The human GIT harbours a diverse and dynamic community of microorganisms, collectively known as the gut microbiome. This intricate ecosystem, comprising bacteria, viruses, fungi, and other microorganisms, plays a pivotal role in maintaining health by influencing digestion, nutrient absorption, and immune function [1-3]. Alterations in gut flora due to various factors such as lifestyle, poor nutrition, age, mode of birth, and the use of medicines like antibiotics can lead to a diverse range of symptoms and result in deleterious consequences [1-4]. Dysbiosis is associated with gastrointestinal disorders, metabolic diseases, and neurological ailments [1,2,5,6]. Moreover, the influence of the gut microbiome extends beyond the confines of the digestive system, exerting profound effects on immune function, including susceptibility to infections [7-9].
Many studies are investigating the potential benefits of restoring gut flora to improve children’s immunity and growth parameters [4,5]. One such approach is the administration of probiotics, which are live, beneficial microorganisms [4-6]. Probiotics, when administered in sufficient dosage (108-1011 CFU), can colonise the GI tract, suppress pathogenic bacteria, and may improve health [10]. Recent years have witnessed a growing interest in the potential therapeutic applications of probiotics to modulate the gut microbiome and promote health [11,12]. The potential benefits of probiotics are vast and multifaceted, encompassing the ability to restore microbial equilibrium, suppress the growth of pathogenic microorganisms, fortify gut barrier function, and even modulate immune responses [13,14]. GBI-30, 6086 (GanedenBC30) has been reported to enhance the immunological response of healthy human adult cells to common viral causes of Upper Respiratory Tract Infections (URTI) ‘ex vivo’ [15].
Malnutrition poses a significant health challenge in underdeveloped nations, often resulting in recurrent infections and impairments in weight, height, and behaviour. This condition places a substantial economic strain on governments and healthcare systems [22-25]. Malnourished children are particularly susceptible to infections due to compromised immune function, and probiotics may offer a potential solution to enhance their immune response and reduce infection rates. However, while several studies have explored the benefits of probiotics in healthy children, there is a significant gap in the literature regarding the specific effects of probiotics on infection rates in malnourished children [18,21,22,24,26]. Moreover, while various probiotic strains have been studied, the potential of B. coagulans SNZ 1969® in this context remains largely unexplored. Therefore, this study was designed to evaluate whether the administration of B. coagulans SNZ 1969® has any role in reducing infections and improving wellbeing in malnourished children. The paramount aim was to provide data that can inform evidence-based interventions aimed at ameliorating the infection challenges in this vulnerable population.
Materials and Methods
This was a randomised, double-blind, placebo-controlled clinical trial conducted from April 2021 to November 2022 through a collaborative effort between the Departments of Pharmacology and Paediatrics at BJGMC and Sassoon Hospitals in Pune, Maharashtra, India. Ethics Committee approval was obtained via letter no. BJGMC/IEC/pharmac/D-0121026-026, and the study was registered prospectively with the Clinical Trials Registry-India (CTRI/2021/03/031870). Informed consent was obtained from the parents or legal guardians of each participant. The study adhered to the principles outlined in the World Medical Association Declaration of Helsinki and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use-Good Clinical Practice (ICH-GCP) guidelines.
Inclusion criteria: Potential participants from the paediatric Outpatient Department (OPD) underwent screening based on specific eligibility criteria. Children aged 1 to 5 years who were malnourished (with a weight-for-height z-score between -3.0 and -2.0) [27] were included in the study.
Exclusion criteria: Children outside the specified age range, those with chronic inflammatory bowel diseases, immunocompromised status or human immunodeficiency virus-positive status, malignancy, metabolic diseases, chronic respiratory tract diseases, malformations, recent surgeries, congenital cardiac defects, food allergies, varied nutritional status, and recent antibiotic use within two weeks prior to enrollment were excluded from the study.
