Autoimmune Hepatitis (AIH) is a rare autoimmune disease that damages the liver over time and, if left untreated, can progress to liver cirrhosis. Clinical manifestations range from asymptomatic patients to those with acute hepatitis or chronic liver failure. Chronic autoimmune liver disease causes persistent liver injury, which results in unresolved inflammation, proliferation of cells, and the formation of extracellular matrix proteins by stellate cells of the liver and portal myofibroblasts. Cirrhosis of the liver and the resulting loss of normal liver function are unavoidable. Cirrhotic patients have a higher risk of morbidity and mortality, and problems of portal hypertension and/or liver dysfunction result in substantial deterioration in the decompensated phase. The theory suggests that among idiopathic or cryptogenic chronic hepatitis, most of them are autoimmune in origin. It is considered one of the rare liver diseases, especially in the Indian population. Portal hypertension is a dangerous complication caused by an obstruction in portal blood flow, such as cirrhosis or portal vein thrombosis. Hepatic Haemangiomas (HH) are benign liver tumours that are made up of groups of blood-filled cavities lined by endothelial cells and fed by the hepatic artery. The vast majority of HH are asymptomatic and are discovered by chance during imaging studies for unrelated pathologies. Hereby, the authors present a case of 53-year-old female with chronic liver disease with ascites, fatty change, and nodular hepatocellular hyperplasia, which was later found to be cirrhosis of autoimmune origin.
Case Report
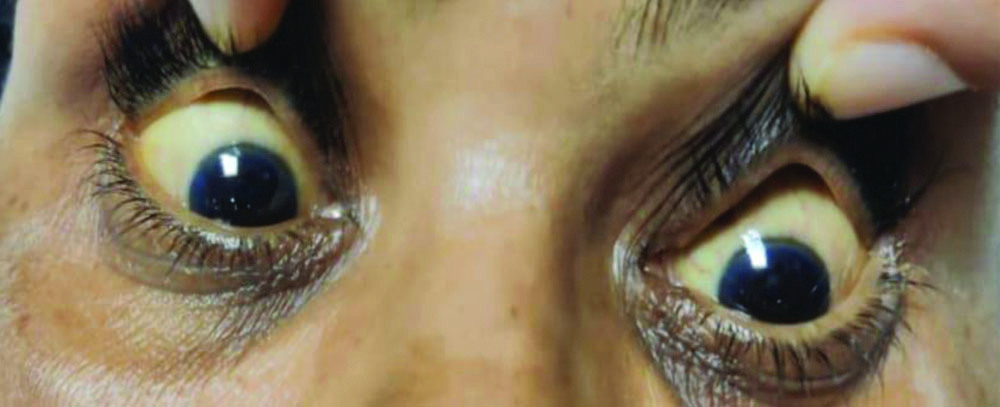
The 53-year-old female presented to the Medicine Department with complaints of yellowish discolouration in the eyes [Table/Fig-1], abdominal distension, and altered behaviour. Relatives noticed mild confusion and disorientation in the 20 days before hospitalisation. There was no significant family history of hepatic disorders. Although there was no history of alcohol intake, the patient had been diagnosed with Type 2 Diabetes Mellitus (T2DM) six years ago and was on 10 units of insulin, as well as hypertension for the past four years and was on captopril 25 mg. The patient had undergone an endoscopy a month prior, revealing mild oesophageal varices and portal hypertensive gastropathy. She had a history of previous hospitalisations for similar complaints. Upon examination, she was afebrile and icteric, with a pulse rate of 92 per minute, blood pressure of 130/90 mmHg, and respiratory rate of 24 per minute. She was drowsy, had skin flaps, and her abdomen was distended. Other system examinations were routine.
Yellowish discolouration in the eyes.
Her laboratory parameters on admission are presented in [Table/Fig-2] below.
Laboratory findings of the patient.
