Chronic Lymphocytic Leukaemia Unmasked by COVID-19 Infection: A Case Report
Bhumika Gupta1, Gyanendra Singh2, Vivek Gupta3, Tushar Kalonia4, Shivani Kalhan5
1 Tutor, Department of Pathology, Government Institute of Medical Sciences, Greater Noida, Uttar Pradesh, India.
2 Assistant Professor, Department of Pathology, Government Institute of Medical Sciences, Greater Noida, Uttar Pradesh, India.
3 Associate Professor, Department of Pathology, Government Institute of Medical Sciences, Greater Noida, Uttar Pradesh, India.
4 Assistant Professor, Department of Pathology, Government Institute of Medical Sciences, Greater Noida, Uttar Pradesh, India.
5 Professor and Head, Department of Pathology, Government Institute of Medical Sciences, Greater Noida, Uttar Pradesh, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Bhumika Gupta, Tutor, Department of Pathology, Government Institute of Medical Sciences, Kasna, Greater Noida, Uttar Pradesh, India.
E-mail: drbhumigupta@gmail.com
Coronavirus Disease 2019 (COVID-19) caused by Severe Acute Respiratory Syndrome (SARS) CoV-2 is an Ribonucleic Acid (RNA) virus which spreads through respiratory route. It was first detected in Wuhan, China in December 2019. World Health Organisation (WHO) declared it as worldwide pandemic in March 2020. Clinically, it can present as a mild flu like disease to severe respiratory illness. Reverse Transcription Polymerase Chain Reaction (RT-PCR) is the method for confirmation. Chronic lymphocytic leukaemia is a lymphoproliferative disorder that presents as absolute lymphocytosis with proliferation of monoclonal mature B lymphocytes, unlike other causes of reactive lymphocytosis. Patients with chronic lymphocytic leukaemia often have defects in humoral and cellular immune system. Therefore, these patients have a strong predisposition to recurrent infections and in this case for COVID-19. The relationship between chronic lymphocytic leukaemic and COVID-19 is not very clearly understood. A 77-year-old male with no significant co-morbidities presented with cough, fever and shortness of breath. His High-Resolution Computed Tomography (HRCT) chest revealed non-specific interstitial pneumonia and was tested positive for COVID-19 infection on RT-PCR. A Complete Blood Count (CBC) showed a high Total Leukocyte Count (TLC) with absolute lymphocytosis. There was absence of lymphadenopathy or splenomegaly. Flow cytometry confirmed the diagnosis of Chronic lymphocytic leukaemia. He was offered convalescent plasma therapy and later had to be intubated because of multiorgan failure. His Absolute Lymphocyte Counts (ALC) more than doubled in a week’s time. COVID-19 infection usually presents with lymphopenia, lymphocytosis should always raise a suspicion of underlying haematological malignancy or secondary infection.
Corona virus disease 2019, Lymphocytosis, Monoclonality
Case Report
A 77-year-old man presented to the Flu Out Patient Department (OPD) with complaints of fever, cough and shortness of breath since two days. At the time of admission his SpO2 was 90% on 15 litre/minute oxygen via non re-breather mask. He had no complaints of weight loss, loss of appetite or night sweating. There were no co-morbidities or significant past medical history or family history. On physical examination there was no lymphadenopathy or organomegaly. HRCT chest showed patchy opacities involving bilateral mid and lower lobes suggestive of non-specific interstitial pneumonia. He tested positive for COVID-19 by RT-PCR from nasopharyngeal and throat swabs. He was shifted to Non-Invasive Ventilation (NIV) mode with FiO2 100% and started on Ceftriaxone 1 gm i.v 12 hourly, Methylprednisolone 40 mg i.v 12 hourly, Clexane 60 mg s/c 12 hourly, Hydroxychloroquine 400 mg BD and other symptomatic treatment.