Sample size estimation: A study by Shukla Y et al., has shown that malnourished children experienced diarrhoea (2.7 episodes with a standard deviation of 3.4 in the last three months) and Acute Respiratory Infections (ARI) (1.6 episodes with a standard deviation of 2.9 in the last 3 months), whereas normal children, on average, presented with diarrhoea (1.08 episodes with a standard deviation of 1.7 in the last three months) and ARI (0.06 episodes with a standard deviation of 0.5 in the last three months) [28]. Based on this study, authors assumed that children aged between 1 and 5 years would have 3 episodes (2.7 rounded up to 3) of either GI or RTIs in three months, with a standard deviation of 3.4. Authors anticipated a reduction from 3 episodes to 1 episode in three months in the probiotic group, taking into consideration the normal children in the referenced study [28].
Considering the above factors, the sample size calculation indicated that 35 participants would be needed in each group to detect the difference in the number of GI or RTI episodes between the probiotic and placebo groups, with 95% confidence, 80% power, α=0.05, and β=0.2. Assuming an approximate 10% loss to follow-up, the required sample size was determined to be 80 subjects, of which 40 would receive probiotics and the remaining 40 would receive a placebo.
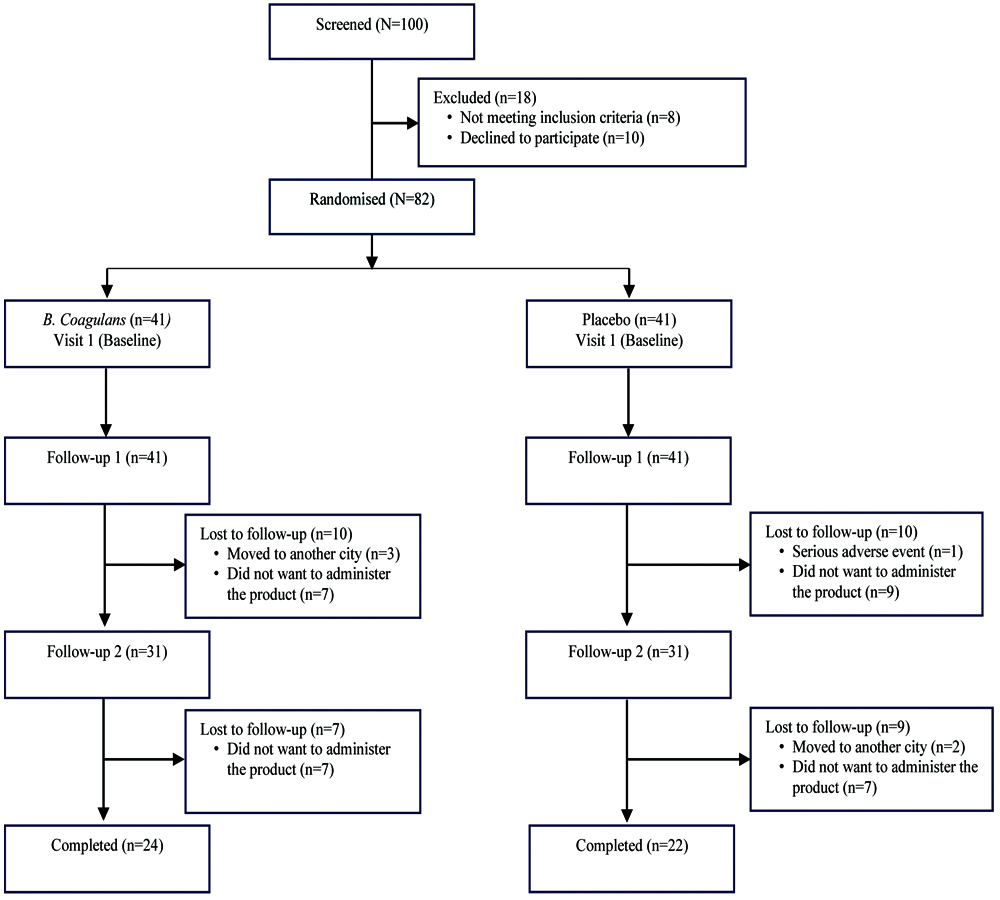
Of the 100 malnourished children screened, a total of 82 were included in the study, with 41 assigned to the probiotic intervention group and 41 to the placebo control group. Of these, only 46 children (24 in the probiotic group and 22 in the placebo group) completed the full three months study, as 36 children dropped out during the study period [Table/Fig-1]. The most common reasons for study discontinuation included children relocating to another city (n=5), participants choosing not to continue (n=30), and one child enrolled in the placebo group who reported a serious adverse event of a body rash. The eligible subjects were randomised into the study arms according to the CONSORT flow chart [Table/Fig-1].
