Intravitreal injection of pharmacological agents has become an effective approach for administering therapeutic levels of drugs for vision-threatening vitreo-retinal diseases of the eye. Various drugs such as corticosteroids, antifungal, antiviral and antibiotics drugs are administered into the vitreous cavity by this method [1].
Recently, intravitreal injections of anti-Vascular Endothelial Growth Factor (anti-VEGF) drugs e.g., ranibizumab, aflibercept and bevacizumab have become a commonly performed procedure in ophthalmology with widespread application in treating neovasularisation and macular oedema associated with retinal disorders [2]. Intravitreal injection is commonly performed under topical anaesthesia. Avoidance of local as well as regional anaesthesia is advantage of the approach, but it is offset by the inevitable pain felt by the patient during the procedure [3]. Many patients require multiple intravitreal injections but intraprocedural as well as postprocedural pain may affect their compliance to the planned subsequent injections [4].
Review of literature has revealed multiple studies pertaining to reduction of pain associated with such procedure on aspects of anaesthetic technique [5-7], needle gauze [7,8], injection technique [3,7,9] and location of injection [10,11], however, no studies were found for the correlation of pain perception with needle bevel tip orientation during intravitreal injection. The effect of needle bevel position as well as design on pain perception has been established in relation to subcutaneous injection [12] and dental local infiltration anaesthesia [13]. Hence, this study was designed to evaluate the influence of needle bevel tip orientation on pain level associated intravitreal injection if any, based on patient feedback.
Materials and Methods
This prospective interventional, single blinded study was conducted at Department of Ophthalmology, Military Hospital Bhopal between December 2017 to January 2020. Institutional Ethical Clearance (IEC/17/01) and written informed consent was taken from all the patients. The study was performed in accordance with the tenets of the Declaration of Helsinki.
Assuming a standard deviation of 17.1 units [10] on VAS pain scoring (0-100 mm) and clinical significant difference in pain perception of atleast 10 units (out of 100) between the two test groups, a sample of 60 subjects was calculated in each group to achieve a power of 89% with level of significance of 5% for this study.
Inclusion and Exclusion criteria: Patients included for the study were those planned for intravitreal ranibizumab injection for conditions such as Neo-vascular ARMD, central DME and macular oedema secondary to RVO. Patients excluded from the study were with any ocular condition that may affect pain perception like previous history of intravitreal injections, surgery involving manipulation of adnexal conjunctiva and sclera except Cataract surgery, past Retinal LASER or Cryo-therapy procedures, herpetic eye diseases, inflammatory conditions of eye and systemic conditions involving use of systemic analgesic or sedatives. Besides this patient having barrier to communication were also excluded.
A total of 120 eyes of 120 patients were included for the study. Patients were allocated into two groups by assigning the successive patient bearing odd serial number to Group 1 and those bearing the even serial number to Group 2 from the Operative patient list prepared on each scheduled day randomly by a paramedical staff who was blinded for the study. In the Group 1 intravitreal injection was given with the bevel tip parallel to limbus margin, while in the Group 2 it was administered with the bevel tip perpendicular to limbus margin [Table/Fig-1].
Intravitreal needle bevel tip orientation while initiating insertion.
a) Parallel to limbal margin in Group 1
b) Perpendicular to limbal margin in Group 2
Mydriasis was achieved by instilling one drop of 1% Tropicamide 30 minutes before the procedure. A drop of of antibiotic (Moxifloxacin 0.5%) was instilled in the eye 10 minutes before injection. All intravitreal injections were performed in the supine position taking aseptic precautions. Two drops of Proparacaine (0.5%) drops (Company: Sunways India Pvt., Ltd.,) was instilled for achieving anaesthesia followed by cleaning of adnexal area with Povidone iodine (10%) and insertion of sterile drape and speculum. Povidone iodine (5%) was applied over the globe and rinsed after 2 minutes using saline. The injection site was 3.5 mm and 4 mm from the limbus margin for the pseudo-phakic and phakic patient, respectively. Injection ranibizumab (0.05 mL=0.5 mg) was loaded from parent injection vial and injected into the vitreous cavity through the infero-temporal quadrant of the indicated eye with straight injection technique using 30-gauze×½ inch needle supplied with the sterile pack of Ranibizumab (Company: Lucentis; Genentech, San Francisco, USA). None of the patient received analgesic or sedatives in preprocedure and postprocedure period.
