Everyday life activities get limited in patients with RA because of severity of pain, fatigue, energy loss, joint deformities etc., [1]. Deformities of small joint of hands are commonly found approximately 80 to 90% in RA [2]. Grip strength and muscle forces are influenced due to muscle atrophy and nodular myositis presence in RA [3]. Muscle changes occur in RA due to direct involvement of neuromuscular system and can be due to pathological process occurring in muscle leading to reduction in contractile ability [4].
Muscle contraction is important for various activities of hand which involves grip force [5]. Grip force and extension force decrease in early RA as compared to healthy individuals due to involvement of metacarpophalangeal joints [6]. Sleep disturbance is also pronounced along with symptoms of pain, fatigue and physical disability, which hamper quality of life in chronic RA [7].
Physical agents such as ice and wax packs and exercise therapy help in pain management, joint tenderness and Activities of Daily Living (ADLs) in RA patients [8]. Strengthening and stretching exercise of hand and wrist improves the dexterity and functions of hands [9]. Fitness exercise and other physical activity have a beneficial effect in chronically ill individuals [10]. Despite conventional therapy, patient with RA have some degree of disability due to pain, synovitis and joint damage. The treatment which is generally given does not prevent or cure the disease [11]. The aim of this study was to find the effectiveness of hand strengthening exercise by use of therapeutic putty on grip strength, key pinch strength, pain severity and hand function in chronic rheumatoid patients.
Materials and Methods
This case-control study was carried out at Pt BDS PGIMS Rohtak. The study was approved by the Ethical and Research Committe of the Institute (vide letter No: IEC/17/387 dated 25.03.17). The data collection was done from July 2016 to June 2019.
Sample size calculation: The sample size was calculated by a Power Analysis Software System (PASS), using the power of study 0.95, effect size was 0.75 and probability error 0.05.
The prevalence of RA is more among females as compared to males [12].
Inclusion criteria: A total of 206 female subjects clinically diagnosed RA (as per American college of Rheumatology 1987 [13]) between the age group of 20-50 years.
Exclusion criteria: Patients on disease modifying anti-rheumatic drugs for more than 2 years and patients with ability to fully extend fingers. Patients with severe anaemia, hypothyroidism, having evidence of renal, cardiac, liver or pulmonary disease, cerebrovascular abnormalities, persistant neurological deficit and recent fractures and fixed musculoskeletal deformities (as per history taken from patients) were excluded from the study.
A written consent was obtained from all subjects. The subjects were randomally divided in to two groups by simple random sampling method: Control group (Group A) and Experimental group (Group B). The exercise protocol was implemented for 12 weeks. Escape treatment, in the form of Paracetamol (PCM), was provided if participants complained of pain after exercise. Subjects were advised not to use PCM 24 hours prior to assessment/reading, so that performance of muscle was not affected.
Protocol for Control Group (N=103)
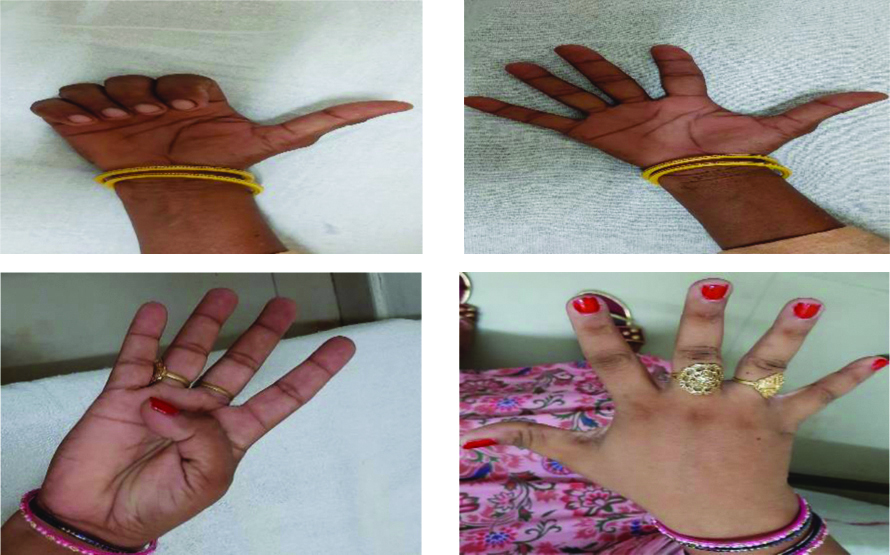
The protocol inlcuded active hand exercise with hot water fomentation (for 20 min). Subjects were detailed about the exercise protocol and they were asked to perform exercise at least 5 times a week with each exercise repeated 10 times, with 10-15 repetition in a progressive manner. The exercises were performed in sitting position with shoulder in neutral position, elbow flexed to 90 degree and forearm resting on arm rest of chair with hands free from any support. Active exercise included finger fist, thumb opposition, ok sign, fingers abduction exercise and tip to tip pinch exercise [14] [Table/Fig-1].
