Neuraxial anaesthesia is commonly performed for all surgical procedures carried on lower abdomen, pelvis and lower limbs to provide adequate surgical anaesthesia and analgesia [1]. Spinal anaesthesia, despite providing a satisfactory surgical exposure, with just a small amount of local anaesthetic, has a drawback due to unpredictable perturbations in the haemodynamic parameters as a result of sympatholysis. These effects are proportional to the level of sympathetic blockade [2]. Intrathecally, administered local anaesthetics and opioids have been shown to have a synergistic analgesic effect [3,4], hence requiring relatively lower dosage. Neuraxial opioids also allow prolonged analgesia in the postoperative period and faster recovery from spinal anaesthesia [5].
Fentanyl, a highly lipid soluble, pure μ-agonist opioid with rapid onset and short duration of action and has been used with various local anaesthetics for a wide variety of surgical procedures [5,6]. But, it has some undesirable side effects like postoperative nausea and vomiting, pruritus, respiratory depression, urinary retention etc., due to mu recepter agonism. Butorphanol is a competitive antagonist at mu opioid receptor and partial agonist at the kappa opioid receptor. It binds to kappa receptor in the brain and spinal cord which is responsible for nociception producing analgesia devoid of mu receptors related side effect. Kappa-agonism also cause dysphoria at therapeutic or supertherapeutic doses and this gives butorphanol a lower potential for abuse than other opioid drugs [7], accounting for its easier availability in comparison to fentanyl and other potent opioids.
So to enlighten further, the present study was done with the primary objective to compare the perioperative analgesic efficacy of intrathecal fentanyl with intrathecal butorphanol along with bupivacaine in infraumbilical surgeries. Secondary objective was to assess which of these two drugs was superior in providing haemodynamic stability, favourable sensory and motor block characteristics and associated with minimum adverse effects.
Materials and Methods
Randomised controlled trial was done in a tertiary level teaching hospital, between March 2017 to February 2018 after taking ethics committee clearance (No: MMC/IEC-2017/1505) and written informed consent from the patients.
Inclusion criteria: Total 110 consenting patients of ASA 1 and 2, age between 18-60 years, either sex posted for infraumbilical surgeries under spinal anaesthesia were included in the study. The step wise procedural flowchart for this study is displayed in [Table/Fig-1].
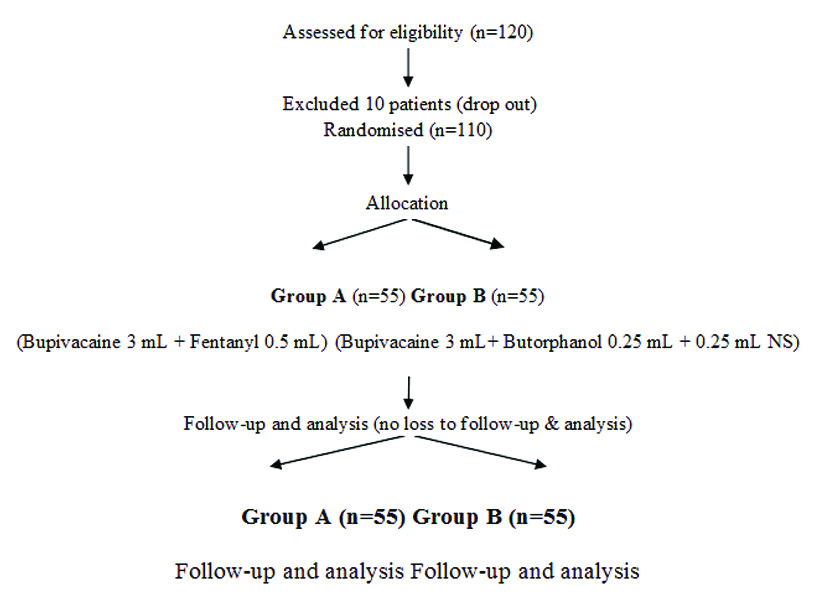
Exclusion criteria: Patients with any cardio-respiratory disease, hepatic and renal disease, Central Nervous System (CNS), endocrine disorder, pregnant patient, conversion to general anaesthesia and failure to spinal anaesthesia were excluded. All patients were allocated randomly according to the computer generated random numbers into two groups A (n=55) and B (n=55). During the planning stage of the study, the sample size was calculated with the help of power analysis. The sample size was calculated on the basis of the duration of postoperative analgesia as the primary outcome measure. It was calculated that 55 subjects were required per group in order to detect a difference of 30 minutes between groups in this parameter with 80% power and 5% probability of Type-I error. This calculation assumed a standard