Endometriosis, a chronic disease characterised by the presence of endometrial tissue (glands and stroma) outside the uterus causes significant interference of the quality of life especially in women of reproductive age. Although, it has been extensively investigated over the past century, it still remains an enigmatic disease [1].
Effects of the disease on physical and mental health is debilitating because misdiagnosis are common and there is a time delay of six years or more between the onset of symptoms to a confirmed diagnosis [2]. The presentation and course of the disease are varied in that in some patients it can persist as a minimal or mild disease, or the disease can be asymptomatic. Remaining cases can present with severe pain or infertility due to tissue infiltration, growth of endometriomas or pelvic adhesions.
Visual inspection of endometriotic lesions by laparoscopy preferably with histological confirmation is the gold standard for diagnosis of endometriosis [3]. Delay in diagnosing endometriosis results in worsening of quality of life of the patient, advancement of the disease and increases the financial burden. Further, laparoscopy a surgical procedure can be associated with few complications. Due to non-availability of good non-invasive or semi-invasive test for diagnosing endometriosis, there is a substantial time delay between onset of pain symptoms and confirmed endometriosis. Development of a simple non-invasive or semi-invasive diagnostic method can greatly help to reduce this delay and prevent severe morbidity in women with minimal to mild endometriosis who cannot be diagnosed clinically or by ultrasonography [4].
Eutopic endometrium in women with endometriosis is found to be biologically different from endometrium in women with no endometriosis. Higher density of small unmyelinated fibres is present in the functional layer of endometrium In women with endometriosis, higher density of small unmyelinated fibres is present in the functional layer of endometrium when compared to women without endometriosis [4]. According to implantation theory, the nerve fibres in endometriotic plaques arise either from nerve fibre precursors from the functional layer of the endometrium or due to ingrowth of local nerve fibres as a result of secretion of Nerve Growth Factors (NGFs) and increased expression of NGF receptors, namely Trk-A and p75 by the implanting endometrium [5].
The aim of the study was to evaluate the role of nerve fibre detection in endometrial biopsy for diagnosing endometriosis.
Materials and Methods
This case-control study was conducted in SRM Medical College Hospital and Research Centre from March 2012 to August 2013. SRM Medical College Hospital and Research Centre, Chengelpet, Tamil Nadu, India, Institutional Ethical committee approval was obtained (239/IEC/2012) and the study was conducted in accordance with the Declaration of Helsinki as revised in 2013.
Inclusion and Exclusion criteria: Women undergoing laparoscopy/laparotomy for pelvic pain or infertility between 18-45 years were included in the study after informed written consent. Patients receiving hormonal therapy three months prior to laparoscopy/laparotomy and not willing to participate in the study were excluded. Detailed clinical history was taken from all participants prior to the procedure.
Study Procedure
The reference standard used for diagnosing endometriosis was by laparoscopic/laparotomy visualisation of the endometriotic lesions and staging of the disease was done using Revised American Society Scoring System [3]. Cases included patients with laparoscopic/laparotomy visualisation of endometriotic lesions and controls included patients without visual evidence of endometriosis. Endometrial biopsies were taken at the time of laparoscopy/laparotomy using standard metallic endometrial curette. In patients who had undergone hysterectomy for endometriosis, endometrial biopsy was taken from the hysterectomy specimen. The quality of the endometrial biopsy was assessed before nerve fibre counting histologically. All samples were stained with Haematoxylin and Eosin for conventional histological assessment. Polyclonal rabbit Anti-protein gene product 9.5 (Dako, Australia), highly specific pan neuronal marker which recognises all types of nerve fibres was used for Immunohistochemistry.
Immunohistochemistry technique: The endometrial biopsies were fixed in 10% buffered formalin and processed routinely to paraffin wax. Cut-sections were taken on coated slides and deparaffinised in xylene then rehydrated through graded alcohol and washed under running tap water. Antigen retrieval was performed using 0.01 m citrate buffer and was washed in deionised water. Peroxidase block was used to neutralise endogenous peroxidase and then washed in Tris Buffered Saline (TBS) and slides were incubated with protein block for five minutes, then washed in TBS and incubated with optimally diluted primary antibody for one hour and washed with TBS, and then were incubated with postprimary antibody for 30 minutes and washed with TBS, then slides were again incubated with Novolink polymer for 30 minutes and washed with TBS. Peroxidase activity was developed using DAB working solution for 5 minutes. Slides were rinsed in water and counterstained with haematoxylin and rinsed again with water. Clear, dehydrate slides are seen using microscope.