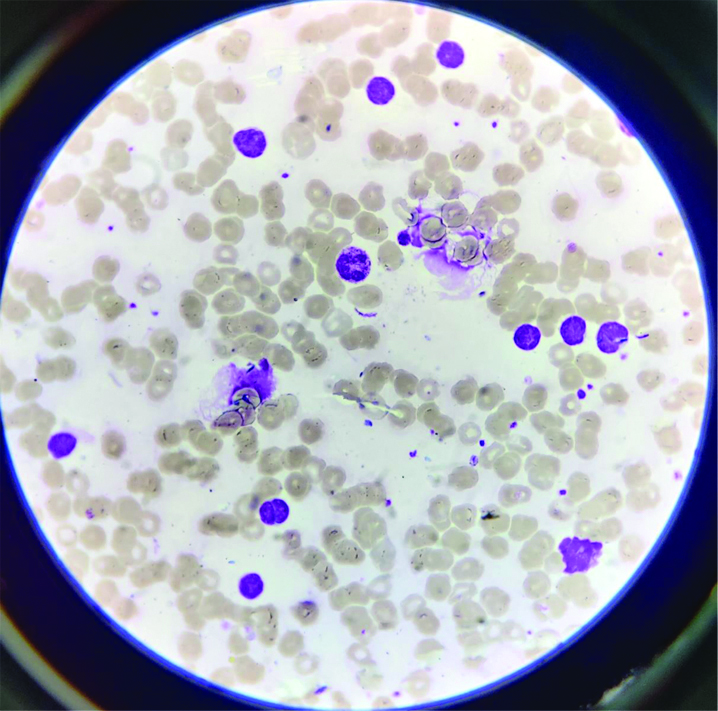
His CBC showed a TLC of 27,600 cells/mm3. The ALC was abnormally high, 16,560 cells/mm3 [Table/Fig-1]. The peripheral blood smear showed marked lymphocytosis consisting mostly of small mature-looking lymphocytes with few smudge cells [Table/Fig-2]. Platelets were within normal limits. Flow cytometry revealed an atypical population of B-lymphoid cells showing lambda light chain restriction and co-expression of CD5 and CD19 with dim to moderate expression of CD20, CD23, sIgM and CD43. There was moderate to bright expression of CD45 and CD200. These atypical cells were negative for CD10, CD11c, CD25, CD38, CD79b, FMC7, CD103 and CD123. Flow cytometry hence confirmed the diagnosis of CLL. In the absence of lymphadenopathy and splenomegaly, he was staged as Rai stage 0 and Binet stage A [1,2]. His C-Reactive Protein (CRP) was 224.3 mg/L, D-dimer was 9.61 mg/L, and serum Lactate Dehydrogenase (LDH) was 732.6 U/L.
Changes in laboratory values during hospitalisation.
| Test | Day |
|---|
| 1 | 2 | 3 | 4 | 5 | 6 |
|---|
| TLC (Cells/mm3) | 27600 | 37700 | 52100 | 60700 | 81300 | 44300 |
| ALC (Cells/mm3) | 16560 | 19981 | 28655 | 30957 | 40650 | 19049 |
| Haemoglobin (g/dL) | 11.9 | 11.5 | 11.6 | 12.3 | 12.9 | 11.7 |
| Platelet count (lac/mm3) | 3.83 | 2.86 | 1.67 | 1.12 | 1.66 | 1.64 |
| Serum urea (mg/dL) | 32 | 39 | 85.4 | 127 | 212 | 253 |
| Creatinine (mg/dL) | 0.9 | 1.26 | 1.52 | 1.69 | 3.64 | 2.93 |
| Uric acid (mg/dL) | 1.2 | 3.0 | 7.0 | 8.7 | 13.4 | 14.6 |
ALC: Absolute leukocyte count; TLC: Total leukocyte count
Peripheral smear showing mature lymphocytes with smudge cells (Leishman’s stain, 400x).