deviation of 45 min for duration of postoperative analgesia and two-sided testing. Extrapolating to two groups, the recruitment target was being set at 110 subjects overall. Around 10% were added to compensate for any loss of power resulting from any drop outs. Group A (Fentanyl) received 3 mL 0.5% hyperbaric bupivacaine and 0.5 mL fentanyl (25 microgram, total 3.5 mL). Group B (Butorphanol) received 3 mL 0.5% hyperbaric bupivacaine and 0.25 mL butorphanol (250 microgram) by insulin syringe and 0.25 mL NS (total 3.5 mL). All intrathecal drugs formulation were prepared under strict aseptic precautions by an another anaesthesiologist who followed opaque sealed envelope technique and did not participated in the procedure of subarachnoid block and in data collections. Another anaesthesiologist, who was not involved in drug formulations, performed the subarachnoid blocks and participated in the data collection. After proper preanaesthetic check-up, patients were taken into the operating room. An 11-point Visual Analogue Scale (VAS) scoring system for assessment of postoperative pain was explained during the preanaesthetic check-up. In the operating room, an intravenous cannulae of 18G were established on non-dominant hand and Ringer lactate solution of 10 mL/kg were started. Baseline HR, SBP, DBP, Mean Arterial Pressure (MAP), Respiratory Rate (RR) and peripheral arterial Oxygen Saturation (SPO2) were recorded.
Subarachnoid block were performed under strict aseptic conditions in the sitting positions at the level of L4-L5 intervertebral space using 26G Quincke spinal needle after infiltrating the skin with 1 mL of 2% lidocaine. Immediately following the subarachnoid block, the patients were put in supine position. Intraoperative vitals (HR, SBP, DBP, MAP, RR, SPO2) were recorded at five minutes intervals for the first 30 minutes from the time of injection of spinal drugs and thereafter every 15 minutes upto completion of surgeries. This data were recorded by the primary investigator, who was unaware of the patient allocation. The highest level of sensory block were determined in the midclavicular line bilaterally by insensitivity to cold alcohol swab tested every two minutes till the maximum height achieved. Sensory testing was performed at every 15 minutes interval till two segment regression of sensory block. Motor block were assessed using the modified bromage scale (1=complete block; 2=Almost complete block; 3=partial block; 4=detectable weakness of hip flexion while supine; 5=no detectable weakness of hip flexion while supine; 6=able to perform partial knee bending), till achievement of the highest motor level. Side effects such as hypotention, bradycardia, nausea, vomiting, sedation, pruritus, shivering and respiratory depression (RR <9 or SPO2 <90) were recorded.
Sedation was recorded by Ramsay sedation score (1=awake and anxious, 2=awake, cooperative, oriented; 3=awake response only to commands; 4=asleep, brisk response to light; 5=asleep, sluggish response to light, 6=asleep, no response to light). The quality of postoperative analgesia was assessed by using VAS at 15 minutes, 30 minutes and thereafter every 30 minutes till two hours postoperatively and then every hourly, till six hours postoperatively. When patients complained of pain for the first time and VAS score >4, rescue analgesia were given with 75 mg aqueous Diclofenac Sodium in 100 mL of 0.9% NS through intravenous route and time were noted.