The slides were viewed by a single pathologist, who was uninformed of the intraoperative findings . The specimen was graded as scanty or satisfactory based on the amount and endometrial mucosal orientation. Specimen was considered satisfactory if well-oriented endometrial mucosa was present in more than one low power field. The PGP stained slides were scanned at 20 and 40 objective magnification using a Leica light microscope. The cases were classified as positive or negative based on the presence or absence of neuronal elements and not on the number of neuronal elements identified.
Statistical Analysis
Association with clinical presentation, hormonal therapy, endometrial cyclical changes and stage of endometriosis was done using Chi-square test with statistical tools from graph pad instat 3 software. Statistical significance was considered if p<0.05.
Results
During the 18 months study period, 64 endometrial biopsies were obtained from women undergoing laparoscopy/laparotomy and three cases were excluded due to poor quality of endometrial biopsy. Women with visual evidence of endometriosis in laparoscopy/laparotomy were taken as cases (n=32). Women without evidence of endometriosis in laparoscopy/laparotomy were taken as controls (n=29).
Majority of patient with endometriosis has undergone laparotomy. Pain symptoms alone was the indication for surgery in 21 (65.6%) in endometriosis group compared to 7 (24.1%) in control group which was found to be significant (p=0.001). Among 32 patients with endometriosis eight had endometrioma [Table/Fig-1].
Demographic characteristics.
| Categories | Cases (32) | Control (29) | p-value |
|---|
| Age (y), Mean±SD (range) | 36.25±7.02 (23-47) | 34.03±9.16 (20-53) | 0.29 |
| Parity |
| Nullipara n (%) | 12 (37.5) | 16 (55.1) | 0.26* |
| Multiparous n (%) | 20 (62.5) | 13 (44.9) |
| Visualisation of endometriotic lesions |
| Laparotomy n (%) | 21 (65.62) | 13 (44.8) | 0.04* |
| Laparoscopy n (%) | 11 (34.38) | 16 (55.1) |
| Previous hormonal therapy | 5 | 4 | 0.13* |
| Symptoms |
| Infertility n (%) | 4 (12.5) | 10 (34.4) | 0.17 |
| Pelvic pain n (%) | 21 (65.6) | 7 (24.1) | 0.001* |
| Infertility+Pelvic pain n (%) | 7 (21.9) | 12 (41.3) | 0.87 |
*Chi-square test; Y: Years
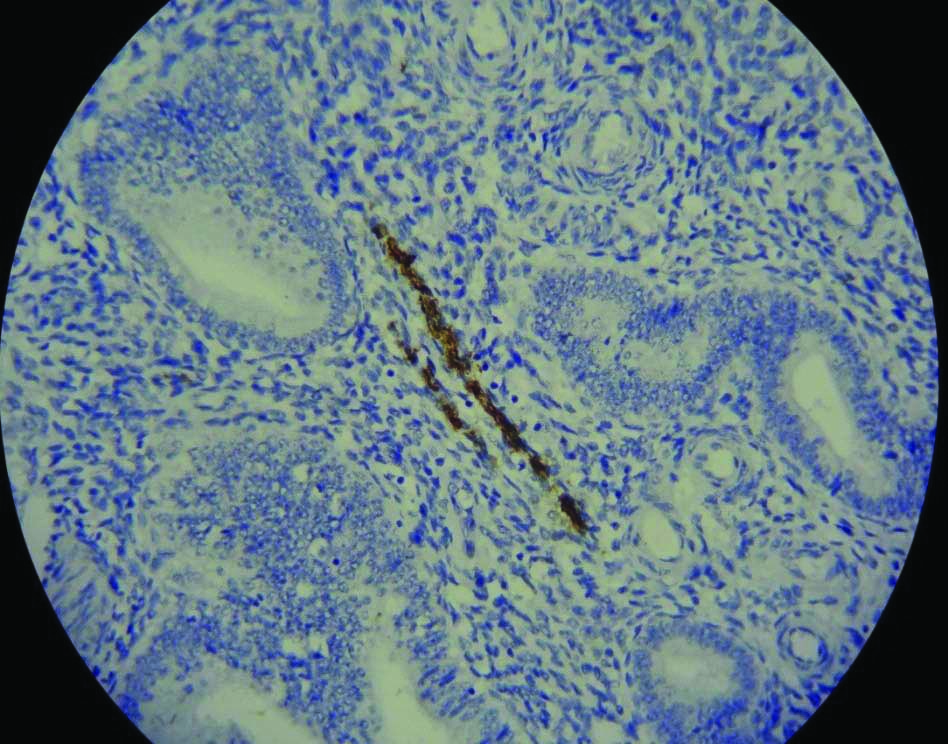
Endometrial nerve fibres were detected in the functional layer in 7/32 (21.8%) of the endometriosis patients, image shown in [Table/Fig-2]. No nerve fibres were detected in the control group. This association of endometrial nerve fibres in women with endometriosis was found to be statistically significant (p=0.01, OR=2.16, 95% CI (1.62-2.87) [Table/Fig-3].