Study Procedure
Eligible participants were enrolled in the study, and an independent third party generated a randomisation code to assign each participant a unique serial number and corresponding drug treatment allocation, either to the probiotic group (B. coagulans SNZ 1969®) or the placebo group. The justification for the dosage of 300 million CFU thrice daily (900 million CFU per day) for a period of three months was based on the Bacillus coagulans GBI-30, 6068 study by Anaya-Loyola MA et al., which administered one billion CFU per day for three months [21]. Each 5 mL of B. coagulans SNZ 1969® contained 300 million CFU (Batch no: SPDS 01) in a vehicle, while the placebo consisted of the vehicle, i.e., pharmaceutical-grade sugar, lactose (in minute quantities), flavouring additives, and stabilisers, devoid of B. coagulans SNZ 1969® [21]. The administration was carried out for a period of three months, and parents or guardians were instructed on the proper storage and consumption of the medication. To ensure adherence to the treatment regimen, participants were provided with diaries to record their intake of the study medication. The study team regularly reviewed these diaries during follow-up visits to monitor adherence. Both study medications were provided by Sanzyme Biologics Pvt. Ltd., in identical bottles, free of charge. All subjects received 5 mL of the respective formulation thrice daily for three months.
Follow-up visits: The study comprised a total of seven visits, including four in-person visits to the OPD and three telephonic check-ins. On the day of randomisation (Visit 1), participants received either the probiotic or placebo, along with diaries and instructions for medication storage and consumption. Subsequent in-person follow-up visits occurred on day 30 (±7 days), day 60 (±7 days), and day 90 (±7 days) postrandomisation. During these in-person OPD visits, body weight, height, head circumference, mid-upper arm circumference, and essential data on symptoms, infections, and treatment compliance were recorded.
Telephonic visits (A, B, and C) were scheduled for day 15, day 45, and day 75 (±7 days). In cases where patients missed their appointments, proactive phone calls were made to remind them to complete their diaries and attend the OPD visit. During the telephonic check-ins, the study investigator engaged with parents or guardians to inquire about critical aspects, including the child’s adherence to the prescribed probiotics, any infections during the monitoring period, reported side-effects, and visits to local general practitioners.
Infection definitions and compliance: GI infections were defined as instances of diarrhoea involving three or more bowel movements of soft or liquid stools within a 24-hour period, with or without fever or vomiting [29]. Severe diarrhoea was identified by high-output diarrhoea with elevated stool volumes (more than eight episodes per day), accompanied by signs of severe dehydration [30].
Respiratory Tract Infections (RTIs) encompassed both URTIs and Lower Respiratory Tract Infections (LRTIs), as diagnosed by a physician. LRTIs included conditions such as pneumonia, bronchitis, and bronchiolitis, while URTIs included various acute conditions such as rhinitis, laryngitis, pharyngitis, and acute otitis media. The criteria for diagnosing pneumonia included tachypnoea, nasal flaring, cough, shortness of breath, fever, headache, and lethargy. Severe pneumonia was identified by tachypnoea, lower chest wall indrawing, stridor in a calm child, cyanosis, and additional symptoms [31]. Chest X-ray findings indicative of pneumonia and an increased white blood cell count were also considered.
Treatment compliance was defined as consuming at least 80% of the prescribed treatment [32] and was confirmed through diary reviews conducted by parents. Cases of GI or RTI symptoms in either study arm were treated according to standard treatment guidelines.