| Laboratory findings | Values at admission | Values at discharge | Reference range |
|---|
| Haemoglobin (g/dL) | 10.4 | - | 13.2-16.6 |
| White blood cell (/mm3) | 13800 | - | 4500-11000 |
| Platelets (/mm3) | 1.67 | - | 150000-450000 |
| MCV (fL) | 80.9 | - | 78.9-98.6 |
| INR | 1.62 | - | 0.8-1.1 |
| PT (sec) | 18.8 | - | 9.4-12.5 |
| APTT (sec) | 41.5 | 29.1 | 25-35 |
| AST (U/L) | 92 | 32 | 13-39 |
| ALT (U/L) | 55 | 33 | 7-52 |
| Alkaline phosphatase (U/L) | 141 | 59 | 34-104 |
| Direct bilirubin (mg/dL) | 3.9 | 0.1 | 0-0.2 |
| Indirect bilirubin (mg/dL) | 1.4 | 0.6 | 0.2-0.8 |
| Total bilirubin (mg/dL) | 5.3 | 0.9 | 0.3-1.0 |
| Total protein (g/dL) | 6.1 | 7.4 | 6.3-8.2 |
| Serum albumin (g/dL) | 2.1 | 4.8 | 3.5-5.0 |
| Serum ammonia (μmol/L) | 171 | 24 | 9-30 |
| Urea (mg/dL) | 99 | 16 | 9- 20 |
| Creatinine (mg/dL) | 3.0 | 1.20 | 0.66- 1.25 |
| Sodium (mmol/L) | 134 | 1.42 | 137-145 |
| Potassium (mmol/L) | 5.4 | 4.5 | 3.5-5.1 |
MCV: Mean corpuscular volume; INR: International normalised ratio; PT: Prothrombin time; APTT: Activated partial thromboplastin time; AST: Aspartate transaminase; ALT: Alanine transaminase
She scored 33 on the Model for End-stage Liver Disease 3.0 (MELD 3.0) and was classified as Child-Pugh class C (score of 12). An ultrasound of the abdomen and pelvis revealed coarse echotexture of the liver, suggestive of cirrhosis of the liver with a focal lesion in segment 4 of the liver, along with gross ascites and grade 1 renal parenchymal disease. Subsequent ultrasonography of the abdomen and pelvis a week later showed gross ascites and partial portal vein thrombosis. The patient was initially administered antibiotics such as Meropenem 2.25 gm thrice a day, Tigecycline 10 mcg once daily, Inj. Albumin 20% at 10 mL/hr., Inj. Mucomix 600 mg twice daily, and other supportive care. The patient’s sensorium was fully restored three days after starting the medication.
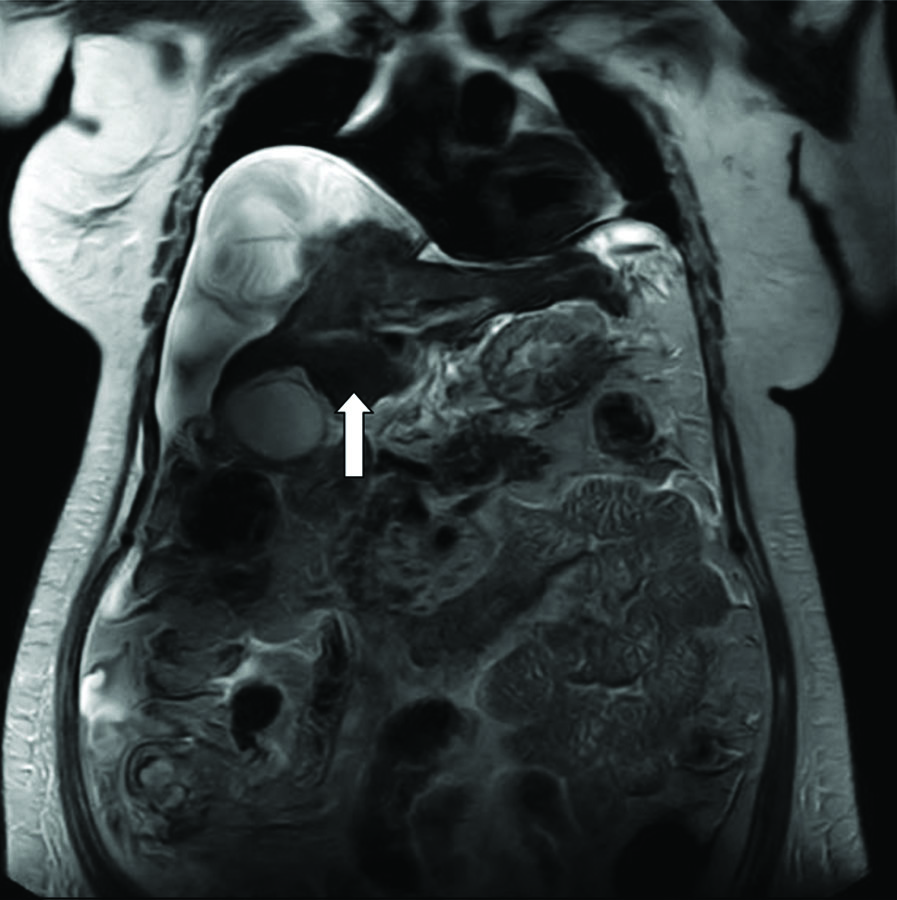
Magnetic Resonance Imaging (MRI)-Magnetic Resonance Cholangiopancreatography (MRCP) suggested chronic liver disease with ascites and revealed T2 hypointense-dependent content in the gall bladder, which may represent concentrated bile, sludge, or microlithiasis. The patient was planned for a triple-phase Computed Tomography (CT) scan, but it could not be done due to raised creatinine [Table/Fig-3]. Blood culture revealed growth of Citrobacter freundii, and urine culture showed growth of E.Coli and Klebsiella pneumonia. Consequently, the patient was started on antibiotics. Although the patient’s sensorium fully recovered, kidney function tests remained deranged in subsequent tests.