Presence of nerve fibres in endometrium; (10X, Immunohistochemistry staining).
Endometrial nerve fibres in cases and controls.
| Endometrial nerve fibres | Cases (n=32) | Controls (n=29) | p-value |
|---|
| Present | 7 (21.8%) | 0 | 0.01OR=2.16*95% CI (1.62-2.87) |
| Absent | 25 (78.1%) | 29 (100%) |
*Chi-square test
Among the seven patients with positive endometrial nerve fibres, six had pain as the predominant symptom. But, there was no significant correlation between the presence of endometrial nerve fibres in endometriosis with pain symptomatology, history of hormonal therapy, endometrial cyclic changes and stages of endometriosis [Table/Fig-4]. Among the seven patients, five patients had endometriotic cyst which was confirmed by histopathology. Remaining two were diagnosed by visualisation of endometriotic lesions.
Association between endometrial nerve fibre detection with pain symptoms, Hormonal therapy, biopsy adequacy, phase of menstrual cycle and stage of endometriosis.
| Variables | Functional layer Nerve Fibres (FNF) present n=7 | Functional layer Nerve Fibres (FNF) absent n=25 | p-value |
|---|
| Symptoms | p=0.92 |
| Pain | 6 | 21 | OR=1.11 |
| No pain | 1 | 4 | 95% CI (0.19-7.35) |
| Hormonal therapy | p=0.48 |
| Present | 0 | 20 | OR=4.02 |
| Absent | 7 | 5 | 95% CI (0.19-81.98) |
| Biopsy adequacy | p=0.87 |
| Satisfactory | 6 | 22 | OR=0.81 |
| Scanty | 1 | 3 | 95% CI (0.07-9.36) |
| Endometrium | p=0.95 |
| Proliferative | 4 | 14 | OR=1.048 |
| Secretory | 3 | 11 | 95% CI (0.19-5.69) |
| Stage of endometriosis | p=0.41 |
| 1 and 2 | 1 | 10 | OR=0.25 |
| 3 and 4 | 6 | 15 | 95% CI (0.025-2.40) |
*Chi-square test
Sensitivity of detecting endometriosis by the presence of nerve fibres in endometrial nerve biopsy specimen was 21.8%, Specificity was 100%, PPV was 100% and NPV was 46.29%.
Discussion
The gold standard for the diagnosis of endometriosis is direct visualisation of endometriotic lesions through laparoscopy preferably with confirmation by histology [3]. But being an invasive procedure it is associated with small risk of surgical complications. Because of non-availability of good non-invasive or semi-invasive test for diagnosis of endometriosis there is a substantial time delay between onset of pain symptoms and surgically confirmed endometriosis. The pathogenesis of endometriosis is not well understood. Eutopic endometrium in women with endometriosis is found to be biologically different from endometrium in women with normal pelvis. There are various molecular and cellular abnormalities in the eutopic endometrium of women with endometriosis which may play a part in the development of endometriosis and its related symptoms [6].
Tokushige N et al., reported the new finding of presence of small unmyelinated sensory C nerve fibres in the functional layer of endometrium in all women with endometriosis. In contrast, there was no nerve fibres in the functional layer in women without endometriosis (p<0.001) [7]. Significantly higher density of nerve fibres in the basal endometrium and in the myometrium was seen in women with endometriosis when compared to women without endometriosis (p<0.001) [8]. They also concluded that these increased nerve fibre density, sensory C fibres and adrenergic nerve fibres in the endometrium may play a key role in pain generation in endometriosis [7]. In this study, 6 out of 7 patients who had FNF had pain symptoms.
