The study of hard tissues in routine histopathological analysis requires the techniques of ground sectioning or decalcification for their demonstration [1]. Ground sections are helpful for studying inorganic components, while decalcification is necessary to study the organic components [2]. Decalcification is the process of removing inorganic calcium salts, leaving the organic portion intact and making the specimen amenable to sectioning [3]. Effective decalcification is essential for the microscopic assessment of hard tissue specimens. Various techniques are used for decalcification, including chelating methods, acid decalcification, and electrical ionisation. The rate of decalcification is influenced by factors such as the concentration of the decalcifying solution, temperature, agitation, density, and thickness of the specimen [2].
Controlling the decalcification step is crucial because it can have a detrimental effect on the specimen and lead to serious consequences in diagnosis [1,4]. Inadequate decalcification may render the tissue unsuitable for further processing and sectioning. Decalcifying agents can alter the proteins and nucleic acids present in the tissue, potentially biasing the morphology and results of histochemical analysis. Long-term exposure to formic acid during decalcification can affect mutations, gene amplifications, and sequencing [1].
Assessment techniques for decalcification include qualitative evaluation of histological preparations. Additionally, endpoint determination of decalcification can be achieved using physical, chemical, and mechanical methods [4,5]. X-ray is one of the most accurate methods for determining the endpoint of decalcification; however, taking X-rays of every specimen is often unnecessary and can increase radiation exposure for the technician. Physical testing involves bending the tissue or inserting a scalpel into the specimen.
The disadvantage of these methods is the risk of introducing tears and pinhole artifacts, which can damage the bone specimen. Chemical determination includes qualitative (precipitation) and quantitative (colourimetric and flame photometric) methods [4]. These procedures can be cumbersome, may deteriorate the specimen, and are often subjective.
The objective of present research was to determine the endpoint of decalcification for hard tissue specimens using a colour palette. The colour palette used in present study was previously patented (202241047215). The present simple and safe method can be employed to determine the endpoint of decalcification without any subjective error or damage to the specimen.
Materials and Methods
The present cross-sectional study was conducted in the Department of Oral Pathology at Saveetha Dental College and Hospitals in Chennai, Tamil Nadu, India, over a period of 5 months. The study was approved by the Institutional Ethical Committee (SRB/SDC/OPATH-2105/23/078).
Inclusion and Exclusion criteria: Freshly extracted teeth without any developmental abnormalities were included in the study, while teeth with attrition, abrasion, or carious lesions were excluded.
Study Procedure
A total of 25 hard tissue specimens were collected, consisting of 10 bone samples and 15 tooth samples. These samples were collected from patients who visited the Department of Oral and Maxillofacial Surgery for orthodontic extractions. Bone samples, measuring 2×2 cm, were collected from excision specimens. The collection of bone samples was performed only after diagnosis and did not affect the grossing or evaluation of the tumour and margins. All samples were fixed in neutral buffered formalin and subsequently subjected to decalcification using 20% formic acid. Every 24 hours, the specimens were evaluated, and 1 mL of decalcifying fluid was withdrawn on days 1, 3, 5, and 7 for further analysis. In the present study, authors employed the Von Kossa technique to determine the endpoint of decalcification. The present technique uses a 1-5% silver nitrate solution and produces a colour change when it reacts with calcium. The colour change ranges from yellow (immediately after additing) to grey and then to black.
Preparation of silver nitrate: Total 25 grams of silver nitrate was added to a test tube containing 500 mL of distilled water. The solution was stored in a light-resistant bottle in the refrigerator.
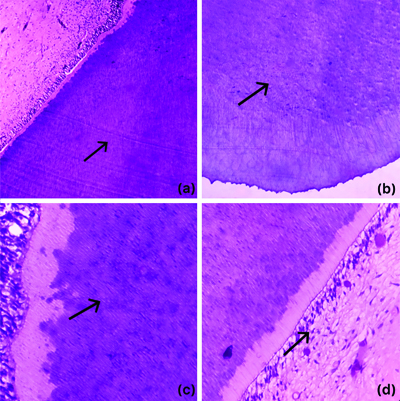
Evaluation of the samples: To evaluate the endpoint of decalcification, 1 mL of decalcifying fluid was taken in a test tube, and 1 mL of silver nitrate solution was added. The solution was mixed and agitated for 5-10 minutes. The colour change was noted after 10 minutes and 24 hours and compared with the colour palette (Official Journal of The Patent Office-202241047215) [Table/Fig-1]. On the right-side of the colour palette, the colours ranged from dark brown and chocolate brown to caramel brown, clay brown, and light brown colour. On the left-side, the colours ranged from silverish black to silverish grey, slate grey, seal grey, and light grey. A darker colour (dark brown/silverish black) indicated a greater amount of calcium release into the decalcifying agent, while a lighter colour (light brown/light silver) indicated a lesser amount of calcium release [Table/Fig-2,3]. The decalcification procedure continued until lighter shades of brown and silver-black were obtained. Once the lighter colour was achieved, the specimen was submitted for tissue processing and subsequently sectioned using a semi-automated microtome. The sections were stained with haematoxylin and eosin stain [Table/Fig-4,5a-d]. The H&E slides of the bone and teeth samples were evaluated by two oral pathologists based on overall morphology and staining, cytoplasmic and nuclear detail, cellular outline, presence of inflammatory cells, and integrity of Red Blood Cells (RBCs). The slides were graded as follows: 1-poor, 2-fair, 3-good, and 4-excellent (as described by Mathai AM et al.) [6].
