow-up details |
|---|
| 142 Y/M | 12 years | Hyperthyroidism on carbimazole 10 mg thrice daily and propranolol 20 mg at night | Low Grade B cell Non-Hodgkin’s Lymphoma (NHL)-consistent with MALT Lymphoma | On regular follow-up in Radiation Oncology Department |
| 243 Y/M | 8 years | Nil | Paraganglioma | On regular follow-up with thyroxine supplementation 75 mcg daily dose |
| 372 Y/F | 4 weeks | History of left hemithyroidectomy 10 years back | Anaplastic carcinoma- Squamous cell carcinoma pattern | Lost to follow-up |
Discussion
Addressing the management of long-standing thyroid neoplasms presents a clinical conundrum, requiring a comprehensive understanding of the complex challenges one could face in the Preoperative diagnosis, treatment planning, intraoperative and postoperative periods, and in reaching postoperative tissue diagnosis. Patients with a history of long-standing thyromegaly mainly present with cosmetic concerns, followed by symptoms arising from compression of nearby vital structures. The initial investigations in a thyroid nodule include an approach similar to the triple assessment of the breast, i.e., detailed history taking and physical examination, imaging, and FNAC. In cases where the clinical euthyroid status is doubtful, a quadruple assessment of the thyroid nodule is advised, which includes thyroid function tests in addition to the triple assessment. This approach may not be suitable for managing tumours of long duration and large size. Clinical examination in most cases of chronic thyroid nodules only provides minimal information about the true nature of the condition. In all our cases, clinical examination did not correlate with radiological findings. Radiological imaging is considered the most important tool in preoperative planning [8].
Supplementary imaging modalities help in planning the type of surgery, anticipating anaesthesia challenges, preparing for difficulties the surgeon might face intraoperatively, and planning reconstruction choices. USG is considered the primary diagnostic modality when evaluating a thyroid nodule. USG helps to assess the size of the lesion, morphological characteristics such as echogenicity, and detect nodal spread. USG also helps pathologists get a fair idea of the Thyroid Imaging Reporting and Data System (TIRADS) status, which may correlate with Bethesda findings, and obtain a more representative tissue sample for cytological examination through USG-guided procedures [9].
However, in cases of large thyroid diseases, the sensitivity of USG as a single imaging modality is very low. Neck CT or MRI more accurately determine the extent of the lesion, the degree of compression of the trachea or oesophagus, and the involvement of lymph nodes and the Recurrent Laryngeal Nerve (RLN). To obtain better delineation and information about vascularity, IV contrast imaging is preferred. Between CT and MRI, CT is preferred as it is associated with less time, fewer motion artifacts with respiration, and is cost-effective with better resolution [10-12].
Long-standing thyroid neoplasms are associated with the displacement of the trachea and tracheal compression, leading to the development of tracheomalacia or involvement of the trachea in cases of aggressive malignancy. The preferred method for securing an airway is awake Fibre Optic Intubation (FOI) with local anaesthesia. In cases where FOI is not possible, alternative options include sevoflurane inhalation induction, ventilation through a bronchoscope, or tracheostomy [13,14]. In present cases, two surgeries were performed with FOI and one with the standard Endotracheal (ET) intubation procedure. However, in two patients, airway transfer to tracheostomy was performed. The indications were bilateral RLN involvement resulting in sacrificing the same, and in the other case, the trachea was infiltrated with a tumour resulting in tracheal resection. Prompt radiological reporting of tracheal involvement in the second case helped the anaesthesia team and surgeons anticipate and plan for this scenario.
Giant goitres pose more hurdles for surgeons intraoperatively due to the distortion of neck anatomy, increased vascular nature of the tumour, and the potential for compression and tumour infiltration of vital structures in the field. The use of heat-producing devices for hemostasis was found to increase the incidence of neuropraxia and paresis of the RLN postoperatively. In present Institution, author use both the ligature technique and vessel sealer [15-17].
Long-standing thyroid neoplasms also challenge the surgeon in locating the parathyroid gland, and it has been found that accidental removal of the parathyroid gland is common in surgeries performed for chronic nodules. The incidence of RLN injury as a result of thyroid surgeries is reported as 0.5-5%. RLN is damaged more in cases of retrosternal goitre where sternotomy is demanded, retraction of the thyroid gland for a long duration, and the use of heat-powered instruments close to the nerve for achieving hemostasis. A nerve monitoring device helps avoid inadvertent damage to the RLN [18].