Statistical Analysis
For statistical analysis, Statistical Package for the Social Sciences (SPSS 24.0 version) and Graph Pad Prism version 5 were used. Data were summarised as mean and standard deviation for numerical variables and percentages for categorical variables. Two-sample t-tests were used for a difference in mean involved independent samples or unpaired samples. Unpaired proportions were compared by Chi-square test or Fischer’s-exact test, as appropriate and a p-value ≤0.05 was considered as statistically significant.
Results
Both groups were comparable with regards to age, sex, Body Mass Index (BMI), ASA grade and duration of surgery and there was no statistically significant difference between them [Table/Fig-2].
Demographic variables of the patients.
| Variables | Group A (Bupivacaine+Fentanyl) Mean±SD | Group B (Bupivacaine+Butorphanol) Mean±SD | p-value |
|---|
| Age in years | 36.5±9.5 | 36.47±10.54 | 0.98 |
| Sex (Male: Female) | 37:18 | 40:15 | 0.53 |
| BMI (Kg/m2) | 22.11±1.52 | 22.21±1.53 | 0.73 |
| ASA I: ASAII | 1.50±0.50 | 1.47±0.50 | 0.71 |
| Duration of surgery (in minutes) | 73.27±14.69 | 75.36±13.53 | 0.44 |
SD: Standard deviation; BMI: Body Mass Index; ASA: American Society of Anesthesiology
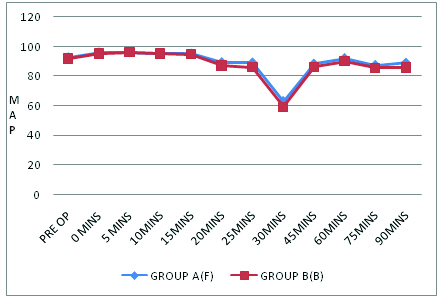
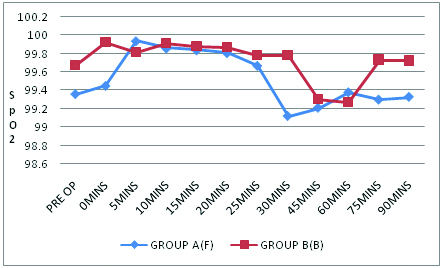
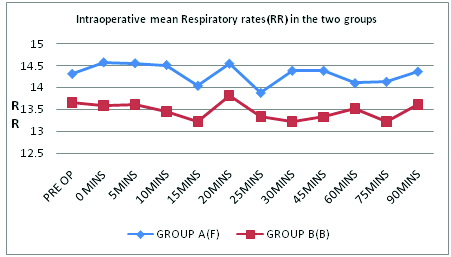
The mean values of HR, SBP and DBP at different time interval were comparable among the groups and there was no significant statistical difference [Table/Fig-3]. The mean values of MAP, SpO2 and Respiratory rates(RR) were comparable amongst the two groups with their differences being statistically insignificant (p value>0.05) in the majority readings, with a few exceptions, which were clinically insignificant [Table/Fig-4,5 and 6]. While, in Group A (Bupivacaine+Fentanyl group) highest sensory level attained was T6 in 32 (58.18%) patients and T8 in 23 (41.82%), in Group B (Bupivacaine+Butorphanol group) 34 (61.82%) patients had highest level T6 and 21 (38.18%) patients had level T8. It can be inferred from [Table/Fig-7] that mean time to two segment regressions from highest level, mean duration of motor block and mean time to rescue analgesic were prolonged in Butorphanol group in comparison to Fentanyl group and stastically significant. The adverse effects were more or less comparable between the groups. However nausea/vomiting (5) and pruritus (4) were observed more in the Fentanyl group than the Butorphanol group (2,2). There was no incidence of any hypotension, bradycardia, sedation, respiratory depression or shivering in both the groups.