Active hand exercise (for control group).
Protocol for Experimental Group (N=103)
The experimental group was detailed about the strengthening exercise protocol with hot water fomentation (for 20 min). They were asked to perform exercise at least 5 times a week with each of the task repeated 10 times with the position of maximum effort held for 3-5 second with a 20 seconds rest between the repetitions, with 10-15 repetition in a progressive manner. The exercises were performed in sitting position with shoulder in neutral position, elbow flexed to 90 degree and forearm rest on arm rest of chair with hand free from any support. Subjects were asked to use therapeutic putty (85g) for finger resistance exercise [15]. The study used soft, medium or firm variety of therapeutic putty based on the strength of hand muscles [Table/Fig-2].
Hand strengthening exercise by using therapeutic putty (for experimental group).

The following four readings were taken after every 4 weeks : 0 week, 4 weeks (post-I); at 8 weeks (post-II); and at 12 weeks (post-III) for all parameters. The study variables were: Grip strength of right hand (measured by digital hand dynamometer) [16] key pinch strength of right hand (measured by digital pinch meter) [17] pain severity score (Brief pain inventory scale) [18] and hand function was measured by hand assessment tool SF-SACRAH score (0 to 100) [19].
Statistical Analysis
Statistical analysis was performed with the software SPSS 16.0 window version. Descriptive analysis of all the variables among both the group was done. Effect of treatment was compared using one-way ANOVA followed by Post-hoc analysis. Comparison was done between the groups for all the variables (grip strength, key pinch strength of right hand, pain severity score, and SF-SACRAH score) at 0 week, 4 week, 8 week and 12 week by using unpaired t-test and significance level was set at p<0.05.
Results
No significant difference in the variables was observed among both the groups including age, duration of disease, and Disease Modifying Anti Rheumatic Drugs (DMARDs) duration, grip and pinch strength of right hand, pain severity score and hand function score [Table/Fig-3].
| Variables | (Control) Group A (Mean±SD) | (Experimantal) Group B (Mean±SD) | t-value | p-value |
|---|
| Age (years) | 40.864±7.034 | 41.064±7.403 | 0.545 | 0.57 |
| Duration of illness (years) | 5.586±3.275 | 5.765±3.315 | 0.712 | 0.45 |
| DMARDs duration | 3.677±1.873 | 3.854±1.908 | 1.039 | 0.30 |
| Right hand grip strength (kg) | 8.810±2.208 | 8.640±2.574 | 0.979 | 0.329 |
| Right hand key strength (kg) | 4.544±1.730 | 4.566±1.732 | 0.041 | 0.967 |
| Pain severity score | 6.243±1.917 | 6.186±1.655 | 0.226 | 0.821 |
| Hand function score SF-SACRAH | 59.275±37.00 | 59.926±27.328 | 0.144 | 0.886 |
t-value calculated by sample independent t-test
DMARDs: Disease modifying anti rheumatic drugs; SF-SACRAH: Score for assessment and quantification of chronic rheumatic affections of hands
[Table/Fig-4] represents the group analysis for all variables in control group (Group A). No significant difference was observed among the variables from baseline to 12th week.
