Spinal anaesthesia, known as SAB, is a neuraxial anaesthesia technique in which a Local Anaesthetic (LA) is placed directly in the intrathecal space. It was first administered by Karl August Bier in 1898 [1]. The first LA isolated was cocaine, the only naturally occurring LA [2]. It is now a preferred technique of anaesthesia for patients scheduled for lower abdominal or lower limb surgeries because of its cost-effectiveness, muscle relaxation, reliability, safety, prolonged intraoperative and postoperative analgesia, and early ambulation [3,4].
The various LA drugs used in spinal anaesthesia are Lidocaine, Bupivacaine, Tetracaine, Mepivacaine, Ropivacaine, Levobupivacaine, and Chloroprocaine. Levobupivacaine is an amino-amide LA drug, the S (-) levorotatory isomer of bupivacaine. Levobupivacaine is a safer and more effective alternative for regional anaesthesia than bupivacaine. It has demonstrated less affinity and depressant effects on myocardial and central nervous system centers in pharmacodynamic studies, with a superior pharmacokinetic profile compared to bupivacaine. It provides a more selective neuraxial blockade due to its faster protein binding property; hence, side-effects such as hypotension, bradycardia, and cardiotoxicity are less compared to bupivacaine [5,6].
Various adjuvants like morphine, fentanyl, sufentanil, ketamine, midazolam, synthetic opioids, and corticosteroids have been used along with LAs for SAB [7]. Dexamethasone is a fluorinated synthetic steroid [8]. It acts by blocking the transmission of nociceptive C-fibers and suppressing ectopic neural discharge. As an additive to intrathecal bupivacaine, it has been shown to improve the duration of surgical and postoperative analgesia without any complications [9].
There are not any studies on the use of dexamethasone with isobaric levobupivacaine under SAB. The present study was planned with the primary objective of comparing levobupivacaine with or without dexamethasone for the effective duration of analgesia.
Materials and Methods
The present pilot randomised, double-blind controlled trail was undertaken in the Department of Anaesthesiology, UCMS-GTB Hospital, New Delhi, India from January 2021 to August 2022 after receiving approval from the Institutional Ethical Committee via letter IECHR/2020/PG/47/3-R1 dated 22-12-2020. The trial was registered with the Clinical Trials Registry-India (ctri.nic.in) before enrolling the patients (Trial registration number CTRI/2021/01/030366).
Sample size: On extensive search on literature, author could not find any study directly related to primary objective. So, they proposed to undertake this as a pilot study with 30 patients in each group. So, a total of 60 patients were recruited.
Inclusion and Exclusion criteria: Patients belonging to ASA grade I or II, between 18-65 years of age, and height 150-180 cm, scheduled for lower abdominal surgeries under SAB were included in the study. Patients who were non consenting for SAB or had an infection at the injection site, coagulopathy, space-occupying lesion, increased intracranial tension, seizure disorder, pre-existing diabetes mellitus, or any spine deformity were excluded from the study.
Study Procedure
Patients were randomly allocated to one of the two groups using a computer-generated random number table [Table/Fig-1]. Concealment was done using consecutively numbered sealed opaque envelopes by the investigator. Upon an extensive literature search, no study directly related to the primary objective was found. Therefore, authors proposed to undertook present as a pilot study with 30 patients in each group. A total of 60 patients were recruited. The patients were visited one day prior to surgery and were explained about the procedure, type, amount of drug being used along with its uses, risks, and side-effects. Written informed consent was obtained. Premedication was administered in the form of tablet Alprazolam 0.25 mg and tablet Ranitidine 150 mg the night before surgery and repeated on the morning of surgery. On the morning of surgery, after preanaesthetic check-up, patients were transferred to the operation theatre and shifted to the operating table. Non invasive oscillometric blood pressure, Lead II Electrocardiography (ECG), and pulse oximetry were attached and monitored. Baseline readings were noted. Intravenous access was established with a wide-bore cannula, and fluid preloading was initiated using Ringer Lactate 15 mL/kg over 15-30 minutes. In Group LS, 12.5 mg of 0.5% isobaric levobupivacaine (2.5 mL) with normal saline (1 mL) was administered, and in Group LD, 12.5 mg of 0.5% isobaric levobupivacaine (2.5 mL) with 4 mg dexamethasone (1 mL) was administered [9-11].