Outcomes
The primary outcome of the study was to determine the number of episodes of GI or RT infections. The secondary outcomes included various measures, such as the total days of illness, the total number of days with symptoms related to RT or GI infections, and the number of Emergency Department (ED) visits prompted by symptoms of RTIs or GI infections. Additional parameters assessed included the percentage of children experiencing episodes of vomiting or diarrhoea, the number of severe diarrhoeal episodes, the number of GI infections lasting beyond two days, and the number of RTIs lasting more than three days. Furthermore, the study examined the proportion of children experiencing at least one episode of GI or RTIs over various timeframes between the groups. Finally, the assessments also included the number of days of work lost by parents and the number of days children were absent from preschool.
Statistical Analysis
The intent-to-treat analysis was conducted to compare both the probiotic and placebo groups. Descriptive statistics, including proportions, means (standard deviations), and medians, were used to assess the characteristics of participants in the two groups. Qualitative data were analysed using the Chi-square test, and where this was not feasible, Fisher’s exact test was employed. Quantitative data were first checked for normality using the Shapiro-Wilk test. Since the data were not normally distributed, they were represented as median±standard deviation and analysed using the Wilcoxon rank-sum test/Mann-Whitney U test. All p-values were two-sided, and differences were considered significant if the p-value was <0.05. Calculations were performed online at: https://www.socscistatistics.com/tests.
Results
Baseline demographics and clinical characteristics: There were no significant differences (p-value >0.05) between the two groups in terms of gender distribution, age, or birth weight at baseline. Similarly, the baseline demographics, birth and feeding history, as well as vaccination status, were not different between the two groups [Table/Fig-2]. Additionally, throughout the study, both groups exhibited similar growth patterns in anthropometric characteristics such as height, weight, head circumference, and mid-upper arm circumference [Table/Fig-3].
Baseline demographics and clinical characteristics.
| Parameters | Probiotic (n=41) | Placebo (n=41) | p-value |
|---|
| Sex: Female, n (%) | 23 (56.1) | 22 (53.6) | 0.823 |
| Age, years (±SD) | 2.85±1.22 | 2.66+1.24 | 0.471 |
| Children with previous history of RTIs, n (%) | 33 (80.4) | 29 (70.7) | 0.306 |
| Children with previous history of AGIs, n (%) | 17 (41.4) | 12 (29.2) | 0.248 |
| Mothers that were C section, n (%) | 12 (29.2) | 14 (34.1) | 0.635 |
| Children that were preterm, n (%) | 10 (24.4) | 7 (17.7) | 0.667 |
| Weight of children at birth, kg (±SD) | 2.59±0.59 | 2.64±0.47 | 0.659 |
| Incubated, n (%) | 8 (19.5) | 4 (9.7) | 0.331 |
| Duration of children breastfed | <1 year | 7 (17.0) | 6 (14.6) | 1 |
| 1-3 years | 23 (56.0) | 22 (53.6) | 1 |
| Still on feed | 11 (26.8) | 13 (31.7) | 0.802 |
| Children that were vegetarian, n (%) | 18 (43.9) | 14 (34.1) | 0.356 |
| Children that included curd in diet, n (%) | 18 (43.9) | 17 (41.4) | 0.832 |
| Children with history of ICU admission, n (%) | 8 (19.5) | 4 (9.7) | 0.333 |
| Weight at visit 1, kg (±SD) | 10.04±2.02 | 9.30±2.10 | 0.147 |
| Height at visit 1, cm (±SD) | 87.10±10.10 | 83.3±11.5 | 0.327 |
| HC at visit 1, cm (±SD) | 46.30±1.74 | 45.90±1.80 | 0.259 |
| MUAC at visit 1, cm (±SD) | 14.10±1.12 | 13.80±1.20 | 0.216 |
| Children going to Anganwadi, n (%) | 22 (53.6) | 13 (31.7) | 0.740 |
| Average number of dependants, (n±SD) | 50.2±1.15 | 4.5±1.16 | 0.077 |
| Monthly income, INR | <10,000 | 1 (2.4) | 1 (2.4) | 0.898 |
| 10,000-20,000 | 37 (90.2) | 38 (92.6) |
| >20,000 | 3 (7.3) | 2 (5.9) |
AGI: Acute gastrointestinal illness; HC: Head circumference; ICU: Intensive care unit; INR: Indian rupees; kg: kilogram; MUAC: Mid upper arm circumference; RTI: Respiratory tract infection
Change in weight, height, head circumference (HC), mid upper arm circumference (MUAC) over study period in the B. coagulans and placebo group.