Mean comparison of various variables with in group A (Control Group).
| Variables | Week (mean±SD) | Mean difference | p-value |
|---|
| Grip strength of right hand | 0 week 8.810±2.208 | 4th week 8.808±2.187 | 0.0019 | 1.00 |
| 4th week 8.808±2.187 | 8th week 8.873±2.185 | -0.0650 | 0.997 |
| 8th week 8.873±2.185 | 12th week 8.802±2.166 | 0.0708 | 0.997 |
| 0 week 8.810±2.208 | 12th week 8.802±2.166 | -0.0077 | 1.00 |
| Key strength of right hand | 0 week 4.544±1.730 | 4th week 4.509±1.677 | 0.0349 | 0.999 |
| 4th week 4.509±1.677 | 8th week 4.570±1.722 | -0.0611 | 0.996 |
| 8th week 4.570±1.722 | 12th week 4.580±1.723 | -0.0097 | 1.00 |
| 0 week 4.544±1.730 | 12th week 4.580±1.723 | 0.0359 | 0.999 |
| Pain severity score | 0 week 6.243±1.917 | 4th week 6.249±2.162 | -0.006 | 1.00 |
| 4th week 6.249±2.162 | 8th week 6.168±2.710 | 0.081 | 1.00 |
| 8th week 6.168±2.710 | 12th week 6.079±2.301 | 0.089 | 0.994 |
| 0 week 6.243±1.917 | 12th week 6.079±2.301 | -0.164 | 0.967 |
| Hand function score | 0 week 59.275±37.001 | 4th week 59.176±36.308 | 0.099 | 1.00 |
| 4th week 59.176±36.308 | 8th week 59.109±36.338 | 0.0669 | 1.00 |
| 8th week 59.109±36.338 | 12th week 59.428±36.894 | -0.318 | 1.00 |
| 0 week 59.275±37.001 | 12th week 59.428±36.894 | 0.152 | 0.314 |
One-way ANOVA followed by Post-hoc analysis
Experimental group
[Table/Fig-5] represents the group analysis within the group in all variable scores of experimental group (Group B). Significant difference was observed among the variables from baseline to 12th week (p<0.001).
Mean comparison of various variables with in group B (experimental Group).
| Variables | Week (Mean±SD) | Mean difference | p-value |
|---|
| Grip strength of right hand | 0 week8.640±2.574 | 4th week10.222±2.108 | -1.084 | 0.012* |
| 4th week10.222±2.108 | 8th week10.921±1.867 | -0.699 | 0.206 |
| 8th week10.921±1.867 | 12th week11.435±2.712 | -0.514 | 0.478 |
| 0 week8.640±2.574 | 12th week11.435±2.712 | 2.298 | <0.001 |
| Key strength of right hand | 0 week4.560±1.730 | 4th week5.409±1.094 | -0.416 | 0.424 |
| 4th week5.409±1.094 | 8th week5.885±1.682 | -0.967 | 0.002* |
| 8th week5.885±1.682 | 12th week6.904±2.261 | -1.019 | 0.001** |
| 0 week4.566±1.730 | 12th week6.904±2.261 | 2.360 | <0.001 |
| Pain severity score | 0 week6.186±1.655 | 4th week5.636±.959 | 0.550 | 0.051 |
| 4th week5.636±.959 | 8th week5.401±.832 | 0.234 | 0.700 |
| 8th week5.401±.832 | 12th week4.487±1.896 | 0.914 | <0.001 |
| 0 week6.186±1.655 | 12th week4.487±1.896 | -1.699 | <0.001 |
| Hand function score | 0 week59.926±27.328 | 4th week51.083±27.488 | 8.842 | 0.134 |
| 4th week51.083±27.488 | 8th week39.795±26.160 | 11.288 | 0.028* |
| 8th week39.795±26.160 | 12th week37.074±26.079 | 2.720 | 0.912 |
| 0 week59.926±27.328 | 12th week37.074±26.079 | -22.852 | <0.001 |
One-way ANOVA followed by Post-hoc analysis
**Highly significant 0.001; *Significant p-value <0.001
Between groups analyses of right hand grip strength showed that there was no significant difference between both the group at the baseline reading i.e., 0 week and a highly significant difference was seen on 12th week [Table/Fig-6]. Control group (group A) showed no significant improvement in the mean score of the variables whereas, experimental group (group B) showed statistically significant improvement.