Consodilated Standard of Reporting Trails (CONSORT) 2010 flow diagram.
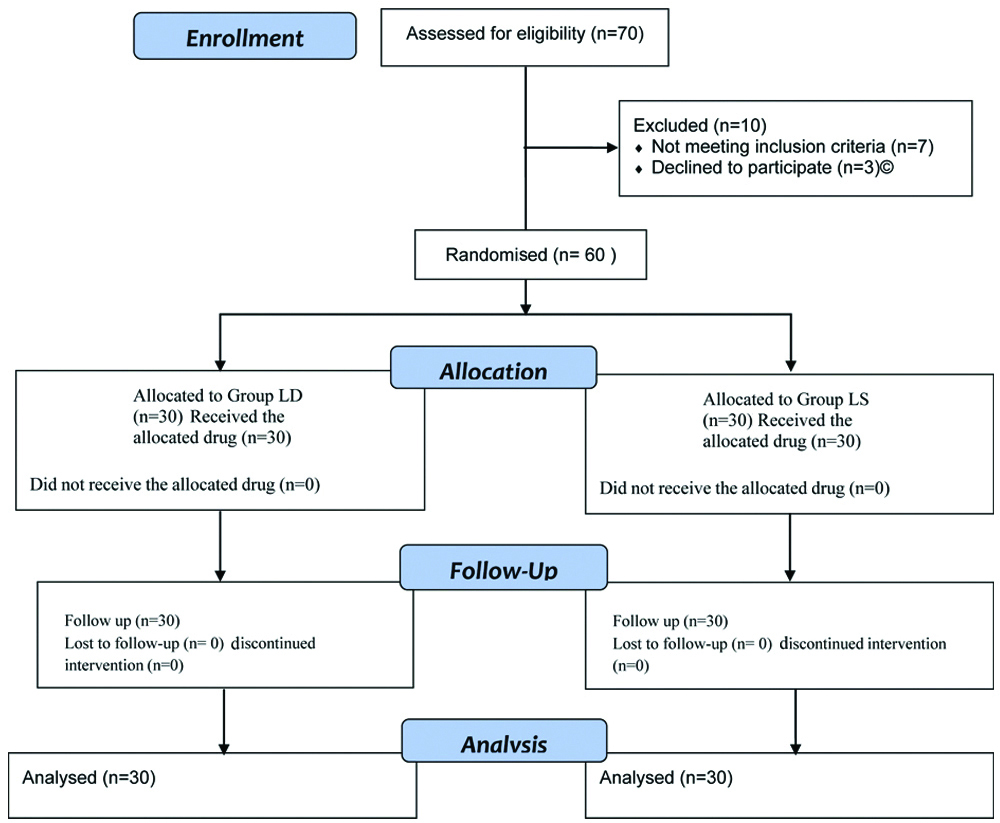
The total volume of intrathecal drug was 3.5 mL in both groups. The study drug was prepared by an anaesthetist not involved in the study. Under continuous monitoring, a SAB was performed with all aseptic precautions following the standard protocol. Patients were assessed for characteristics of sensory and motor block, haemodynamic parameters, pain score, and side-effects. Both the patient and the anaesthesiologist assessing the block characteristics and other parameters were blinded to group allocation.
The SAB was administered in the sitting position with a midline approach under all aseptic precautions. A 25 G Quincke’s spinal needle was inserted at the L2-L3/L3-L4 intervertebral space with the bevel facing cephalad, and the SAB was given with 3 mL of the study drug according to the allocated group after confirming free flow of cerebrospinal fluid.
The drug was injected at a rate of 0.5 mL/sec. After removing the spinal needle, the patient was placed supine.
The following outcomes were assessed:
Primary outcomes were the duration of effective analgesia (time from the SAB to the time of the patient’s first complaint of pain with Virtual Analogue Scale (VAS) ≥3). Secondary outcomes included the quality of sensory block, quality of motor block, and side-effects such as nausea, vomiting, hypotension, and bradycardia. Parameters monitored included heart rate, mean blood pressure, arterial oxygen saturation, and respiratory rate every five minutes for the first 30 minutes, then every 15 minutes until the end of surgery. Any episode of hypotension, indicated by a decrease in Systolic Blood Pressure (SBP) by more than 20% from the preoperative baseline value and/or SBP <90 mmHg, was managed by rapid infusion of additional intravenous fluids and injection of 6 mg mephentermine intravenously. Heart rate was also assessed, and bradycardia (heart rate less than 50/min) was treated with 0.6 mg atropine intravenously. Intraoperative nausea, vomiting, headache, dizziness, and any other side-effects were noted and treated accordingly. The level of sensory block was assessed using the pinprick method in the mid-clavicular line with a 26 G hypodermic needle every two minutes until the level stabilised for three consecutive tests, and the highest level of sensory block was noted.