| B. coagulans | Visit 1 | Follow-up 1 | Follow-up 2 | Follow-up 3 |
|---|
| Weight (kg) | 10.04±2.02 | 10.31±2.03 | 10.48±1.09 | 10.5±1.7 |
| Height (cm) | 87.1±10.2 | 87.7±10.7 | 87.9±11.6 | 88±11.2 |
| HC (cm) | 46.3±1.74 | 46.7±6.7 | 47.1±1.33 | 47.5±1.3 |
| MUAC (cm) | 14.1±11.2 | 14.3±1.03 | 14.6±0.89 | 14.7±0.9 |
| Placebo |
| Weight (kg) | 9.30±2.1 | 9.6±1.9 | 9.8±1.8 | 10.2±1.8 |
| Height (cm) | 83.3±11.5 | 83.4±11.3 | 83.8±13.0 | 84.3±10.5 |
| HC (cm) | 45.9±1.8 | 46.1±1.6 | 46.2±1.4 | 46.4±1.5 |
| MUAC (cm) | 13.8±1.2 | 14.1±1.8 | 14.2±0.9 | 14.3±0.9 |
HC: Head circumference; MUAC: Mid upper arm circumference
Efficacy outcomes: The study found fewer episodes of GI or RTIs in the probiotic group compared to the placebo group (1.32 vs. 1.48), although the difference was not statistically significant (U=689.5, p-value=0.077). No significant differences (p-value >0.05) were observed in the proportion of children experiencing at least one episode of GI and RTIs over various timeframes between the two groups [Table/Fig-4].
Comparison of infection rates, illness duration, and impact between probiotic and placebo groups.
| Parameter | Probiotic (n=41) | Placebo (n=41) | p-value |
|---|
| Number of episodes of Gastrointestinal (GI) or respiratory tract infections, mean±SD | 1.32±0.57 | 1.48±0.67 | 0.077 |
| Proportion of children experiencing at least one episode of acute GI infections over study period, n (%) |
| Follow-up 1 (Day 1-30) | 2 (4.87) | 4 (9.75) | 0.675 |
| Follow-up 2 (Day 31-60) | 0.00 | 2 (4.87) | 0.43 |
| Follow-up 3 (Day 61-90) | 0.00 | 2 (4.87) | 0.43 |
| Proportion of children experiencing at least one episode of Respiratory Tract Infection (RTI) over study period, n (%) |
| Follow-up 1 (Day 1-30) | 37 (90.2) | 39 (95.12) | 0.675 |
| Follow-up 2 (Day 31-60) | 24 (58.5) | 27 (65.8) | 0.22 |
| Follow-up 3 (Day 61-90) | 0.00 | 6 (14.6) | 0.44 |
| Proportion of children developed LRTI, n (%) | 1 (2.4) | 2 (4.87) | 0.17 |
| Number of Emergency Department (ED) visits, n (%) | 1 (2.4) | 3 (7.3) | 0.615 |
| Number of GI infections lasting longer than 2 days, n (%) | 3 (7.3) | 4 (9.7) | 1 |
| Number of Respiratory Tract Infections (RTI) lasting longer than three days, n (%) | 34 (82.9) | 38 (92.6) | 0.311 |
| Total days of illness, mean±SD | 6.36±2.61 | 8.09±3.27 | 0.009 |
| Total days with GI symptoms, mean±SD | 0.24±0.88 | 0.53±1.22 | 0.4593 |
| Total days with respiratory symptoms, mean±SD | 6.12±2.19 | 7.56±3.65 | 0.0687 |
| Number of workdays lost by parents, mean±SD | 1.20±0.71 | 1.30±1.06 | 0.24 |
| Number of pre-school days lost by children, mean±SD | 0.30±0.91 | 0.50±1.38 | 0.928 |
GI: Gastrointestinal; LRTI: Lower respiratory tract infection
Vomiting and diarrhoea were less common in the probiotic group {4 (9.7%) and 3 (7.5%), respectively} compared to the placebo group {8 (19.5%) and 4 (9.7%), respectively}, but the differences were not statistically significant (p-value >0.05). No severe diarrhoea episodes were reported in either group.