| Heart Rate (HR) Mean±SD | Systolic BP (*) Mean±SD | Diastolic BP (*) Mean±SD |
|---|
| Time intervals | Group A | Group B | Group A | Group B | Group A | Group B |
|---|
| On Arrival AT O.T (#) | 85.06±7.21 | 86.35±5.21 | 129.27±5.67 | 127.56±6.35 | 74.78±8.41 | 73.64±7.94 |
| Just Before SAB ($) | 85.75±5.74 | 84.86±4.66 | 128.47±5.58 | 127.56±6.35 | 80.29±2.95 | 78.84±3.43 |
| After 5 min | 86.20±5.44 | 87.46±5.96 | 126.72±5.23 | 126.12±5.97 | 81.20±0 .98 | 81.17±0 .99 |
| After 10 min | 84.18±6.91 | 86.58±5.30 | 125.23±5.38 | 124.38±5.96 | 80.69±1.5 | 80.51±1.28 |
| After 15 min | 85.04±6.61 | 85.89±5.08 | 123.87±5.43 | 122.76±5.81 | 81.38±1.43 | 81.20±1.53 |
| After 20 min | 83.75±7.37 | 86.95±5.94 | 119.23±9.95 | 112.18±11.55 | 75.39±9.53 | 72.93±10.52 |
| After 25 min | 86.56±4.98 | 86.42±4.45 | 116.72±8.72 | 113.90±11.71 | 75.97±9.65 | 72.69±10.92 |
| After 30 min | 86.89±5.96 | 86.98±5.98 | 117.32±9.28 | 114.25±10.64 | 75.79±9.57 | 73.88±10.87 |
| After 45 min | 84.78±6.99 | 85.46±5.04 | 116.21±10.23 | 112.56±10.92 | 74.98±9.45 | 71.42±9.91 |
| After 60 min | 84.69±6.92 | 87.49±4.45 | 128.46±5.43 | 125.36±9.61 | 74.51±9.90 | 72.80±10.39 |
| After 75 min | 85.70±6.92 | 86.24±3.74 | 119.85±12.5 | 116.29±15.33 | 72.57±9.93 | 70.53±10.58 |
| After 90 min | 86.53±5.58 | 87.26±3.47 | 124.46±11.69 | 118.61±11.55 | 72.31±12.16 | 69.14±11.11 |
BP(*): Blood pressure; O.T(#): Operation theatre; SAB($): Sub arachnoid block or Spinal anaesthesia; SD: Standard deviation
Intraoperative mean map (Mean Arterial Pressure) in two Groups.
p significant only at 25 mins (0.02)
Intraoperative mean SPO2 levels in the two Groups.
p significant only at 30 mins (0.04)
Intraoperative mean respiratory rates in the two Groups.
p significant only at 20 mins (0.05)
Block and analgesia characteristics.