Quality of sensory block was evaluated by the time of onset of sensory block at the level of T10 dermatome, maximum block height, time to achieve maximum block height, two-segment regression of sensory block (from the maximum height of the block), time of regression of sensory block to the T12 dermatome, and duration of effective analgesia: the time from the SAB to the time of the patient’s first complaint of pain with VAS ≥3, and the patient was given rescue analgesia in the form of intravenous paracetamol 1 gm.
The assessment of motor blockade in the lower limb was performed according to the Modified Bromage Scale (MBS) [12]. The MBS was noted at the time when the sensory block height was achieved at the T10 level. The quality of motor block was assessed based on the time taken to achieve maximum motor blockade, the maximum motor blockade achieved, and the MBS at the time of rescue analgesia.
According to the protocol, if the sensory block level of T10 was not achieved within 20 minutes after the intrathecal injection of the drug, the patient was planned to receive general anaesthesia. However, no such cases were encountered in the present study. Pain was assessed using a 0-10 cm Visual Analogue Scale (VAS), where 0 represents no pain and 10 represents the worst imaginable pain. The VAS score was recorded at all the time points mentioned above, along with the haemodynamic parameters.
When the patient first complained of pain with a VAS score ≥3, rescue analgesia of Inj. paracetamol i.v. 15 mg/kg was given, and the time was noted. After completion of the surgery, the patients were transferred to the postoperative ward, and monitoring of VAS score and MBS continued every 30 minutes until the patient’s first complaint of pain with a VAS score ≥3. At this point, after administering the rescue analgesia, the study was terminated. For the rest of the postoperative period, analgesia was provided according to the standard protocol. Side-effects such as nausea, vomiting, urinary retention, hypernatremia, and hyperglycaemia were noted and treated accordingly.
Statistical Analysis
Since, it’s a pilot study, there was no hypothesis testing conducted. A post-hoc power calculation has been performed on the primary outcome, which is the duration of effective analgesia, and it was found to be ≥90%. Unpaired t-tests/Mann-Whitney U tests were used to determine significance. A p-value <0.05 was considered to be significant.
Results
The demographic profile of the patients was comparable between the two groups as shown in [Table/Fig-2]. The time of onset of sensory block was shorter in the LD group than in the LS group, and the time to regression to T12 was longer in the LD group has been demonstrated in [Table/Fig-3]. The duration of effective analgesia was also longer in the LD group.
Demographic profile between two groups.
| Parameters | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Age (year)* | 37.80±9.98 | 38.63±12.37 | 0.775 |
| ASA (I:II)# | 24:6 | 23:7 | 0.754 |
| Sex# |
| Male:Female | 17:13 | 21:9 | 284 |
| Weight (kg)* | 57.10±7.85 | 60.77±6.40 | 0.052 |
| Height (cm)* | 162.53±7.79 | 164.60±6.77 | 0.277 |
| BMI (kg/m2)* | 21.70±2.21 | 22.40±2.43 | 0.248 |
| Duration of surgery (min)* | 81.67±26.50 | 106.33±32.42 | 0.002 |
| Duration of effective analgesia (min)^ | 138.50±22.52 | 247.00±36.69 | <0.001 |
Y: Year; kg: Kilogram; cm: Centimeter; min: Minutes
p<0.05 is considered statistically significant, *Values are expressed as Mean±SD, #Values are expressed as ratio; BMI: Basal metabolic index
Sensory block between two groups.