The duration of GI and RTI episodes lasting longer than 2 and 3 days, respectively, showed no significant differences (p-value >0.05) between the groups [Table/Fig-4]. However, the probiotic group exhibited a trend toward reduced infection duration. A lower incidence of LRTIs was observed in the probiotic group {1 (2.4%)} compared to the placebo group {2 (4.87%)}, although this difference was not statistically significant (p-value=0.17). A statistically significant reduction in total illness days was demonstrated in the probiotic group (p-value=0.009; [Table/Fig-4]). Favourable trends were observed in the probiotic group for total days with respiratory symptoms (6.12±2.19 vs. 7.56±3.65; U=643, p-value=0.0687) and GI symptoms (0.24±0.88 vs. 0.536±1.2; U=760, p-value=0.4593), although these were not statistically significant.
Emergency department visits for GI and RTI symptoms were slightly lower in the probiotic group compared to the placebo group {1 (2.4%) vs. 3 (7.3%); p-value=0.615}.
No significant differences were observed in parents’ lost workdays (U=713.5, p-value=0.24) or children’s missed preschool days (U=830.5, p-value=0.928) between the groups [Table/Fig-4].
Safety outcomes: In the safety analysis, the administration of both B. coagulans SNZ 1969® and the placebo was well-tolerated throughout the study, with no reports of severe adverse events. However, it is worth noting that one case of a body rash was reported in the placebo group, whereas no similar events were reported in the probiotic group.
Discussion
In paediatric healthcare, preventing infections is a paramount concern. Several research investigations have highlighted the advantages of probiotics in preventing infectious diseases. However, there is a lack of studies assessing the efficacy of B. coagulans, commonly known as Sporlac in India, in reducing infections among malnourished children, who represent a vulnerable population. Among the diverse range of probiotic strains, B. coagulans SNZ 1969® has gained attention for its potential in preventing infections. However, its use in children, especially high-risk populations such as underweight children, has been relatively underexplored. Therefore, this study was devised to assess the effectiveness of B. coagulans in reducing infections. One group was given B. coagulans SNZ 1969®, while another group received a matching placebo for a duration of three months. The study also assessed the impact of B. coagulans SNZ 1969® treatment on reducing the total number of days of illness and various aspects of RTIs and GI infections. The probiotic B. coagulans SNZ 1969® treatment was well-tolerated, with no adverse events reported in the study.
The results revealed that B. coagulans SNZ 1969® had a significantly positive impact on reducing the total days of illness. This was in line with a meta-analysis conducted by King S et al., which also reported that individuals receiving probiotic interventions experienced significantly shorter illness episodes compared to those who received a placebo [22]. Furthermore, the study observed favourable trends for the probiotic group in terms of other assessments, which included the occurrence of at least one GI or RTI episode within specific timeframes, the duration of illness, infections lasting longer than 2-3 days, and ED visits. However, these favourable trends associated with B. coagulans SNZ 1969® supplementation could not achieve statistical significance. These findings are consistent with studies by Hojsak I et al., and Sazawal S et al., which yielded mixed results in the prevention of GI and RTI infections among children [26,33].
The study also found that the occurrence of URTIs and LRTIs was lower in the probiotic group than in the placebo group. This aligns with findings from a Mexican study, wherein the administration of the probiotic Bacillus coagulans GBI-30, 6086® showed reduced incidences of URTIs and GI infections [21]. Similarly, the number of ED medical visits for children with symptoms of GI infections and RTIs was lower in the B. coagulans SNZ 1969® group than in the placebo group. These findings suggest that B. coagulans SNZ 1969® may have a positive immunomodulatory effect in children, potentially leading to a reduction in the incidence of URTIs and LRTIs. However, further studies with larger sample sizes and longer follow-up periods are needed to confirm these findings.