Primary Thyroid Lymphoma (PTL) is rare, with an incidence of 0.5 to 5% of all thyroid malignancies and 2.5 to 7% of all extranodal lymphomas [19-21]. The most common types of PTL are MALT lymphoma, accounting for more than 50% of thyroid lymphomas, and MALT lymphoma is very uncommon [22,23]. A prerequisite for the occasional MALT lymphoma is the presence of lymphoid tissue+ in the affected organ, which correlates with present case as the patient had long-standing Hashimoto’s thyroiditis.
The most common malignancy in chronic thyroid nodules is of follicular cell origin. Primary thyroid paragangliomas are uncommon neuroendocrine tumours, supposed to originate from the inferior laryngeal parenchyma. A comprehensive search of PubMed showed that only 35 cases have been reported in the English literature [24]. As per present study, the median age is 48.2 years, and there is a marked predilection for female patients, with a male-to-female ratio of 1:5:3. However, present case involved a male patient and was published for its extensive rarity. This patient did not have detectable active catecholamine synthesis.
Approximately 10% of patients with paraganglioma are familial due to germ line mutations in Succinate Dehydrogenase B (SHDB), C (SDHC), D (SDHD), or SDH assembly factor 2 (SDHAF2); therefore, molecular genetic studies may be useful [25-28]. Due to the unexpected occurrence and overlapping features, thyroid paraganglioma is frequently misdiagnosed as other common types of thyroid neoplasms, which may result in inappropriate treatment. Therefore, increased awareness of its potential occurrence in the thyroid gland and familiarity with its characteristic features are important for both clinicians and pathologists to avoid diagnostic and therapeutic pitfalls.
Anaplastic thyroid carcinoma is a rare, almost uniformly fatal follicular cell-derived thyroid carcinoma that can have a variety of histologic patterns. Squamous cell carcinoma of the thyroid was previously listed separately in the World Health Organisation (WHO) tumour classification. However, it is now regarded as a subtype of anaplastic thyroid carcinoma and clarified by the WHO as anaplastic thyroid carcinoma-squamous cell carcinoma pattern [29]. The BRAF V600E mutation is seen, with a history of or concurrent papillary thyroid carcinoma or other differentiated thyroid carcinomas [30,31]. The present case involves a papillary carcinoma-classic variant in the right lobe with metastasis in Level VI lymph nodes on the right and left-side. RAS and BRAF V600E mutations are considered early driver mutations, while Tumour Protein 53 (TP53) and Telomerase (TERT) promoter mutations are late events in the pathogenesis of anaplastic thyroid carcinoma [32].
Upon reviewing the literature, to the best of authors knowledge, squamous cell carcinoma transformation of papillary carcinoma is rare, and almost all of them were seen adjacent to the primary neoplasm in the thyroid gland only. The present case showed papillary carcinoma-classic variant in the right lobe with a separate mass in the nodal dissection specimen that was attached to the vagus nerve, showing an anaplastic carcinoma-squamous cell carcinoma variant. This type of presentation has not been reported before and hence, was published for its rarity.
These imposing thyroid enlargements often cause tracheomalacia, which can result in immediate postoperative airway collapse or tracheal stenosis later on. Methods to avoid postoperative airway narrowing include keeping the endotracheal tube in place for longer periods or performing an elective tracheostomy in suspected cases of tracheomalacia [33]. In first case, extubation was performed after 36 hours because of the intraoperative finding of thinning in the areas of the first and second tracheal rings. Additionally, longstanding thyroid neoplasms make locating the parathyroids difficult, leading to accidental removal. In author’s experience, we had to autotransplant the dissected parathyroids inside the ipsilateral sternocleidomastoid.
In conclusion, longstanding thyroid swellings present challenges for clinicians and pathologists.
Conclusion(s)
Long-standing thyroid nodules do have the potential to become malignant neoplasms. The most common malignancy in such cases is papillary thyroid carcinoma. Other rare malignant neoplasms are also possible, such as poorly differentiated and dedifferentiated tumours, various types of extranodal lymphomas, and other tumours of epithelial and soft tissue origin. To reach a conclusive diagnosis in doubtful and less common entities, histopathological and cytopathological examinations have high diagnostic value. It is important to evaluate patients for the presence of any distant metastasis and to schedule frequent follow-up visits to detect early disease recurrence.