| Parameters | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Onset of sensory block (min)^ | 10.30±3.06 | 9.13±2.98 | 0.140 |
| Maximum block height* | T8 (T6-T8) | T6 (T6-T6) | <0.001 |
| Time to achieve maximum block height (min)^ | 15.20±3.07 | 13.73±2.53 | 0.048 |
| Time of segment regression T12 (min)^ | 92±23.21 | 181.00±36.93 | <0.001 |
| Time for two segment regression (min)^ | 39.33±8.80 | 84.17±14.32 | <0.001 |
^values are expressed as Mean±SD *Values are expressed as Median (IQR) p<0.05 is considered statistically significant, min=minutes
Tests used: Unpaired t-test/Mann whitney U tests
The Maximum Motor Blockade (MBS) was higher in the LD group, and the mean time for maximum motor blockade was longer in the LS group [Table/Fig-4]. The maximum motor blockade MBS at T10 was higher in the LD group. The mean heart rate was comparable at all corresponding time points in both groups except at 45 minutes has been presented in [Table/Fig-5]. The mean blood pressure was similar at all corresponding time points in both groups, as shown in [Table/Fig-6]. As illustrated in [Table/Fig-7], two patients in the LD group developed hypotension. Four patients in the LD group and five patients in the LS group experienced nausea. Additionally, the mean respiratory rate and mean oxygen saturation were comparable between the two groups at all time points, as shown in [Table/Fig-8,9]. Similarly, intraoperative VAS scores were comparable between the two groups except at 120 minutes, as shown in [Table/Fig-10].
Motor block between two groups.
| Parameters | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Time to achieve maximum blockade (min)^ | 20.67±3.14 | 19.43±3.31 | 0.145 |
| Maximum block achieved (MBS)* | 2 (2-3) | 3 (3-3) | <0.001 |
| MBS at T10* (At the time of onset of sensory block) | 2 (2-3) | 3 (3-3) | <0.001 |
| MBS at rescue analgesia* | 0 (0-0) | 0 (0-0) | 0.317 |
^values are expressed as Mean±SD, *median (IQR), min: minutes p<0.05 is considered statistically significant
Tests used: Unpaired t-test/Mann-Whitney U tests
Intraoperative heart rate between two groups.
| Time (min) | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Baseline | 78.10±11.43 | 78.93±11.26 | 0.777 |
| 5 | 75.97±12.38 | 76.33±11.90 | 0.907 |
| 10 | 74.00±12.14 | 75.93±11.96 | 0.537 |
| 15 | 71.43±9.99 | 74.20±12.38 | 0.345 |
| 20 | 70.30±9.02 | 73.87±12.20 | 0.203 |
| 25 | 68.57±10.30 | 74.30±11.76 | 0.049 |
| 30 | 69.60±10.26 | 72.50.±12.37 | 0.327 |
| 45 | 69.93±10.02 | 75.70±11.17 | 0.040 |
| 60 | 70.93±11.13 | 74.23±11.00 | 0.257 |
| 75 | 72.07±12.80 | 73.96±10.70 | 0.614 |
| 90 | 74.60±11.95 | 74.23±10.28 | 0.917 |
| 105 | 75±18.35 | 76.15±11.58 | 0.874 |
| 120 | 78.80±8.20 | 72.08±11.35 | 0.248 |
values are expressed as Mean±SD, min: minutes p<0.05 is considered statistically significant
Intraoperative mean blood pressure between two groups.
| Time (min) | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Baseline | 92.20±6.16 | 94.17±6.32 | 0.227 |
| 5 | 87.87±8.17 | 89.60±9.10 | 0.441 |
| 10 | 85.20±8.17 | 86.43±8.72 | 0.574 |
| 15 | 82.27±9.15 | 83.93±9.55 | 0.493 |
| 20 | 80.83±8.08 | 82.50±9.63 | 0.471 |
| 25 | 80.57±8.06 | 83.90±8.67 | 0.129 |
| 30 | 81.70±8.69 | 83.40±7.01 | 0.408 |
| 45 | 83.00±8.47 | 84.30±8.68 | 0.560 |
| 60 | 84.24±7.97 | 84.90±7.52 | 0.745 |
| 75 | 85.67±11.85 | 87.04±8.85 | 0.676 |
| 90 | 86.93±8.06 | 84.62±8.13 | 0.384 |
| 105 | 86.20±7.79 | 88.00±7.49 | 0.657 |
| 120 | 91.60±9.55 | 89.85±6.55 | 0.659 |
values are expressed as Mean±SD, min: minute; p<0.05 is considered statistically significant
Side-effects between two groups.