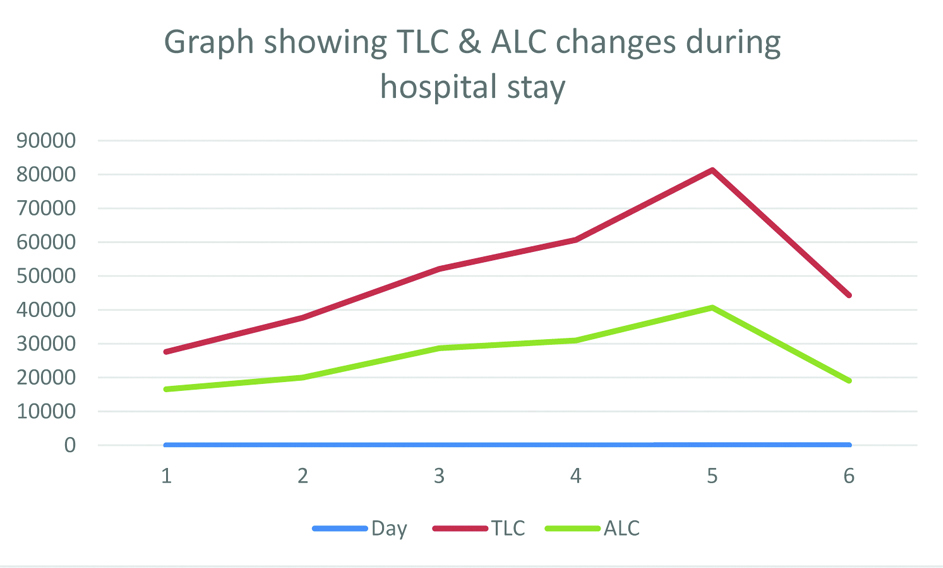
The following day his chest X-ray worsened and showed increase in patchy opacities present in bilateral mid and lower lung zones. His antibiotics were switched to Tazomac 4.5 gm intravenous (i.v) 8 hourly and Clindamycin 600 mg i.v 8 hourly. He received daily dose of Methylprednisolone 40 mg i.v 12 hourly throughout the duration of his hospital stay, but TLC and ALC were unaffected. TLC kept on increasing from 52,100 cells/mm3 on day three to 81,300 cells/mm3 on day 5th. ALC was almost more than doubled from 16560 cells/mm3 on day 1 to 40650 cells/mm3 on day fifth [Table/Fig-1,3]. Patient was also offered convalescent plasma therapy but despite all measures, patients saturation couldn’t be maintained, he was intubated on day 5 and was started on i.v Noradrenaline to maintain his falling systolic blood pressure to 60 mmHg. In addition, he developed acute kidney injury [Table/Fig-1] and multiple organ failure.
Graph showing Total Leukocyte Count (TLC) and Absolute Lymphocyte Count (ALC) changes during hospitalisation.
On seventh day of admission his oxygen saturation dropped to 70% and he developed an asystole, his pulse and blood pressure was not recordable. Cardiopulmonary Resuscitation (CPR) was started as per Advanced Cardiac Life Support (ACLS) protocol and vasopressors were given, but despite all these measures, the patient could not be revived.
Discussion
COVID-19 disease mediated by Novel coronavirus SARS-CoV-2 was first detected in Wuhan, China, in December 2019. By the end of March 2020, the infection was declared a worldwide pandemic [3]. The clinical presentation varies from mild flu like disease to severe respiratory illness. Severe and sometimes fatal illness with respiratory co-morbidites are often associated with bilateral ground-glass opacities on chest imaging, occurs mainly in older individuals and those with comorbidities such as diabetes, COPD, and cardiac failure [4]. Patients with haematologic malignancies, especially those with lymphocytic leukaemia, have a high risk of infection owing to the underlying immunodeficiency and inadequate immune response to infection [5].
Chronic lymphocytic leukaemia is a lymphoproliferative neoplasm that is characterised by clonal proliferation of mature B cells. The diagnosis is based on presence of at least >5×109/L B lymphocytes in peripheral blood, sustained for at least 3 months. The clonality of the mature B lymphocytes needs to be confirmed using flow cytometry by demonstrating an immunoglobulin light chain restriction [6]. Reactive lymphocytosis is the main differential diagnosis for CLL which can be seen in infections or other types of lymphoid neoplasms. However, unlike CLL, lymphopenia is a more common finding in patients with COVID-19 than lymphocytosis [7], making the diagnosis of COVID-19 more challenging especially if the patient is not previously diagnosed with CLL [8].
Most of the CLL patients are asymptomatic and are often diagnosed during routine work up or underevaluation of enlarged lymph nodes [6]. The index patient also had no previous record of lymphocytosis or any lymph node enlargement.
The elderly and those with underlying co-morbidities are more susceptible to the COVID-19 disease. There are only few cases reported in the literature of CLL patients with COVID-19, but it can have a major impact on their illness [8]. CLL patients often have defects in humoral and cellular immune system, with a strong predisposition to recurrent infections, especially bacterial infections. T-cells, which are responsible for the immune response against viral infection, are depleted in patients with CLL, in particular if they have received chemotherapy [9]. The index patient didn’t receive any chemotherapy and that surely had major impact on the course of the disease.
In a report by Paneesha S et al., they documented four patients with untreated CLL who contracted SARS-CoV-2 infection confirmed by PCR of sample collected by throat swab at the time of admission [10]. They followed severe clinical course of the disease with a high rate of mortality. An increase in the lymphocyte count was observed in all four cases by 3-4 times of the baseline values. They suggested COVID-19 infection with therapy-naive CLL may be associated with considerably greater clinical risk. In this case, similar pattern was observed, the ALCs almost more than doubled in a short span of a weeks time [Table/Fig-3].