| Group A Mean±SD | Group B Mean±SD | p-value |
|---|
| Time of onset of sensory block (in minutes) | 3.08±0.22 | 3.15±0.25 | 0.13 |
| Highest sensory level | T6 (T6-T8) | T6 (T6-T8) | 0.84 |
| Time to two segment regression from highest sensory level (in minutes) | 41.94±1.73 | 50.56±4.43 | <0.0001 |
| Time of onset of motor block (in minutes) | 5.28±0.32 | 5.27±0.32 | 0.96 |
| Duration of motor block (in minutes) | 81.23±4.87 | 109.83±2.61 | <0.0001 |
| Time to rescue analgesic (in minutes) | 289.27±7.37 | 378.41±10.25 | <0.0001 |
SD: Standard deviation; p less than 0.001 significant
Discussion
Comparative studies between butorphanol and fentanyl or other pure mu receptor agonists, or even butorphanol vs plain bupivacaine heavy, as adjuvants to spinal anaesthesia are relatively fewer in number. Most of the available literature have been mostly focused on lower limb orthopaedic surgeries [1,7,12-14], or endoscopic urological surgeries [15,16] and gynaecological and obstetric surgeries [17-19]. The study by Upasna B et al., was the only contemporary study on comparison of intrathecal Fentanyl and Butorphanol as adjuvants to spinal anaesthesia in different varieties of infraumbilical surgeries comparable to this study [20]. Hence, this study was undertaken to compare these two drugs in a variety of infraumbilical surgeries to get a better idea regarding their perioperative analgesic and anaesthetic efficiency as opposed to only one variety of surgery. There was also a controversy on the optimum intrathecal dose of butorphanol with variations in the range of 25 μgm-200 μgm in the above referred studies. The studies conducted by Gupta K et al., and Reddy NG et al., both of them being based on lower limb orthopaedic surgery [1,7], used a dose of 200 μgm intrathecal butorphanol along with bupivacaine heavy. Similar dose was also used by Singh SN et al., in abdominal and vaginal hysterectomies under spinal anaesthesia without any significant side effects [17]. Thus, a dose of 25 μgm Fentanyl and 250 μgm of Butorphanol as adjuvants with Bupivacaine heavy 3 mL, in subarachnoid block was used in this study with an intention of testing the perioperative efficacy, especially that of butorphanol as against a relatively fixed and already established dose of fentanyl. The other major concern was whether this dose of butorphanol was associated with an increase in adverse effects in comparison with other studies.
Haemodynamic parameters like mean HR, SBP and DBP were within acceptable limits. Both mean SBP and DBP were lower in the butorphanol group as compared to the fentanyl group, although it was not statistically significant, whereas there was no such trend on comparison of mean HRs between the groups, just like the study of Reddy NG et al., [7]. This was unlike the findings of Upasna B et al., where both mean HRs and blood pressure were significantly lower in the butorphanol group between 45-90 minutes of intraoperative period [20]. Haemodynamic parameters were within acceptable limits in line with the study of Reddy IR et al., [12]. Time for onset of sensory block of the fentanyl group (3.0836±0.2158 min) was less than that of butorphanol group (3.1509±0.2464 min) but results were comparable (p>0.05). This was similar to the findings of Kumar A et al., where time to onset of sensory block in fentanyl group was 8±1.4 minutes, while it was 8±3.2 minutes in the butorphanol group [16]. The longer period in the latter study may be due to their much lower dosage of spinal drugs. The maximum sensory level achieved was T6 in both groups in this study much like Reddy NG et al., where they had used 200 μgm of intrathecal butorphanol and bupivacaine heavy 3 mL [7].
Significant finding in this study was in respect of two segment regression time. It was significantly prolonged in butorphanol group B (50.5636±4.4379 min) compared to fentanyl group A (41.9455±1.7365 min). Results obtained were comparable to previous study by Reddy NG et al., where incidence of two segment regression in sensory level in fentanyl group was 40.1542±1.6254 minutes and in butorphanol group 51.4231±4.2389 minutes [7]. However, similar trend was also observed in the study conducted by Singh V et al., where time for sensory regression to S2 from highest sensory was 158±22 minutes in butorphanol group, which was significantly higher than 135±35 minutes in fentanyl group although 25 μgm of fentanyl and butorphanol were used intrathecally, unlike the current study where 250 μgm of butorphanol was used instead [14]. Both these adjuvant opioids did not cause any significant increase in onset of motor blockade which was consistent with the findings of Reddy NG et al., and Kumar B et al., [7,13]. However in Group B, the mean duration of motor block was 109.83±2.61 minutes, which was significantly more than Group A at 81.2364±4.8799 minutes. These findings were quite consistent with Reddy IR et al., (butorphanol group 178.99±13.32 min as opposed to 168.8±9.18 min in fentanyl group) and Upasna B et al., (246±42.6 min in butorphanol group as opposed to 180±16.8 min in fentanyl group [12,20]. This was contrary to the findings of Kumar B et al., who had found no statistical significant difference between duration of motor block between these groups [13]. One explanation may be as they had used 2.5 mL of 5% Bupivacaine and 25 μgm of butorphanol, while in this study 3 mL of 5% Bupivacaine and 250 μgm of Butorphanol was used instead. However, if mean duration of surgery in these groups are compared (73.27±14.69 min for Group A and 75.36±13.53 min for Group B), the motor block was not that inconvenient.
