Netarsudil Associated Limbal Follicles: A Case Report
Rinky Thakur1, Lalit Mohan2, Bibhuti Prassan Sinha3
1 Senior Resident, Department of Pharmacology, Indira Gandhi Institute of Medical Sciences, Patna, Bihar, India.
2 Professor and Head, Department of Pharmacology, Indira Gandhi Institute of Medical Sciences, Patna, Bihar, India.
3 Professor and Head, Department of Ophthalmology (R.I.O.), Indira Gandhi Institute of Medical Sciences, Patna, Bihar, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Rinky Thakur, Senior Resident, Department of Pharmacology, Indira Gandhi Institute of Medical Sciences, Patna-800014, Bihar, India.
E-mail: rinkythakur09@gmail.com
Primary Open Angle Glaucoma (POAG) is one of the leading causes of irreversible blindness worldwide. Netarsudil, a Rho-kinase inhibitor and norepinephrine transporter, has recently been approved for reducing Intraocular Pressure (IOP), which is the single most clinically modifiable risk factor for this condition. Here, a case of a 53-year-old female with POAG who developed follicles along the corneal limbus following treatment with Netarsudil eye drops has been reported. The patient had been non-compliant with her POAG treatment for the past five years and presented with diminished vision (right eye greater than left) and raised IOP of 28 mmHg and 26 mmHg in the right and left eyes, respectively. The patient was started on Netarsudil (0.02%) eye drops once daily. After three weeks, the IOP had reduced to 19 mmHg and 18 mmHg in the right and left eyes, respectively; however, the patient still complained of itching and redness. A slit-lamp examination showed few follicular lesions on the limbus. Initial treatment for allergic conjunctivitis was ineffective, leading to a worsening of symptoms. Netarsudil was then discontinued, and dorzolamide was initiated instead, resulting in the resolution of symptoms within one week. The patient continued on dorzolamide without further issues. While Netarsudil is effective in reducing IOP, many of its potential adverse effects may still be unknown due to limited clinical experience. The present case report aims to contribute to the understanding of the safety profile of this drug. More such reports in the future will enhance this knowledge and help clinicians manage similar adverse situations.
Adverse drug reaction, Corneal limbus, Glaucoma, Rho kinase inhibitors
Case Report
A 53-year-old female patient was presented with complaints of diminished vision (right eye >left eye) and mild pain in both eyes, accompanied by a sensation of heaviness, grittiness, and occasional watering, which had been ongoing for five years. Upon taking her history, she revealed that she had been diagnosed with glaucoma approximately five years ago, for which she had been treated with eye drops. However, she had not been very compliant with the treatment. She had completely discontinued her treatment for about six months and did not have any documentation related to her past treatment.
The patient did not give history of any chronic illness; she was non-diabetic and non-hypertensive. There was no significant drug history other than that for glaucoma, and no family history relevant to her current condition. Additionally, there was no known allergy to any drug or substance. Findings of the eye examination were as shown in [Table/Fig-1].
Eye examination findings at first patient visit. These findings were obtained before starting Netarsudil.
| On examination |
|---|
| OD (Right eye) | OS (Left eye) |
|---|
| UCVA | Finger count+ | 6/36 |
| BCVA | 6/36 | 6/18 |
| IOP (mmHg) | 28 | 26 |
| On slit-lamp examination |
| OD (Right eye) | OS (Left eye) |
| Lids | WNL | WNL |
| Conjunctiva | Clear | Clear |
| Cornea | Clear CCT=512 μm | Clear CCT=520 μm |
| Anterior chamber | Deep/Quiet | Deep/Quiet |
| Iris | WNL | WNL |
| Pupil | Sluggish reaction | Round, Regular, Reactive |
| Lens | IMSC | IMSC |
| Fundus | CDR=0.8:1Fovea- dull | CDR=0.6:1Foveal reaction + |
WNL: Within normal limits; UCVA: Uncorrected visual acuity; BCVA: Best corrected visual acuity; CCT: Central corneal thickness; CDR: Cup disc ratio; IMSC: Immature senile cataract
The patient was diagnosed with POAG and was started on Netarsudil 0.02% eye drops once daily. She was followed-up after two weeks, at which point the IOP had reduced to 20 mmHg in the right eye and 17 mmHg in the left eye; however, there was mild redness.