After completion of the procedure, the patient was taken to the recovery room and asked to rate their perceived pain on a VAS of 0 (no pain) to 10 (unbearable/worst pain). The VAS is a frequently used tool for the assessment of range of pain perception and has been used in similar studies measuring ocular comfort [1,5,6,8,10,11,14,15]. It is a horizontal line measuring 10 cm. A score of 0 presents “no pain at all” to a score of 10 representing “worst pain ever”. Patients were explained about the VAS along with method of marking by a trained paramedical staff before undergoing the procedure. Within 5 minutes of receiving injection, they were asked to place a line perpendicular to the VAS line at point corresponding to perceived pain during the injection with a standard set of questionnaire pertaining to pain levels by a trained paramedical staff in the absence of surgeon. The distance of the marked line on the VAS was measured in millimeter and transformed into a score between (0-100).
To achieve standardisation, all injections in the study were performed in the infero-temporal quadrant with the same needle size (30 gauze of a single company), same drug by a single ophthalmologists on patient indicated for their first intravitreal injections.
Statistical Analysis
The data was recorded and compiled in Microsoft Excel worksheet and analysed by using statistical software Statistical Package for Social Sciences (SPSS) Version 24.0 (IBM Corp. Released 2016. IBM SPSS Statistics for Windows, Version 24.0 Armonk, NY: IBM Corp.). Independent t-test was used for the comparison of the two groups. Chi-square test was used for the comparison of nominal variables of subject groups. Pearson’s correlation test was used to evaluate the relationship between quantitative variables and pain scores. The p-values <0.05 were considered as statistically significant.
Results
Subject characteristics of two study groups as depicted in [Table/Fig-2] included 53 (44.17%) male and 67 (55.83%) female, with a mean age of 65.16±7.91 years (range 44–83 years). Injected eyes included 63 (52.50%) right eyes and 57 (47.50%) left eyes. The diagnosis of the patients is summarised in [Table/Fig-3] with most common indications being Diabetic Retinopathy. No adverse effect other than mild sub-conjunctival haemorrhage was encountered in the study.
Subject characteristics and pain scores of two study groups (120 patients).
| Variables | VAS score |
|---|
| Group 1 (n=60) | Group 2 (n=60) | Cumulative (n=120) | p-value |
|---|
| Age (years)Mean±SD | 64.95±8.38 | 65.37±4.93 | 65.16±7.91 | 18.13±7.65 | 0.74*(r=0.12)† |
| ≤65 y=55>65 y=65 | - | - | - | 17.37±7.5018.78±7.78 | 0.32* |
| Sex | M=25F=35 | 22.08±5.6424.70±5.60 | M=28F=32 | 12.87±4.2312.48±5.57 | M=53F=67 | 17.22±6.7518.86±8.28 | 0.25* |
| Laterality | R=31L=29 | 22.88±5.0924.39±6.33 | R=32L=28 | 11.84±3.8013.59±5.94 | R=63L=57 | 17.27±7.1219.09±8.17 | 0.19* |
| Diabetes | D=32ND=28 | 25.22±6.0121.77±4.85 | D=34ND=26 | 12.61±5.2912.73±4.57 | D=66ND=54 | 18.72±8.4717.42±6.52 | 0.36* |
| Lens status | P=38PSD=22 | 22.19±5.6826.05±5.01 | P=46PSD=14 | 11.58±4.5915.10±5.39 | P=84PSD=36 | 16.38±7.3522.22±6.82 | 0.0001* |
M: Male; F: Female; R: Right; L: Left; D: Diabetic; ND: Nondiabetic; P: Phakic; PSD: Pseudophakic, ≤65y=less than/equal to 65 year of age, >65 y=more than 65 years of age; VAS: Visual analogue scale (0-100 mm)
Data are as mean±standard deviation or n unless otherwise specified
*Determined by t-test
†Determined Pearson’s (r) correlation coefficient test
The diagnoses of the patients in the study groups.