Although Tokushige N et al., has shown a higher prevalence of endometriosis in secretory phase, in 2009 Bokor A et al., found no difference in nerve fibre density in relation to the phase of the menstrual cycle similar to our study [7,8].
Hormonal therapy, one of the modalities to treat endometriosis was found to decrease nerve fibre density. It has been reported that these endometrial nerve fibres are not detected in most women on hormonal therapy [9]. Small C-nerve fibres were uniquely present only in the functional layer of endometrium in women with endometriosis and was not present in the functional layer in women with other uterine pathologies like adenomyosis, endometrial hyperplasia, chronic endometritis, leiomyomas and endometrial polyps [10]. Leslie C et al. who performed conventional endometrial biopsies for 68 patients who underwent laparoscopy for suspected endometriosis found that functional layer nerve fibres were detected in 15 (22%) endometrial biopsies including 9/47 with peritoneal endometriosis and 6/21 (29%) without endometriosis. There was no correlation between functional layer nerve fibres and clinical symptoms, phase of endometrial cycle, or hormonal therapy similar to our study [11].
In women with laparoscopic diagnosis of endometriosis, Al-Jefout M et al., found that 2.7 nerve fibres per mm2 was the mean nerve fibre density in the functional layer of the endometrium . Nerve fibre density was significantly more in women with pain symptoms when compared to women with infertility but no pain symptoms were found (2.3 and 0.8 nerve fibre per mm2, respectively, p=0.005) [10].
Bokor A et al., found there was no uniform distribution of these nerve fibres throughout the endometrium. The density of nerve fibres was markedly varied ranging from 0 to 30/mm2. There was no significant difference in nerve fibre densities in women with minimal endometriosis (2.1+2.87/mm2) and mild endometriosis (1.84+2.59/mm2, p=0.46) [8].
In contrast to others, Donnez O et al., concluded that endometrial biopsies were mostly negative for nerve fibres. Instead nerve fibres were detected in endometriotic lesions and their density was more in deep nodular lesions when compared to peritoneal (p<0.01) or ovarian (p<0.001) lesions. NGF expression was also more in the stroma of deep-infiltrating lesions (p<0.05). Their conclusion was deep nodular lesions are more neuroattractive because of NGF [12].
The specificity and sensitivity of diagnosis of endometriosis for detection of nerve fibres in endometrial biopsy in endometriosis was 83 and and 98%, respectively in the study by Al-jefout M et al., [10]. Bokor A et al., concluded that minimal-mild endometriosis can be predicted by the neural markers PGP9.5, Vasoactive Intestinal Peptide (VIP) and anti-Substance P (SP) with 95% sensitivity and 100% specificity [8]. In contrast in our study, the specificity and sensitivity was 100% and 21.8%, respectively.
In our current study, presence of FNF correlated poorly with the diagnosis of endometriosis by laparoscopy/laparotomy. Many factors including procedural factors may have resulted in these discordant findings. Al-Jefout M et al., in his study used a specific endometrial sampling device to get full thickness endometrial specimens [10]. In contrast, the curettage technique used in our study and routinely used in practice may provide only small tissue fragments that may not be specifically oriented for microscopic examination.
In the study by Bokor A et al., endometriosis were confirmed by both laparoscopy and histopathology whereas in our study histological confirmation for all could not be possible as many patients had undergone only diagnostic laparoscopy. The basal and functional layers of the endometrium are not sharply defined histologically. This may limit the use of endometrial nerve fibre analysis in routinely obtained biopsy specimens [8].
Another difference between our study and the previous studies are that the nerve fibres were assessed using conventional light microscopy. Photomicrography and image analysis are more sensitive for nerve fibre detection.
Limitation(s)
The most important limitation of endometrial FNF analysis in this study is low sensitivity 21.8%.
Conclusion(s)
Endometrial nerve fibre assessment for identifying endometriosis in women undergoing laparoscopy/laparotomy using routine endometrial sampling and immunohistochemical analysis was not found to be sensitive for the diagnosis of endometriosis. These findings were influenced due to a number of factors and cannot be recommended as an alternative to laparoscopy. In future large prospective studies with improved methodology may help to find whether endometrial nerve fibre assessment will help in identifying endometriosis in early stage so that treatment can be started early.
*Chi-square test; Y: Years
*Chi-square test
*Chi-square test