| Side-effects | Group LS (n=30) n (%) | Group LD (n=30) n (%) | p-value |
|---|
| Hypotension | 0 (0) | 2 (6.6%) | 0.150 |
| Bradycardia | 0 | 0 | - |
| Pruritus | 0 | 0 | - |
| Headache | 0 | 0 | - |
| Dizziness | 0 | 0 | - |
| Nausea | 5 (16.66%) | 4 (13.33%) | 0.718 |
| Vomiting | 1 (3.33%) | 1 (3.33%) | 1.000 |
| Shivering | 3 (10%) | 1 (3.33%) | 0.28 |
| Urinary retention | 0 | 0 | - |
| Hypernatremia | 0 | 0 | - |
| Post op Blood sugar level (mg/dL)* | 124.73±21.93 | 160.73±31.80 | <0.001 |
No.: Number; pts: patients; %: Percentage; *Mean±standard deviation
p<0.05 is considered statistically significant
Respiratory rate between two groups.
| Time (min) | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Baseline | 14.77±1.16 | 15.27±1.01 | 0.08 |
| 5 | 15.27±0.74 | 15.27±0.86 | 1.00 |
| 10 | 15.20±1.09 | 15.50±0.90 | 0.25 |
| 15 | 15.60±0.85 | 15.50±0.63 | 0.60 |
| 20 | 15.30±0.91 | 15.20±1.03 | 0.69 |
| 25 | 15.23±1.04 | 15.43±0.77 | 0.40 |
| 30 | 15.40±0.96 | 15.27±0.94 | 0.59 |
| 45 | 15.23±0.81 | 15.50±1.00 | 0.26 |
| 60 | 14.97±0.82 | 15.40±1.03 | 0.07 |
| 75 | 15.27±1.16 | 15.54±1.02 | 0.44 |
| 90 | 15.60±1.12 | 15.23±1.17 | 0.33 |
| 105 | 15.80±2.04 | 15.62±0.87 | 0.78 |
| 120 | 15.20±0.447 | 15.08±1.49 | 0.86 |
Saturation of oxygen (SpO2) between two groups.
| Time (min) | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Baseline | 100.00±0.00 | 100.00±0.00 | 1.00 |
| 5 | 99.93±0.25 | 99.93±0.25 | 1.00 |
| 10 | 99.93±0.25 | 99.93±0.36 | 1.00 |
| 15 | 99.90±0.30 | 100.00±0.00 | 0.07 |
| 20 | 99.90±0.30 | 100.00±0.00 | 0.07 |
| 25 | 99.90±0.40 | 100.00±0.00 | 0.17 |
| 30 | 100.00±0.00 | 99.97±0.18 | 0.32 |
| 45 | 99.97±0.18 | 100.00±0.00 | 0.32 |
| 60 | 100.00±0.00 | 99.97±0.18 | 0.28 |
| 75 | 100.00±0.00 | 99.96±0.19 | 0.45 |
| 90 | 99.93±0.25 | 100.00±00.0 | 0.19 |
| 105 | 99.80±0.44 | 100.00±00.0 | 0.09 |
| 120 | 99.80±0.44 | 100.00±00.0 | 0.09 |
Intraoperative VAS between two groups.
| Time (min) | Group LS (n=30) | Group LD (n=30) | p-value |
|---|
| Baseline | 5 (5-6) | 5 (5-6) | 0.920 |
| 5 | 4 (4.00-4.25) | 4 (3-4) | 0.043 |
| 10 | 3 (2-3) | 3 (0.75-3) | 0.064 |
| 15 | 0 (0-0.25) | 0 | 0.063 |
| 20 | 0 | 0 | 1.00 |
| 25 | 0 | 0 | 1.00 |
| 30 | 0 | 0 | 1.00 |
| 45 | 0 | 0 | 1.00 |
| 60 | 0 | 0 | 1.00 |
| 75 | 0 | 0 | 1.00 |
| 90 | 0 | 0 | 1.00 |
| 105 | 0 | 0 | 1.00 |
| 120 | 2 (0-2.50) | 0 | 0.003 |
Discussion
The prospective double-blind randomised study was conducted to compare the analgesic efficacy of levobupivacaine alone and levobupivacaine with dexamethasone in lower abdominal surgeries under SAB.