| Treatment groups | Diabetic Retinopathy (DME) | Neo-vascular ARMD | BRVO | p-value |
|---|
| Group 1 (n=60) | 29 | 26 | 5 | 0.83* |
| Group 2 (n=60) | 28 | 25 | 7 |
| Total (n=120) | 57 | 51 | 12 |
ARMD: Age related macular degeneration; DME: Diabetic macular oedema; BRVO: Branch retinal vein occlusion
*Determined by X2 test
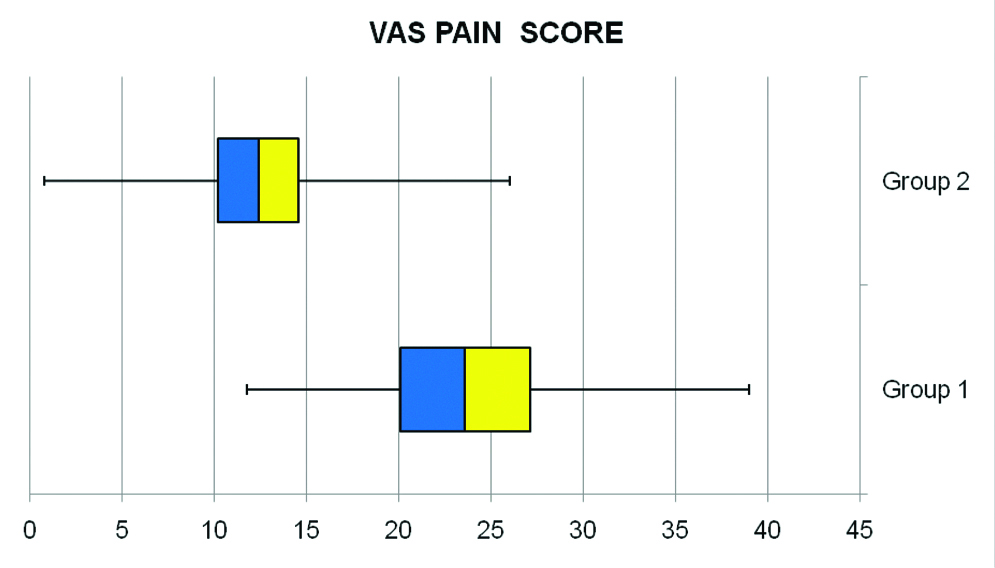
The mean age was similar (p=0.74) in both groups i.e., 64.95±8.38 year in Group 1 and 65.37±4.93 year in Group 2. In Group 1, 25 patients were male and 35 were female. In Group 2, 28 patients were male and 32 were female. The number of patients with respect to sex was similar between groups (p=0.25). Overall, pain scores of all subject patients on the VAS (0-100 mm) ranged from 0.8 to 39, with a mean of 18.13±7.65. The mean VAS pain score in Group 2 was 12.66±4.95, which was lower than mean VAS pain score of 23.61±5.72 in Group 1 [Table/Fig-4]. The analysis of mean VAS pain scores between two groups with two different mode of orientation of needle bevel tip orientation revealed statistically significant difference between the two test groups (p=0.0001) [Table/Fig-5].
Mean VAS pain scores reported in two study groups.
| Group 1 (n=60) | Group 2 (n=60) | p-value |
|---|
| 23.61±5.72 | 12.66±4.95 | 0.0001* |
Data are as mean±standard deviation
*Determined by t-test
Mean Visual Analogue Scale (VAS) pain scores reported in two study groups plotted in Box and whisker chart. The patient’s pain level was evaluated by using a VAS (0-100 mm), where 0=no pain/no distress and 100=agonising pain/unbearable distress.
There was no correlation of pain with age with coefficient correlation (r) of 0.12. Analysis of the mean pain scores (VAS) of the two sub-groups of age (less than/equal to 65 year of age and more than 65 years of age) among both groups revealed statistically nonsignificant difference (p=0.32), however, statistical significant difference of VAS pain score was revealed for the lens status between two groups (p<0.0001) with higher pain scores among Pseudo-phakics compared to Phakic patients [Table/Fig-2]. In 3 months follow-up of each patient, none of the patient had significant sub-conjunctival haemorrhage, secondary cataract due to lens touch, retinal detachment and endophthalmitis.