[1]. Knobel M, Etiopathology, clinical features and treatment of diffuse and multinodular nontoxic goitresJ Endocrinol Invest 2016 39(4):357-73. [Google Scholar]
[2]. Fuhrer D, Constitutive TSH receptor activation as a hallmark of thyroid autonomy. Endocrine 2020 68(2):274-78. [Google Scholar]
[3]. Du W, Liu ST, Li P, Sun LY, Zhao M, Qi JX, Intra-and postoperative complications in 137 cases of giant gland tumourOnco Lett 2012 4(5):965-69. [Google Scholar]
[4]. Fernandes GA, Matos LL, Dedivitis RA, Risk factors for malignancy in patients with multinodular goitreInt Arch Otorhinolaryngol 2022 27(1):e138-42. [Google Scholar]
[5]. Ajarma KY, Al-Faouri AF, Al Ruhaibeh MK, Almbaidien FA, Nserat RT, Al-Shawabkeh AO, The risk of thyroid carcinoma in multinodular goitre compared to solitary thyroid nodules: A retrospective analysis of 600 patientsMed J Armed Forces India 2020 76(1):23-29. [Google Scholar]
[6]. Simsir IY, Cetinkalp S, Kabalak T, Review of factors contributing to nodular goitre and thyroid carcinomaMed Princ Pract 2020 29(1):01-05. [Google Scholar]
[7]. Ali SZ, Baloch ZW, Cochand-Priollet B, Schmitt FC, Vielh P, VanderLaan PA, The 2023 Bethesda System for reporting thyroid cytopathologyThyroid 2023 33(9):1039-44. [Google Scholar]
[8]. Elakkiya R, Sivamarieswaran R, Gopinathan A, Balamurugan R, Triple assessment in diagnosis of thyroid nodules and its comparison with histopathologyCureus 2023 15(3):e36021 [Google Scholar]
[9]. Hambly NM, Gonen M, Gerst SR, Li D, Jia X, Mironov S, Implementation of evidence-based guidelines for thyroid nodule biopsy: A model for establishment of practice standardsAJR Am J Roentgenol 2011 196(3):655-60. [Google Scholar]
[10]. Yi KH, Lee EK, Kang HC, Koh Y, Kim SW, Kim IJ, 2016 revised Korean thyroid association management guidelines for patients with thyroid nodules and thyroid cancerInt J Thyroidol 2016 9(2):59-126. [Google Scholar]
[11]. Russ G, Bonnema SJ, Erdogan MF, Durante C, Ngu R, Leenhardt L, European thyroid association guidelines for ultrasound malignancy risk stratification of thyroid nodules in adults: The EU TIRADSEur Thyroid J 2017 6(5):225-37. [Google Scholar]
[12]. Shetty SK, Maher MM, Hahn PF, Halpern EF, Aquino SL, Significance of incidental thyroid lesions detected on CT: Correlation among CT, sonography, and pathologyAJR Am J Roentgenol 2006 187(5):1349-56. [Google Scholar]
[13]. Patil VH, Huddar KS, Barsagde WS, Rao AU, Airway management of huge thyroid swelling having tracheal deviation: Challenges facedIndian J Clin Anaesth 2022 9(4):514-17. [Google Scholar]
[14]. Kaur H, Kataria AP, Muthurmalingapandian M, Kaur H, Airway considerations in case of large multinodular goitreAnesth Essays Res 2017 11(4):1097-100. [Google Scholar]
[15]. Smith RB, Coughlin A, Thyroidectomy hemostasisOtolaryngol Clin North Am 2016 49(3):727-48. [Google Scholar]
[16]. Balghari KW, Abbas N, Khan MA, Ilyas M, Islam MU, Ijaz MT, Assessment of safety of bipolar diathermy regarding recurrent laryngeal nerve damage in thyroid surgeryPak Postgrad Med J 2020 30(02):61-64. [Google Scholar]
[17]. Jawad NK, Al-Harbawi LQ, Ullawerdi KS, Compare using of LigaSure and conventional bipolar device in total thyroidectomy, retrospective studyTeikyo Med J 2021 44(06):2955-60. [Google Scholar]
[18]. Tressalet C, Chigot JP, Menegaux F, How to prevent recurrent nerve palsy during thyroid surgery?Ann Chir 2006 131(2):149-53. [Google Scholar]