A total of 60 patients were included in present study. In a previous study, done in 2018 by Singh A et al., a comparison of 3 mL 0.5% levobupivacaine with the same volume of 0.5% racemic bupivacaine intrathecally for patients undergoing inguinal hernia surgeries was conducted. The study concluded that levobupivacaine is an effective alternative to bupivacaine as it has a shorter duration of sensory and motor block. It also allows early ambulation after surgeries with a lower incidence of intraoperative hypotension [13]. Based on the aforementioned study, authors decided to use a lower volume of isobaric levobupivacaine, specifically 2.5 mL of 0.5%, along with either normal saline or 1 mL of dexamethasone 4 mg as an intrathecal LA to compare analgesic efficacy in the conduct of lower abdominal surgeries. Authors observed that the addition of 4 mg (1 mL) of dexamethasone to 2.5 mL of 0.5% isobaric levobupivacaine provides a clinically longer duration of effective analgesia with early onset and better quality of sensory and motor block than 2.5 mL of 0.5% isobaric levobupivacaine alone. There were no severe side-effects observed except for statistically significant high blood sugar levels in the levobupivacaine and dexamethasone group, which were within the normal plasma blood sugar range and required no active intervention. Previous researchers have used dexamethasone as an adjunct to spinal anaesthesia in doses of 4-8 mg and have observed that the addition of dexamethasone improved the duration of analgesia, sensory and motor block with no significant haemodynamic changes and postoperative complications [13,14]. In the present study, after an extensive search, authors decided to use 4 mg of dexamethasone (1 mL) in addition to 2.5 mL of 0.5% isobaric levobupivacaine in a SAB.
The present study found that the duration of effective analgesia was significantly higher in the levobupivacaine with dexamethasone group (247.00±36.69 min) compared to the levobupivacaine group (138.50±22.52 min) with p<0.001. It was observed that dexamethasone with isobaric levobupivacaine provided a clinically longer duration of effective analgesia by approximately 110 minutes. Similar studies conducted by previous researchers have also observed that the addition of dexamethasone to a LA like bupivacaine increases the duration of sensory block and postoperative analgesia [9,11]. Bani-hashem N et al., studied the effect of adding intrathecal dexamethasone 8 mg to bupivacaine for spinal anaesthesia in orthopaedic surgery. The duration of sensory blockade and post-operative analgesia was significantly higher in the bupivacaine with dexamethasone group (119±10.69 and 401.92±72.44 minutes) compared to the bupivacaine group alone (89.44±8.37 and 202±43.67 minutes), respectively. They concluded that dexamethasone provided a better quality of block compared to bupivacaine alone. It also prolongs postoperative analgesia [9]. Haque MM et al., compared the efficacy of 0.5% hyperbaric bupivacaine with dexamethasone (5 mg) versus 0.5% hyperbaric bupivacaine alone in spinal anaesthesia for patients undergoing lower abdominal urological and lower limb orthopaedic surgeries. Total 72 adult patients were included. The study concluded that the duration of sensory blockade and postoperative analgesia was significantly higher in the bupivacaine with dexamethasone group (122.11±10.59 and 412.82±71.51 minutes) compared to the bupivacaine group alone (92.32±8.34 and 122.11±10.59 minutes) [11].
In present study, the onset of sensory block was 9.13±2.98 minutes in the levobupivacaine and dexamethasone group, and 10.30±3.06 min in the levobupivacaine group (p=0.140). This is supported by a previous study conducted by El-Shourbagy MA et al., who also observed similar findings [10]. The addition of dexamethasone to a LA like bupivacaine shortens the onset of sensory block to 5.3±1.7 minutes for the bupivacaine and dexamethasone group, compared to 5.9±1.9 minutes for the bupivacaine group [14]. Therefore, authors concluded that the addition of dexamethasone to levobupivacaine leads to an earlier onset of sensory block.