Discussion
Although, the intravitreal injections are given under surface anaesthesia, mild to moderate pain is still perceived by the patient during needle insertion which may lead to sudden involuntary ocular movement, which can further lead to secondary complication in form of sub-conjunctival haemorrhage, injury to lens and retinal detachments [16]. Besides this, perceived pain may lead to nonadherence of the patients to the planned future intravitreal injections [17]. Hence, for optimising patient comfort and compliance, it is imperative to persistently bring out the factors affecting pain with such injections and simultaneously, initiate attempts to reduce associated pain by modifying factors such as technique of anaesthesia, selection of appropriate needle gauge and modification of needle design.
Review of literature has revealed multiple studies pertaining to reduction of pain associated with such procedure on aspects of anaesthetic technique [5-7], needle gauze [7,8], injection technique [3,7,9] and location of injection [10,11]. Further, it will be prudent to address the bio-mechanical factors in form of interaction between needle design like bevel angle, bevel lancet tip shape, length of bevel, bevel heel surface cut, bevel grind with anatomical and physiological factor of the injection site, which may influence pain associated with such injections.
The mean pain score (VAS) during intravitreal injection procedure in this study was 18.13±7.65. The mean pain score (VAS) in Group 1 and Group 2 was 23.61±5.72, and 12.66±4.95, respectively. Overall, the pain scores (VAS) in both study groups were mild in severity. These scores were similar to previous studies stating milder level of pain perception with intravitreal injections [8,12]. The overall pain level scores obtained in this study for intravitreal injections were corroborating with the pain score of 16.8±2.3 as recorded by Moisseiev E et al. in their study for pain evaluation in intravitreal injection of ozurdex and bevacizumab [10,11]. However, the mean pain score (VAS) ranging from as low as 2.77±2.12 to as high as 36.3±13.3 has been recorded in various studies [8,18].
In this study, the mean pain score (VAS) of 12.66±4.95 in Group 2 was lower than mean pain score (VAS) of 23.61±5.72 in Group 1 atleast by 10 units, which is of clinically significance, besides being statistical significant (p=0.0001). This difference is attributed to interplay of initial biomechanical interaction between the bevel needle tip and area of insertion during needle insertion, having influence on pain perception, which is different with different orientation of needle and contributing to the different pain scores. This is assumed that, the initial insertion of the bevel needle tip parallel to limbus margin in Group 1 may have traversed relatively greater number of pain fibres compared to initial insertion of bevel needle tip perpendicular to limbus margin in Group 2. The sensory and pressure receptors situated on the sclera, episclera and conjunctiva of the eye are triggers of pain signal generation on initiation of intravitreal injection [19]. The sensory innervation of anterior sclera and episclera is by the long posterior ciliary nerve derived from the ophthalmic branch of trigeminal nerve, which enters the sclera near optic nerve and course forward towards ciliary body along the with long posterior and short posterior artery in longitudinal fashion. There is no anatomical difference in corneo-scleral nerve density between quadrants, but larger nerves are distributed along long axis of the eye [20]. Electron microscopy and immunohistological staining have revealed predominantly longitudinal pattern course and branching pattern of these nerves in sclera, supra-choroidal space, and choroid [21,22]. Hence, it is reasonable to assume that relatively more pain receptors may have stimulated consequent to more pain fibres traversed while initiating insertion of the bevel of the needle tip as parallel to the limbus margin compared to perpendicular to limbus margin.
Secondly, the peak penetration force of needle injection may be lesser while injecting with bevel tip perpendicular to the limbus compared to while injecting parallel to the limbus as the rigid collagen fibres of sclera in this region have been mapped to be meridonial oriented compared to circumferential pattern at limbus and near optic nerve head [23]. It has been established that pain perception scores are correlated with the peak penetration force of needle insertion [24].