[19]. Al Hassan MS, El Ansari W, Darweesh A, Sharaf Eldeen MZ, Obiedat S, Abdelaal A, Mucosa-associated lymphoid tissue lymphoma (MALT lymphoma) in the thyroid mimicking a painless subacute (De Quervain’s) thyroiditis on presentation, fine needle aspiration and cytology, and ultrasound findings: A rare case reportInt J Surg Case Rep 2023 106:10814710.1016/j.ijscr.2023.108147 [Google Scholar] [CrossRef]
[20]. Kossev P, Livolsi V, Lymphoid lesions of the thyroid: Review in light of the revised European-American lymphoma classification and upcoming World Health Organization classificationThyroid 1999 9(12):1273-80. [Google Scholar]
[21]. Green LD, Mack L, Pasieka JL, Anaplastic thyroid cancer and primary thyroid lymphoma: A review of these rare thyroid malignanciesJ Surg Oncol 2006 94(8):725-36. [Google Scholar]
[22]. Acar N, Acar T, Avcı A, Haciyanli M, Approach to primary thyroid lymphoma: Case seriesTurk J Surg 2019 35(2):142-45. [Google Scholar]
[23]. Karvounis E, Kappas I, Angelousi A, Makris GM, Kassi E, Mucosa-associated lymphoid tissue lymphoma of the thyroid gland: A systematic review of the literatureEur Thyroid J 2020 9(1):11-18. [Google Scholar]
[24]. Yu BH, Sheng WQ, Wang J, Primary paraganglioma of thyroid gland: A clinicopathologic and immunohistochemical analysis of three cases with a review of the literatureHead Neck Pathol 2013 7(4):373-80.Epub 2013 Aug 13; PMCID: PMC382479310.1007/s12105-013-0467-723943066 [Google Scholar] [CrossRef] [PubMed]
[25]. Phitayakorn R, Faquin W, Wei N, Barbesino G, Stephen AE, Thyroid-associated paragangliomasThyroid 2011 21(7):725-33.10.1089/thy.2010.0362 [Google Scholar] [CrossRef]
[26]. Evankovich J, Dedhia RC, Bastaki JM, Tublin M, Johnson JT, Primary sclerosing paraganglioma of the thyroid gland: A case reportAnn Otol Rhinol Laryngol 2012 121(8):510-15. [Google Scholar]
[27]. Castelblanco E, Gallel P, Ros S, Gatius S, Valls J, De-Cubas AA, Thyroid paraganglioma. Report of 3 cases and description of an immunohistochemical profile useful in the differential diagnosis with medullary thyroid carcinoma, based on complementary DNA array resultsHum Pathol 2012 43(7):1103-12. [Google Scholar]
[28]. Armstrong MJ, Chiosea SI, Carty SE, Hodak SP, Yip L, Thyroid paragangliomas are locally aggressiveThyroid 2012 22(1):88-93. [Google Scholar]
[29]. Basnet A, Pandita A, Fullmer J, Sivapiragasam A, Squamous cell carcinoma of the thyroid as a result of anaplastic transformation from BRAF-positive papillary thyroid cancerCase Rep Oncol Med 2017 2017:4276435 [Google Scholar]
[30]. Xu B, Fuchs T, Dogan S, Landa I, Katabi N, Fagin JA, Dissecting anaplastic thyroid carcinoma: A comprehensive clinical, histologic, immunophenotypic, and molecular study of 360 casesThyroid 2020 30(10):1505-17. [Google Scholar]
[31]. Chen TY, Lorch JH, Wong KS, Barletta JA, Histological features of BRAF V600E-mutant anaplastic thyroid carcinomaHistopathology 2020 77(2):314-20. [Google Scholar]
[32]. Volante M, Lam AK, Papotti M, Tallini G, Molecular pathology of poorly differentiated and anaplastic thyroid cancer: What do pathologists need to know?Endocr Pathol 2021 32(1):63-76. [Google Scholar]
[33]. Agarwal A, Mishra AK, Gupta SK, Arshad F, Agarwal A, Tripathi M, High incidence of tracheomalacia in longstanding Goitres; Experience from endemic goiter regionWorld J Surg 2007 31(4):832-37. [Google Scholar]