Safarpour D et al., also reported a case, where an 80-year-old patient was diagnosed with severe COVID-19 infection and CLL [5]. During hospitalisation his ALCs were increased by more than three folds from his baseline counts. The patient was not offered any chemotherapy. The patient couldn’t survive as he developed Multiple Organ Dysfunction Syndrome (MODS), which was very similar to this case. This indicates that the COVID-19 infection might be associated with an increase in the clonality of the B cells. As no previous records of absolute lymphocytosis were available, but there is a possibility of unreported monoclonal B-cell lymphocytosis that got accelerated into CLL by the COVID-19 infection as seen in other respiratory tract infection [11]. Another possibility could be asymptomatic CLL that was unmasked by the COVID-19 infection.
Conclusion(s)
Lymphocytosis is usually an uncommon finding with COVID-19 infection and its presence should always raise the suspicion of an additional underlying disease or haematological malignancy such as CLL. The interaction between COVID-19 infection and CLL is not very clearly understood. More reported cases and close follow ups are needed to bridge the gaps in the existing knowledge.
ALC: Absolute leukocyte count; TLC: Total leukocyte count
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. NA
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Feb 01, 2021
Manual Googling: Mar 20, 2021
iThenticate Software: May 31, 2021 (16%)
[1]. Rai KR, Sawitsky A, Cronkite EP, Chanana AD, Levy RN, Pasternack BS, Clinical staging of chronic lymphocytic leukemiaBlood 1975 46(2):219-34.10.1182/blood.V46.2.219.2191139039 [Google Scholar] [CrossRef] [PubMed]
[2]. Binet JL, Leporrier M, Dighiero G, Charron D, D’Athis P, Vaugier G, Clinical staging system for chronic lymphocytic leukemiaCancer 1977 40:855-64.10.1002/1097-0142(197708)40:2<855::AID-CNCR2820400239>3.0.CO;2-1 [Google Scholar] [CrossRef]
[3]. Cucinotta D, Vanelli M, WHO Declares COVID-19 a PandemicActa Biomed 2020 91(1):157-60. [Google Scholar]
[4]. Kumar V, Abbas AK, Aster JC, Perkins JA, Robbins Basic Pathology (Tenth edition.) 2018 PhiladelphiaElsevier [Google Scholar]
[5]. Safarpour D, Srinivasan K, Farooqui M, Roth C, Ghouse M, A case of COVID-19-induced lymphocytosis in a patient with treatment-naive CLL: Should it be treated?Clin Lymphoma Myeloma Leuk 2020 S2152-2650(20):30516-4. [Google Scholar]
[6]. Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Döhner H, iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLLBlood 2018 131(25):2745-60.10.1182/blood-2017-09-80639829540348 [Google Scholar] [CrossRef] [PubMed]
[7]. Sun S, Cai X, Wang H, He G, Lin Y, Lu B, Abnormalities of peripheral blood system in patients with COVID-19 in Wenzhou, ChinaClin Chim Acta 2020 507:174-80.10.1016/j.cca.2020.04.02432339487 [Google Scholar] [CrossRef] [PubMed]
[8]. Jin XH, Zheng KI, Pan KH, Xie YP, Zheng MH, COVID-19 in a patient with chronic lympho-cytic leukaemiaLancet Haematol 2020 7:e351-52.10.1016/S2352-3026(20)30074-0 [Google Scholar] [CrossRef]
[9]. Melchardt T, Weiss L, Greil R, Egle A, Viral infections and their management in patients with chronic lymphocytic leukemiaLeuk Lymphoma 2013 54(8):1602-13.10.3109/10428194.2012.75517823206225 [Google Scholar] [CrossRef] [PubMed]
[10]. Paneesha S, Pratt G, Parry H, Moss P, Covid-19 infection in therapy-naive patients with B-cell chronic lymphocytic leukemiaLeuk Res 2020 93:10636610.1016/j.leukres.2020.10636632388230 [Google Scholar] [CrossRef] [PubMed]
[11]. Anderson LA, Landgren O, Engels EA, Common community acquired infections and subsequent risk of chronic lymphocytic leukaemiaBr J Haematol 2009 147(4):444-49.10.1111/j.1365-2141.2009.07849.x19725826 [Google Scholar] [CrossRef] [PubMed]