MRI-MRCP showing T2 hypointense-dependent content in the gall bladder.
The patient was readmitted for further evaluation of the focal lesion in segment 4 of the liver, as shown in the ultrasound of the abdomen and pelvis, three weeks after the initial visit. An ultrasound-guided Fine Needle Aspiration Cytology (FNAC) of the liver space-occupying lesion was performed, revealing nodular hepatocellular hyperplasia with non specific chronic inflammatory infiltrate and fatty change, but no plasma cell infiltrate or malignant cells were observed. Treatment included azathioprine 50 mg/day and prednisolone 30 mg/day for one week, along with supportive measures such as monitoring liver enzymes, vitamin K, and managing symptoms like insulin regular for diabetes mellitus. Despite abnormal liver and kidney function test results, the patient remained oriented and haemodynamically stable, and was discharged with scheduled follow-up.
Discussion
There are three types of autoimmune hepatitis: types 1, 2, and 3. The majority of autoimmune hepatitis patients have type 1 disease, which is characterised by Antinuclear Antibodies (ANA) and anti-smooth muscle antibodies. Anti-Liver Kidney Microsomal (LKM) type 1 antibody is associated with type 2, which is classified into two subtypes: 2a and 2b. The target antigen in type 2a is liver kidney cytochrome monooxygenase P450 2D6 [1]. Autoimmune hepatitis has an unknown pathophysiological mechanism. According to theory and research, autoimmune hepatitis is a condition that develops in a genetically predisposed individual who is also affected by environmental triggers [2].
In a similar case of autoimmune hepatitis type I by Shrestha M et al., the female patient presented with epigastric pain and swelling, with a history of jaundice and the presence of diabetes mellitus as a co-morbid condition. The symptoms later progressed into liver cirrhosis with lobular inflammation, not associated with portal hypertension, just like in our case. However, the inflammation resulted in the demise of the patient, despite a mild rise in transaminase and high protein levels. Additionally, the patient did not respond to immunosuppressive therapy [3]. In present case, the patient responded well to corticosteroids [2] and showed improvement.
In another case of a woman with recurrent jaundice, abdominal pain, and hepatosplenomegaly, similar to present case, patient also presents with altered behaviour. There is a moderate elevation in levels of transaminase, featuring chronic inflammation of hepatocytes, and the presence of elevated Immunoglobulin G (IgG) confirming the presence of AIH. The patient responded well to steroid therapy, similar to present case [4].
Patients who respond well to corticosteroids often have extra-hepatic immune-mediated illnesses such as diabetes. In type 2b, upto 70% of cases are associated with anti-LKM-1 antibodies. Anti-soluble Liver Antigen (SLA) and anti-liver and Pancreas Antigen (LPA) antibodies are linked to type 3 autoimmune hepatitis. The liver exhibits steatosis and severe septa development. Within two years, the illness typically progresses rapidly to macronodular cirrhosis. While the disease remains inactive during remissions, restoration of standard hepatic architecture does not occur [5].
Autoimmune hepatitis is more common in women and has a bimodal distribution, with peaks in childhood and in the fifth decade. However, AIH affects both genders of all ages, and there have been reports of newly diagnosed AIH in patients as old as 80 years [1]. Upon investigation, hyperbilirubinemia, elevated IgG, markedly increased serum transaminases, and pancytopenia were observed. Liver biopsy remains an essential tool for diagnosis [1].
The mainstay of autoimmune hepatitis management is glucocorticoid therapy. Prednisolone is commonly used and is equally effective. It is initiated at a dose of 60 mg/day, then tapered over a month to 20 mg/day, or started at 30 mg/day along with azathioprine (50 mg/day). With azathioprine maintained at 50 mg/day, prednisolone is tapered over a month to a maintenance dose of 10 mg/day. Combination therapy offers the advantage of reducing complications of steroid therapy. Treatment should be continued for atleast 12-18 months [6,7]. Maintenance therapy with only azathioprine or only prednisolone has also proven beneficial. In medically refractory cases, high doses of combination therapy can be given (prednisolone 30 mg/day+azathioprine 150 mg/day). In severe autoimmune hepatitis, the only reliable treatment is liver transplantation if bilirubin does not improve after two weeks of therapy [8].