Most significant finding of this study was that time for first request of rescue analgesia was prolonged in Butorphanol group (378.41±10.25 min) compared to Fentanyl group (289.27±7.37 min). Both fentanyl and butorphanol along with bupivacaine provided adequate analgesia and anaesthesia, but butorphanol was more superior in delaying time of rescue analgesic which supports the result obtained from studies of Kumar B et al., time of consumption of rescue analgesia in fentanyl group was 308±14.9 minutes which was significantly less than in butorphanol group at 365.9±12.3 minutes [13]. Similar statistically significant findings were also observed in studies of Reddy IR et al., and Upasna B et al., [12,20].
Pruritus (7.3%vs 3.6%) and nausea/vomiting (9.1% vs 3.6%) were much more common in the fentanyl group in comparison to the butorphanol group which was quite in line with the findings of Reddy IR et al., [12]. It has been already shown that butorphanol can antagonise pruritus and nausea produced by morphine (μ-agonist) while at the same time prolong the duration of analgesia [21], these findings do not corroborate with the findings of Singh V et al., as they have found more pruritus in butorphanol group and hypotension more in fentanyl group [14]. This difference may be due to the difference in doses, type of surgery and racial variations. None of the groups had episodes of hypotension similar to the findings of Reddy NG et al., [7]. Addition of fentanyl (20-25 μg) to low-dose bupivacaine (4 mg) has been reported to increase the perioperative quality of spinal blocks with fewer cardiovascular changes in elderly patients [22]. None of the groups had episodes of hypotension which means that butorphanol much like fentanyl has a scope of use as an adjuvant in spinal anaesthesia in elderly patients with cardiovascular morbidities. Delayed respiratory depression is more commonly associated with poorly lipid-soluble narcotic drugs, like morphine [23]. It was suggested by Bromage PR that lipid-soluble, highly protein bound narcotic analgesics might have lesser probability to exhibit these characteristics and this seems to be true for butorphanol and fentanyl alike [24]. The patients were continuously observed for respiratory depression and sedation in this study and no significant respiratory depression was noted much like Reddy IR et al., and Upasna B et al., thus again pointing out that both these drugs in this route may have fewer side effects [12,20].
Limitation(s)
The inclusion of a control group in this study could have further supported the findings. Postoperative analgesia was monitored only for six hours and total rescue analgesia doses were not recorded. Comparative analysis with respect to gynaecological, orthopaedic and general surgery patients was not undertaken due to inadequate sample size for subgroup analysis. The biggest limitation of the study was attributed to the fact that equipotent intrathecal doses of fentanyl and butorphanol were not known. Future studies need to be directed towards this dearth of knowledge.
Conclusion(s)
Principal findings of the study was that the addition of 25 μg fentanyl or 250 μg butorphanol as adjuvants to hyperbaric bupivacaine 3 mL in intrathecal route for infraumbilical surgeries offered better haemodynamic stability and provided effective and relatively safe anaesthesia. However, butorphanol was significantly better than fentanyl in providing longer duration of analgesia. Butorphanol, with its low abuse potential due to diaphoresis and relatively easier availability in the market in comparison to fentanyl, thus provides us with a suitable alternative as an adjuvant in spinal anaesthesia, although more studies are required in the future to determine its optimum dose.
SD: Standard deviation; BMI: Body Mass Index; ASA: American Society of Anesthesiology
BP(*): Blood pressure; O.T(#): Operation theatre; SAB($): Sub arachnoid block or Spinal anaesthesia; SD: Standard deviation
SD: Standard deviation; p less than 0.001 significant