Shows the comparison of the colour obtained in the test tube with the colour palette. On the right-side of the colour pallete the colour ranged from dark brown, chocolate brown colour, caramel brown, clay brown to light brown colour. On the left-side it ranged from silverish black to silverish grey, slate colour, seal grey, and light grey.

This figure represents the colour change that was noted 10 minutes after the addition of silver nitrate on day 1, day 3, day 5, and day 7. The colour change ranged from darker brown to light brown. Light brown denotes the end point of decalcification.

This figure represents the colour change that was noted 24 hours after the addition of silver nitrate on day 1, day 3, day 5, and day 7. The colour change ranged from silverish black to light grey. Light grey denotes the end point of decalcification.

a&b) Photomicrograph shows H&E-stained Sections of bone with osteons with concentric lamellae and interstitial lamellae (H&E, 100X); c&d) Osteocytes within the osteocyte lacunae (H&E, 400X).
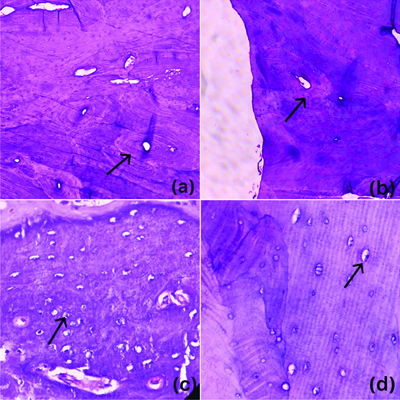
a-c) Sections of tooth with dental tubules (H&E 100X); (d) Dentin-pulp interface with odontoblasts and pulp tissue (H&E,400X).
Statistical Analysis
The grades obtained were entered into Microsoft Excel 2021. Descriptive analysis and percentage analysis were conducted. Kappa statistics were performed to evaluate inter-rater reliability.
Results
Different grades of brown and silverish-black colours were observed. The colour gradation indicated the amount of calcium released in the solution. The darker colours indicated incomplete decalcification, while the lighter colours indicated that the decalcification was complete. It was noted that different shades of brown were obtained when the colour change was noted after 10 minutes of adding the silver nitrate solution. Different shades of silverish-black were observed after 24 hours. The colours obtained were matched with the corresponding colours on the colour palette.
On day 1, a chocolate brown colour was observed after 10 minutes of adding the silver nitrate solution; on day 3, caramel brown; on day 5, clay brown; and on day 7, light brown. When the test tubes were evaluated after 24 hours, day 1 showed silverish black, day 3 showed silverish grey, day 5 showed seal gray, and day 7 showed light grey. Once the lighter colours were obtained, the samples were processed, sectioned, and evaluated by two oral pathologists. Kappa statistics showed a score of 0.835, indicating very good interobserver agreement. Total 8 (80%) samples showed excellent staining, while 2 (20%) samples showed a good staining score. With respect to the teeth samples, 14 (93.3%) showed excellent staining, and 1 (6.66%) tooth showed a good staining score.
Discussion
The chemical method of decalcification is the most widely used technique in routine histopathological analysis. This method uses acids (both strong/inorganic and weak/organic) or chelating agents for decalcification [7]. A good decalcifying agent should effectively remove calcium with minimal damage to the specimen and should not impair subsequent staining procedures. In addition to the chemical agents used, other factors that play an important role in decalcification include the size and type of tissue, the type of fixation used, the concentration of the decalcifying agent, agitation, temperature, and suspension [2].