Comparison of grip strength, key strength of right hand, pain severity score and hand function score between Group A and B.
| Variables | Week | Group A (Mean±SD) | Group B (Mean±SD) | t-value | p-value |
|---|
| Right hand grip strength | 0 week | 8.810±2.208 | 8.640±2.574 | 0.501 | 0.617 |
| 4th week | 8.808±2.187 | 10.222±2.108 | -4.721 | <0.001* |
| 8th week | 8.873±2.185 | 10.921±1.867 | -7.228 | <0.001** |
| 12th week | 8.802±2.166 | 11.435±2.712 | -7.697 | <0.001** |
| Right hand key strength | 0 week | 4.544±1.730 | 4.560±1.730 | 0.185 | 0.85 |
| 4th week | 4.509±1.730 | 4.917±1.426 | -1.879 | 0.062 |
| 8th week | 4.580±1.722 | 5.885±1.682 | -5.540 | <0.001** |
| 12th week | 4.544±1.730 | 6.904±2.261 | -8.296 | <0.001** |
| Pain severity score | 0 week | 6.243±1.917 | 6.186±1.655 | 0.226 | 0.821 |
| 4th week | 6.249±2.162 | 5.834±.959 | 1.750 | 0.082 |
| 8th week | 6.168±2.710 | 5.401±.832 | 2.743 | 0.007* |
| 12th week | 6.079±2.301 | 4.487±1.896 | 5.417 | <0.001** |
| Hand function score | 0 week | 59.275±37.00 | 59.926±27.328 | -0.144 | 0.886 |
| 4th week | 59.176±36.308 | 51.083±27.488 | 1.804 | 0.073 |
| 8th week | 59.109±36.338 | 39.795±26.160 | 4.378 | <0.001** |
| 12th week | 59.428±36.894 | 37.074±26.079 | 5.021 | <0.001** |
Between group analyses of right hand grip strength was done using unpaired t-test
**Values are highly significant
Discussion
This study was conducted to describe the effect of hand strengthening exercise by using therapeutic putty along with conventional physiotherapy treatment on various function of hand in chronic RA female patients. Brorsson S et al., had introduced therapeutic putty as hand strengthening exercise tool. Therapeutic putty is composed of borosiloxane chain compounds. It exhibits resistance to deforming force which is proportional to the force applied to them. This resistance properties makes it useful as therapeutic putty [15]. Fleckenstein JL et al., proved that exercise performance with progressive resisted technique causes more protein synthesis in muscle thus increased the muscle volume [20].
The study by van den Endea CHM et al., also supports the index study. They found an intensive role of conservative exercise programme in reducing the level of pain in RA patients [21]. Dogu B et al., also showed that strengthening exercise of hand decrease pain, improved hand function and quality of life in RA patients [22].
Schnornberger CM et al., reported that strengthening exercise improve the joint stability which help in pain reduction and better work performance in RA female sufferers [23]. Results of this study were in agreement with the result of Ronningen A and Kjeken I. They suggested that strengthening exercise for flexors and lumbricals helped to improve the kinaesthetic sense and joint stiffness which enhances joint loading for joint functions and proprioception of weight bearing for upper limbs [24].
The data showed that hand strengthening exercise by using therapeutic putty was more beneficial than the active exercise of hand in Indian female population. It is widely acknowledged that regular physical activity or exercises provides multiple health benefits for the general population and patients with chronic diseases, including RA.
Limitation(s)
Only females were included and the age range was wide. Some of them were unmarried and some were menopausal. These factors led to difficulties in comparison.
Conclusion(s)
This study was designed to evaluate the effects of hand strengthening exercise by using therapeutic putty on strength of right hand, pain severity and function in chronic rheumatoid hand. The finding supports the use of hand strengthening exercise as an adjunct to routine physiotherapy treatment in the rehabilitation of Chronic RA patients.
Future exploration is expected to discover the impact of hand strengthening exercises in quality of life in chronic RA population.
t-value calculated by sample independent t-test
DMARDs: Disease modifying anti rheumatic drugs; SF-SACRAH: Score for assessment and quantification of chronic rheumatic affections of hands
One-way ANOVA followed by Post-hoc analysis
One-way ANOVA followed by Post-hoc analysis
**Highly significant 0.001; *Significant p-value <0.001
Between group analyses of right hand grip strength was done using unpaired t-test
**Values are highly significant