The median maximum block height was T6 in the dexamethasone with levobupivacaine group compared to T8 in the levobupivacaine group (p<0.001), which was statistically significant. The time to achieve maximum sensory block was 13.73±2.53 minutes in the dexamethasone with levobupivacaine group compared to 15.20±3.07 minutes in the levobupivacaine group, which was also statistically significant (p=0.048) and thus favourable for lower abdominal surgery. This is contrary to the previous study conducted by Bani-hashem N et al., which compared the addition of dexamethasone to bupivacaine with bupivacaine in orthopaedic surgery. The study showed that the onset time (time from injection of the drug into the intrathecal space to peak of sensory and motor block) was longer (11.27±2.08 minutes) in the bupivacaine with dexamethasone group compared to the bupivacaine group (10.95±1.87 minutes) with a p-value of 0.57 [9]. In the present study, the MBS was higher in the levobupivacaine and dexamethasone group 3 (3-3) as compared to the levobupivacaine group 2 (2-3) with p<0.001. The mean time to achieve the maximum motor blockade (assessed by the MBS) was 19.43±3.31 minutes and 20.67±3.14 minutes (p=0.145) in the levobupivacaine with dexamethasone and levobupivacaine groups, respectively. It was comparable between both groups.
Hypotension was observed in 2 (6.6%) in the levobupivacaine with dexamethasone group, while it was not observed in any patient in the levobupivacaine group. In contrast to present study, the study by Bani-hashem N et al., found that the incidence of hypotension was equal in the dexamethasone with bupivacaine group 7 (28%) and the bupivacaine group 7 (28%) patients [11]. Additionally, side-effects such as nausea, vomiting, and shivering were non significant and comparable between both groups. These results are in agreement with studies conducted by previous researchers [9,10]. Bani Hashem N et al., observed nausea and vomiting in 8% and shivering in 38% of the bupivacaine + dexamethasone group compared to the group in which only bupivacaine was given (20% and 32%, respectively) with a p-value of more than 0.005 [9]. Similarly, El-Shourbagy MA noted nausea, vomiting in 10%, and shivering in 42% of the bupivacaine+dexamethasone group compared to the control group (24% and 36%) with a p-value of more than 0.005 [10]. In present study, significant alterations of blood sugar levels were observed in patients who were administered dexamethasone along with levobupivacaine (160.73±31.80 mg/dL) compared to patients who were administered levobupivacaine with saline (124.73±21 mg/dL) intrathecally with a p-value of <0.001, which was statistically significant. Dexamethasone belongs to the class of long-acting synthetic corticosteroids with powerful anti-inflammatory as well as analgesic properties. It is known to cause hyperglycaemia as a side-effect by increasing gluconeogenesis, thereby inhibiting the action of insulin, along with enhancing the effect of counter-regulatory hormones such as glucagon and epinephrine. It also causes the activation of peroxisome proliferator-activated receptor α receptors, thereby further enhancing its gluconeogenic effect [14]. Various studies have previously proven the analgesic and anti-inflammatory effects of dexamethasone when used intrathecally [13], the results of which were found to be similar to the present study. The present study is the first study that has compared isobaric levobupivacaine with dexamethasone and isobaric levobupivacaine in lower abdominal surgeries under SAB.
Limitation(s)
The sample size was small. Further studies with a larger sample size, using various doses of dexamethasone along with other adjuvants and LAs with a different study population, can be planned for more conclusive results.
Conclusion(s)
Authors concluded that the addition of 4 mg (1 mL) of dexamethasone to 2.5 mL of 0.5% isobaric levobupivacaine provides a clinically longer duration of effective analgesia with early onset and better quality of sensory and motor block than 2.5 mL of 0.5% isobaric levobupivacaine alone. There were no severe side-effects observed except for statistically significant high blood sugar levels in the levobupivacaine and dexamethasone group, which were within the normal plasma blood sugar range and required no active intervention. Therefore, authors recommended using 4 mg of dexamethasone with 2.5 mL of 0.5% isobaric levobupivacaine for lower abdominal surgeries under SAB to achieve effective analgesia.
Y: Year; kg: Kilogram; cm: Centimeter; min: Minutes
p<0.05 is considered statistically significant, *Values are expressed as Mean±SD, #Values are expressed as ratio; BMI: Basal metabolic index
^values are expressed as Mean±SD *Values are expressed as Median (IQR) p<0.05 is considered statistically significant, min=minutes
Tests used: Unpaired t-test/Mann whitney U tests
^values are expressed as Mean±SD, *median (IQR), min: minutes p<0.05 is considered statistically significant
Tests used: Unpaired t-test/Mann-Whitney U tests
values are expressed as Mean±SD, min: minutes p<0.05 is considered statistically significant
values are expressed as Mean±SD, min: minute; p<0.05 is considered statistically significant
No.: Number; pts: patients; %: Percentage; *Mean±standard deviation
p<0.05 is considered statistically significant