This is believed that keeping the orientation of needle tip bevel orientation perpendicular to limbus margin while inserting the intravitreal injection can further reduce the pain associated with intravitreal injections and may improve patient compliance particularly with larger gauze needles. Besides, it will be interesting to further investigate; if modification of calibre of needle shaft from conventional cylindrical pattern to spindle or diamond pattern along with orientation along longitudinal axis of eyeball may further reduce pain associated with such injections.
Additional characteristics of the pain associated with intravitreal injections were revealed in this study. No correlation of pain with age with coefficient correlation (r) of 0.12 was found. Analysis of the mean pain scores (VAS) of the two sub-groups of age (less than 65 year of age and more than 65 years of age) among both groups revealed statistically nonsignificant difference (p=0.32) [Table/Fig-1]. The mean pain score (VAS) was 17.37±7.50 in age group ≤65 years and 18.78±7.78 in age group >65 years. Moisseive E et al., and Onakpoya OH and Akinwande JG, have also found no correlation between pain score and age [15,25]. It has been shown that nerve density decreases with age and more significantly over 70 years [25], a fact that could suggest older patients experience less pain during intravitreal injections, however, in our study mean pain score (VAS) was higher in higher age groups.
No significant difference was found between pain perception of male and female sub-groups, (p=0.25), however the overall mean pain score (VAS) in female sub-group (18.86±8.28) was higher than male subgroup (17.22±6.75). In most studies, no relationship has been reported between gender and pain perceived during intravitreal injection [11,19,20]. Higher pain sensitivity of female may be assumed due to influence of specific sex hormones on pain sensitivity.
The presence of Diabetes Mellitus had no effect on pain scores (p=0.36.), however the overall mean pain score (VAS) in Diabetic sub-group (18.72±8.47) was higher than nondiabetic sub-group (17.42±6.52). Infact, diabetic patient should have perceived lesser pain on account of reduced corneo-scleral sensitivity [26,27]. The reason could be because of subclinical ocular irritation and inflammation among diabetic affected patients. Moisseiev E et al., have also reported no association between intravitreal injection pain scores and presence of diabetes mellitus [11].
In this study, statistical significant difference in mean pain scores (VAS) was found for the lens status (p≤0.0001) with Pseudo-phakes sub-group (22.22±6.82) having higher pain scores than Phakic sub-groups (16.38±7.35). Similar findings have been reported in study conducted by Moisseiv E et al., for evaluation of pain in intravitreal injection for Ozurdex and Bevacizumab [10]. Authors assume that relatively anterior location of intravitreal injection insertion among Pseudo-phakes may have stimulated posterior-most part of ciliary body triggering more pain generation; however, we believe it to be clinical insignificant as it is not affecting treatment decisions.
Although the injecting ophthalmologist was right-handed, no statistical difference was found between pain levels in right eye and left eye (p=0.19). Besides this, no significant difference was found between pain scores and indications of the injections.
Limitation(s)
This study was based on subjective assessment method of pain sensation which is variable and cannot be directly measured. Beside this study could not be designed as double-blinded study, as it was not possible to blind the operating ophthalmologists for the use of the specific orientation of bevel needle tip on the specific study patients, which might have introduced surgeon bias, however, standardised surgical protocol was designed to limit this bias. Besides this, we used straight injection technique for needle insertion which is associated relatively higher amount of vitreous reflex. Further studies with larger series are needed to confirm the difference of pain scores with different initial orientation of needle bevel tip during intravitreal injection especially with wider gauze of needle.
Conclusion(s)
This is the study comparing the pain perception with the different orientation of needle bevel tip during intravitreal injections. Pain associated with intravitreal injection may be further minimised by orienting needle bevel tip as perpendicular to limbal margin while intiating insertion.
M: Male; F: Female; R: Right; L: Left; D: Diabetic; ND: Nondiabetic; P: Phakic; PSD: Pseudophakic, ≤65y=less than/equal to 65 year of age, >65 y=more than 65 years of age; VAS: Visual analogue scale (0-100 mm)
Data are as mean±standard deviation or n unless otherwise specified
*Determined by t-test
†Determined Pearson’s (r) correlation coefficient test
ARMD: Age related macular degeneration; DME: Diabetic macular oedema; BRVO: Branch retinal vein occlusion
*Determined by X2 test
Data are as mean±standard deviation
*Determined by t-test