In line with other parameters, the incidence of vomiting and diarrhoea was also lower in the probiotic group than in the placebo group. These findings are consistent with those of a previous study by Sur D et al., which found that probiotic intake can reduce the incidence of acute diarrhoea in children [34]. While the probiotic group had fewer days of preschool missed by children compared to the placebo group, no significant difference was observed when the two groups were compared. Similarly, there was no significant difference between the probiotic and placebo groups regarding the number of workdays lost by parents. In line with these results, a meta-analysis demonstrated that children who received probiotics had fewer days of absence from daycare or school compared to those who received a placebo [35,36]. Another study by Cazzola M et al., on the efficacy of synbiotic supplementation in preventing common winter diseases in children found a reduced number of school days missed due to sickness in the synbiotic group compared to the placebo group [35].
In terms of safety, both B. coagulans SNZ 1969® and the placebo were well-tolerated throughout the study. There was only one reported adverse event observed in the placebo group, while none were reported in the probiotic group, reinforcing the superior safety profile of B. coagulans SNZ 1969®. Therefore, B. coagulans SNZ 1969® can be considered relatively safe, which aligns with earlier published reports [37-39].
In summary, the study found that treatment with B. coagulans SNZ 1969® was generally well-tolerated and had a significant positive effect on reducing the total number of sick days. It also showed promising trends in decreasing Respiratory Tract (RT) infections and GI infections compared to a placebo. However, it is important to note that while there was a downward trend favouring the probiotic group, statistical significance was not reached for some assessments, including the occurrence of various RTIs and GI events, the percentage of children experiencing GI infections or RTIs, and infections lasting longer than 2-3 days. Although the study focused on moderately underweight children, its findings have broader implications, suggesting potential benefits of B. coagulans SNZ 1969® supplementation for the general population, which needs evaluation in the respective clinical set-up. This probiotic emerges as a promising option not only for children with nutritional deficiencies but also for their healthy counterparts.
Limitation(s)
To the best of current knowledge, this was the first study that assessed the efficacy and safety profile of B. coagulans SNZ 1969® in underweight children under five years of age. Although valuable insights were gained, several limitations were identified. These include a small sample size and difficulties in recruitment due to the Coronavirus Disease-2019 (COVID-19) pandemic, which may have impacted the statistical significance of the findings. Monitoring compliance with the administered syrup was also challenging. Furthermore, the rate of premature discontinuation was high. Although parents did not provide any specific reasons, it could have been due to the taste of the medication or the need to administer it three times a day for an extended period, as per the protocol.
Conclusion(s)
The study results revealed that B. coagulans SNZ 1969® had a significant positive impact on reducing the total days of illness and showed favourable trends toward a decrease in RTIs and GI infections compared to a placebo. These findings contribute to the ongoing research on the potential advantages of probiotics, underscoring the importance of considering variables such as duration, probiotic strain, and the specific population in future investigations. Therefore, it is recommended to conduct a confirmatory study with a larger sample size, incorporating additional microbiological and immunological parameters to elucidate the underlying mechanisms responsible for the observed trends and to substantiate potential benefits for at-risk populations.
Author’s contribution: MJF, BRD, AAK: conceptualisation, study design, protocol development, CTRI registration, study conduct, subject recruitment and follow-up, data collection, data analysis, data interpretation, manuscript drafting with critical review and final version approval of the manuscript. RJS: conceptualisation, study design, protocol development, CTRI registration and final version approval of the manuscript.
AGI: Acute gastrointestinal illness; HC: Head circumference; ICU: Intensive care unit; INR: Indian rupees; kg: kilogram; MUAC: Mid upper arm circumference; RTI: Respiratory tract infection
HC: Head circumference; MUAC: Mid upper arm circumference
GI: Gastrointestinal; LRTI: Lower respiratory tract infection