Despite being the most effective treatment for all types of liver disease, liver transplantation plays only a minor role in AIH, accounting for roughly 4% of all liver transplants performed in the United States and Europe. Indeed, the cornerstone of AIH management is avoiding liver transplantation through earlier diagnosis and adequate immunosuppressive medication. Patients who are unable to respond to immunosuppressive treatment or who have end-stage liver disease require liver transplantation [9]. Hepatic Haemangiomas (HHs) are primarily asymptomatic and do not require treatment, but cavernous haemangiomas are unpredictable and often followed by severe complications, necessitating regular follow-up and intervention if needed [10]. It should be noted, however, that the correct diagnosis necessitates not only practical skills and appropriate knowledge of the methods to be used, but also clinical expertise and critical judgment in identifying potential false negatives and false positives of the tests used [11].
Cirrhosis develops as a result of chronic liver illness and progresses to compensated, decompensated, and ultimately fatal cirrhosis. Because death in cirrhosis is clearly related to the development of decompensation, identifying predictors of decompensation among compensated patients is warranted [12].
Conclusion(s)
The present case study describes a middle-aged woman who developed autoimmune liver disease, leading to a variety of liver complications. Because the condition is rare and does not regularly present to clinicians, it is critical for healthcare workers to properly diagnose and treat the problem. The disease is potentially treatable if detected early, but it can quickly progress to end-stage liver disease, which is fatal.
MCV: Mean corpuscular volume; INR: International normalised ratio; PT: Prothrombin time; APTT: Activated partial thromboplastin time; AST: Aspartate transaminase; ALT: Alanine transaminase
[1]. Cleveland Clinic (Internet]. (cited 2023 Dec 15]. Autoimmune HepatitisAvailable from: https://my.clevelandclinic.org/departments/digestive/medical-professionals/hepatology/autoimmune-hepatitis [Google Scholar]
[2]. Lohse AW, Mieli-Vergani G, Autoimmune hepatitisJ Hepatol 2011 55(1):171-82.10.1016/j.jhep.2010.12.012 [Google Scholar] [CrossRef]
[3]. Shrestha M, Subedi SC, Shah S, Acharya J, Regmi M, Mehta N, Autoimmune hepatitis leading to liver cirrhosis: A case reportJNMA J Nepal Med Assoc 2022 60(256):1059-62.10.31729/jnma.7808 [Google Scholar] [CrossRef]
[4]. Jemilohun AC, Adewoye OG, Liver cirrhosis from autoimmune hepatitis in a nigerian woman: A case reportAnn Ib Postgrad Med 2017 15(2):133-36. [Google Scholar]
[5]. Homberg JC, Abuaf N, Bernard O, Islam S, Alvarez F, Khalil SH, Chronic active hepatitis associated with antiliver/kidney microsome antibody type 1: A second type of “autoimmune” hepatitisHepatology 1987 7(6):1333-39. [Google Scholar]
[6]. Wies I, Brunner S, Henninger J, Herkel J, Kanzler S, zum Büschenfelde KH, Identification of target antigen for SLA/LP autoantibodies in autoimmune hepatitisLancet 2000 355(9214):1510-15. [Google Scholar]
[7]. Read AE, Sherlock S, Harrison CV, Activejuvenile’cirrhosis considered as part of a systemic disease and the effect of corticosteroid therapyGut 1963 4(4):378-93. [Google Scholar]
[8]. Johnson PJ, McFarlane IG, Williams R, Azathioprine for long-term maintenance of remission in autoimmune hepatitisN Engl J Med 1995 333(15):958-63. [Google Scholar]
[9]. Manns MP, Lohse AW, Vergani D, Autoimmune hepatitis-Update 2015J Hepatol 2015 62(S1):S100-11.10.1016/j.jhep.2015.03.005 [Google Scholar] [CrossRef]
[10]. Bajenaru N, Balaban V, Săvulescu F, Campeanu I, Patrascu T, Hepatic hemangioma-reviewJ Med Life 2015 8(Spec Issue):04-11. [Google Scholar]
[11]. Procopet B, Berzigotti A, Diagnosis of cirrhosis and portal hypertension: Imaging, non-invasive markers of fibrosis and liver biopsyGastroenterology Report 2017 5(2):79-89. [Google Scholar]
[12]. Ripoll C, Groszmann R, Garcia-Tsao G, Grace N, Burroughs A, Planas R, Hepatic venous pressure gradient predicts clinical decompensation in patients with compensated cirrhosisGastroenterology 2007 133(2):481-88. [Google Scholar]