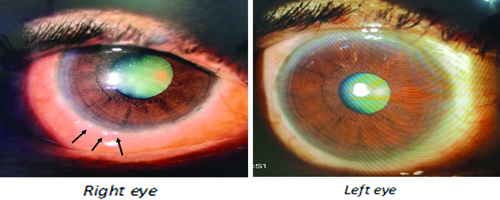
During her next visit one week later, the patient complained of itching and redness in both eyes. The IOP in the right and left eyes was 19 and 18 mmHg, respectively. The right eye slit-lamp examination revealed a few small follicular lesions on the limbus and redness, while the left eye showed only mild redness [Table/Fig-2]. The patient was started on Loteprednol (a corticosteroid) and Carboxymethylcellulose (a lubricant) eye drops, with a provisional diagnosis of allergic conjunctivitis, and was scheduled for follow-up after one week.
Follicular lesions at the corneal limbus of the right eye (arrow) and mild redness in the left eye as seen on slit-lamp examination.
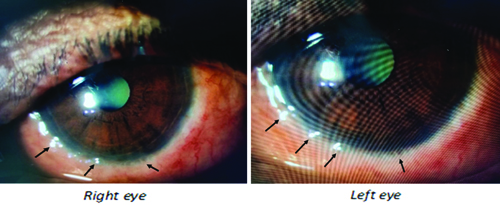
When the patient returned after one week, the redness had increased, and the follicular lesions at the limbus had also increased in size and number, as seen on slit-lamp examination [Table/Fig-3]. Her IOP was 19 and 18 mmHg. It was suspected that the reaction could be due to Netarsudil. Consequently, the drug was withdrawn, and dorzolamide eye drops were started instead. The corticosteroids were tapered, while the lubricant was continued.
Follicular lesions at the limbus (arrow) after one month of treatment with Netarsudil as seen on slit-lamp examination.
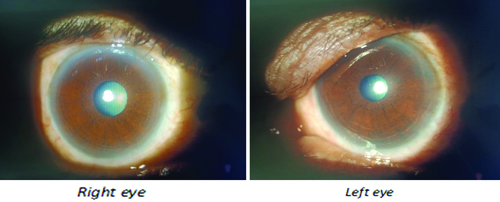
After one week, the patient returned again; the redness had decreased, and the limbal follicular lesions had also disappeared [Table/Fig-4]. The patient had no complaints, and her IOP was 20 mmHg in the right eye and 18 mmHg in the left eye. Therefore, she was continued on dorzolamide. The patient came back for follow-up after one month (while on dorzolamide) and reported no complaints [Table/Fig-5]. The IOP in both eyes was 18 mmHg.
Disappearance of limbal follicular lesions and improvement in conjunctival redness after one week of Netarsudil withdrawal.
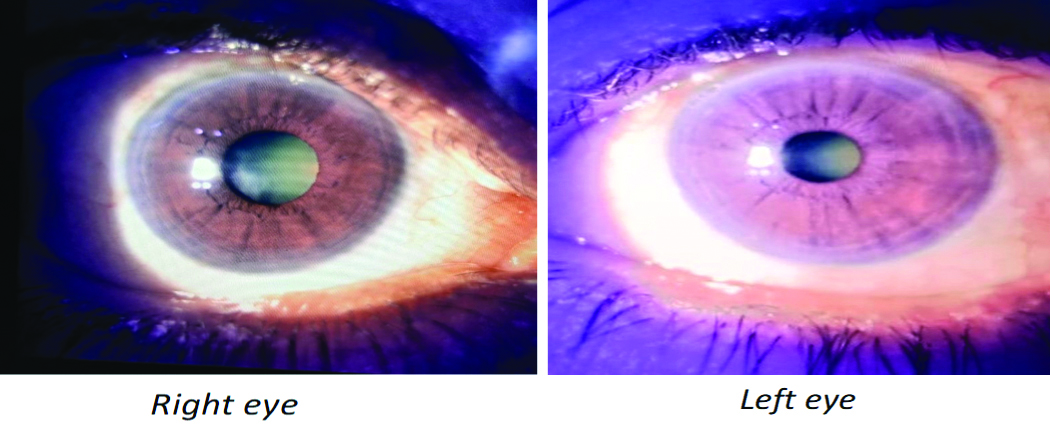
Slit-lamp examination after one month of treatment with dorzolamide.
So, the authors concluded that the Adverse Drug Reaction (ADR) was probably/likely due to Netarsudil, as per the WHO-UMC causality assessment scale [1]. According to the Naranjo Algorithm ADR Probability Scale [2], the total score was 6, indicating that it was probably due to the drug. The reaction was classified as Level 2 (Mild) on the Modified Hartwig and Siegel Severity Assessment Scale [3]. Furthermore, it was deemed not preventable as per the Modified Schumock and Thornton Scale [4].
Discussion
The patient was started on Netarsudil for POAG following which she developed redness and follicular lesions at the limbus that were non-responsive to corticosteroid eye drops. The redness and follicular lesions regressed following the withdrawal of this medication.