The most critical aspect of decalcification is determining whether sufficient calcium has leached out of the hard tissue specimen to allow for further processing steps. Incompletely decalcified tissue can lead to poor microtomy, damaging both hard and soft tissue, resulting in excess basophilia in H&E staining and disrupted soft tissue. Conversely, excessive decalcification can result in the loss of basophilia and an increase in eosin staining, leading to poor preservation of nuclear and cytoplasmic details [8]. The endpoint determination of decalcification can be performed through mechanical, physical, or chemical methods, as well as radiography or X-ray methods and weight change calculations [3]. A less invasive method for endpoint determination involves testing the decalcifying agent itself rather than testing the specimen itself. This method is most often used with a formic acid decalcifier [2].
It has been reported that radiographs are the most reliable method for determining the endpoint of decalcification, while chemical tests are considered less reliable, as any stronger acid other than 10% formic acid may yield false positive results in cloudy precipitation tests [3,9]. Secondary calcium phosphate may reprecipitate during the decalcification process, which could affect the efficacy of the radiographic method for endpoint determination. Flame photometry is also used to determine the endpoint of decalcification. This technique reduces the need to repeat experiments, saving both money and time, and allows for the measurement of calcium even in small samples and at low concentrations [7]. A variation of the classic oxalate test has been employed to test acid decalcifying solutions for endpoint determination in Ethylene Diamine Tetraacetic Acid (EDTA) decalcification. Charnwichai P et al., performed EDTA decalcification using an ultrasonic cleaner/microwave oven and achieved better results [10]. A colourimetric method for determining the endpoint of decalcification using flame photometry and spectrophotometry has also been reported [4].
In the present study, authors used the von Kossa technique. Von Kossa, a Hungarian physician, developed this method over 120 years ago, treating kidney sections with a 1-5% silver nitrate solution, which resulted in a yellow colouration of the calcium present in the tissue. This transient colour gradually changed to grey and then to black. Until now, this remains the most popular method for detecting mineralisation [11]. The von Kossa stain was initially referred to as a “silver nitrate method for calcium deposits” [12]. It is widely used in histopathology to detect the presence of abnormal calcium deposits in tissues. The key colourimetric factor in present technique is the silver nitrate solution. The principle of this technique is based on the transformation of calcium salts into silver salts. When calcium ions are bound to phosphates, they are replaced by silver ions from the silver nitrate solution. When exposed to a light source, the silver phosphates transform into metallic silver deposits through photochemical degradation. The von Kossa stain is useful for detecting calcifications in tumours, assessing static cancellous histomorphometry in rodents, providing a sharp distinction between mineralised and unmineralised bone, evaluating mineralisation in cultured cells, monitoring bone resorption, and identifying calcinosis conditions such as chondrocalcinosis, nephrocalcinosis, and cutaneous calcinosis, as well as metastatic calcinosis [13-16]. The von Kossa stain clearly demonstrates calcium better than haematoxylin and is considered a superior technique when combined with specimen radiography to provide a good radiologic-pathologic correlation [17].
Guerlain J et al., stated that microcalcifications are a specific feature of thyroid malignancy and are associated with psammoma bodies, which contain microcrystals of calcium phosphate apatite [18]. The von Kossa staining can detect these phosphates in the presence of acidic materials without quantifying the amount of calcium, thus enabling the detection of malignant microcalcifications associated with psammoma bodies [12]. Furthermore, Kunitake JAMR et al., mentioned the use of the von Kossa stain in identifying calcium deposits in breast biopsies, including acinar secretions, cyst fluid, ductal ectasia, and papillomatosis [17]. Proper decalcification of bone is essential for accurate diagnosis [19].
In the present study, the utility of the von Kossa stain/silver nitrate crystals has been employed for endpoint determination of decalcification. The calcium in the decalcifying agent reacts with the silver nitrate solution, producing a colour change. A transition from a darker shade to a lighter shade indicates a decrease in calcium concentration in the solution. A lighter shade denotes the completion of decalcification.
Limitation(s)
There were few limitations in present research. Authors used only 20% formic acid as a decalcifying agent; other commonly used decalcifying agents and different concentrations of formic acid need to be evaluated. Furthermore, authors only considered normal bone and tooth samples for assessing the endpoint of decalcification using the colour palette. Pathological tissues and other calcified samples were not addressed in present study.
Conclusion(s)
The present study aims to enumerate the use of a colour palette with silver nitrate solution as a simpler method for detecting the decalcification endpoint without using the specimen itself. This method provides both immediate and confirmatory results through colour changes, thereby minimising subjective decisions. Consequently, it can be beneficial for all bone pathologies, particularly for larger bone specimens where physical testing, radiographs, and flame photometry are challenging. Further, research can be conducted on this colour palette, which could be modified into even simpler devices that may also have applications in forensics.