Netarsudil has been known to cause corneal hyperaemia and corneal verticillata in the past [5]. It has also been reported to cause corneal oedema in a case report [6], although a case series has shown that ROCK inhibitors could actually facilitate corneal healing and reduce corneal oedema [7]. The drug has also been reported to cause epithelial keratopathy [8] and reticular corneal epithelial oedema [9].
The present case report documents an adverse effect that is different from those reported so far. Although, the patient improved after discontinuing the drug and the implications were not grave, this clinical scenario as an adverse effect should be kept in mind by all practicing ophthalmologists so that they can act promptly if they encounter any such findings with this drug.
The efficacy of Netarsudil has been established in several trials [10,11]. Netarsudil also has an acceptable safety profile, as reported in a pooled analysis that estimated the rate of serious ADRs (both ocular and non-ocular) to be around 3.3% with monotherapy, which was not very different from that of Timolol (~3.2%) [12]. However, as the drug gains popularity among clinicians, its prescription is expected to increase in the near future, and more adverse effects are likely to emerge. We have reported one such case in the present study.
Conclusion(s)
The present case report highlights a potential adverse effect of Netarsudil, which presents as follicles at the corneal limbus. It underscores the importance of regular monitoring and follow-up for patients on this drug. Although, Netarsudil has been found to be an effective and safe drug for POAG, its safety profile continues to evolve with growing clinical experience. Future prescriptions, documentation, and reporting are therefore warranted in order to unravel the potential safety concerns and provide clinicians with the necessary information regarding this drug.
WNL: Within normal limits; UCVA: Uncorrected visual acuity; BCVA: Best corrected visual acuity; CCT: Central corneal thickness; CDR: Cup disc ratio; IMSC: Immature senile cataract
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Jun 15, 2024
Manual Googling: Jul 18, 2024
iThenticate Software: Jul 30, 2024 (3%)
[1]. Center UMThe use of the WHO-UMC system for standardised case causality assessment 2018 Available from: https://www.who-umc.org/media/164200/who-umc-causality-assessment_new-logo.pdf [Google Scholar]
[2]. Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, A method for estimating the probability of adverse drug reactionsClin Pharmacol Ther 1981 30(2):239-45.10.1038/clpt.1981.154 [Google Scholar] [CrossRef]
[3]. Hartwig SC, Siegel J, Schneider PJ, Preventability and severity assessment in reporting adverse drug reactionsAm J Hosp Pharm 1992 49(9):2229-32. [Google Scholar]
[4]. Schumock GT, Thornton JP, Focusing on the preventability of adverse drug reactionsHosp Pharm 1992 27(6):538 [Google Scholar]
[5]. Hoy SM, Netarsudil ophthalmic solution 0.02%: First global approvalDrugs 2018 78(3):389-96. [Google Scholar]
[6]. Chu MJ, Song M, Palmares T, Song A, Song J, Rhopressa-induced corneal edema: A case reportJ Med Case Rep 2021 15(1):182 [Google Scholar]
[7]. Fernandez MM, Reticular epithelial edema in edematous corneas treated with netarsudilOphthalmology 2018 125(11):1709 [Google Scholar]
[8]. Ramakrishnan MS, Addis VM, Lehman AY, Sankar PS, Netarsudil-associated epithelial keratopathyAm J Ophthalmol Case Rep 2020 19:10080010.1016/j.ajoc.2020.100800 [Google Scholar] [CrossRef]
[9]. Tran JA, Jurkunas UV, Yin J, Davies EC, Sola-Del Valle DA, Chen TC, Netarsudil-associated reticular corneal epithelial edemaAm J Ophthalmol Case Rep 2022 25:10128710.1016/j.ajoc.2022.101287 [Google Scholar] [CrossRef]
[10]. Serle JB, Katz LJ, McLaurin E, Heah T, Ramirez-Davis N, Usner DW, Two phase 3 clinical trials comparing the safety and efficacy of netarsudil to timolol in patients with elevated intraocular pressure: Rho Kinase elevated IOP treatment trial 1 and 2 (ROCKET-1 and ROCKET-2)American Journal of Ophthalmology 2018 186:116-27. [Google Scholar]
[11]. Zaman F, Gieser SC, Schwartz GF, Swan C, Williams JM, A multicenter, open-label study of netarsudil for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension in a real-world settingCurr Med Res Opin 2021 37(6):1011-20. [Google Scholar]
[12]. Singh IP, Fechtner RD, Myers JS, Kim T, Usner DW, McKee H, Pooled efficacy and safety profile of netarsudil ophthalmic solution 0.02% in patients with open-angle glaucoma or ocular hypertensionJ Glaucoma 2020 29(10):878-84. [Google Scholar]