Systemic Strongyloidiasis- Is It Dissemination or Hyperinfection?
Srinivasan Radhakrishnan1, Swathy Moorthy2, Shilpa Rao3, Shiny Queensty4, Krishnaswamy Madhavan5
1 Postgraduate Resident, Department of General Medicine, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
2 Associate Professor, Department of General Medicine, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
3 Assistant Professor, Department of General Medicine, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
4 Postgraduate Resident, Department of Microbiology, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
5 Professor, Department of General Medicine, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Swathy Moorthy, Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India.
E-mail: drswathymoorthy@sriramachandra.edu.in
An intestinal nematode, with clinical presentations ranging from asymptomatic state to catastrophic outcomes, Strongyloides stercoralis can be a potential cause of mortality, especially in patients with immunosuppressed state. Here, we present a patient with relatively short duration of immunosuppression, developing systemic strongyloidiasis and eventually succumbed after a brief duration of ailment. The index case satisfied the description of a case of hyperinfection. However, there existed a challenge in ruling out dissemination in her since she could not be evaluated completely due to a fulminant course.
Fulminant, Immunosuppressed, Nematode, Strongyloides stercoralis
Case Report
A 58-year-old woman, presented to the outpatient department with multiple episodes of vomiting for five days, reduced appetite and fatigue accompanied by three to five episodes of foul smelling, watery stools for two days. She was a homemaker, diagnosed with rheumatoid arthritis one year ago, initiated on corticosteroids since six months (began with a daily dose of 30 mg of oral prednisolone, tapered over the last four months with a present prescription for 5 mg of oral prednisolone daily). She was suspected to have interstitial lung disease, six months ago.
She was apparently asymptomatic five days ago, with no history of worsening cough or shortness of breath, fever, dysuria or abdominal pain. She was conscious, oriented, afebrile, moderately built, dehydrated with tachycardia and hypotension. Systemic examination was essentially normal barring the Velcro crepitations heard in bilateral infra-axillary and infra-scapular areas.
Her baseline laboratory investigations revealed neutrophilic leukocytosis with eosinophils (1.3%), hyponatraemia with hypochloraemia, 2-fold elevation of transaminases and hyperbilirubinemia (2.32 mg/dL). HIV by ELISA was non-reactive.
She was initiated on IV fluids, parenteral antibiotics (Inj.Ciprofloxacin, Metronidazole). Despite adequate hydration and antibiotic therapy the patient had refractory hypotension. She required intensive care management with inotropic support.
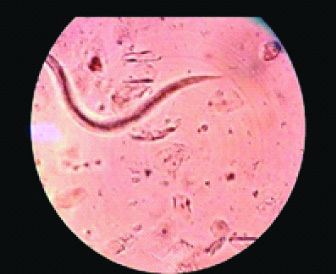
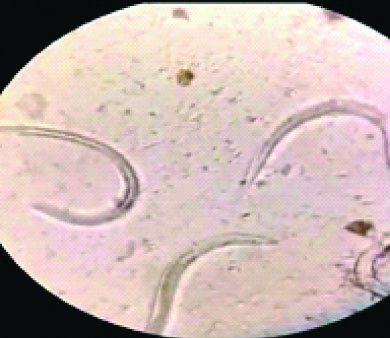
Stool examination showed multiple rhabditiform larvae of Strongyloides stercoralis as seen in [Table/Fig-1]. Ivermectin was initiated based on the weight of the patient (9 mg/day for 2 days). In view of suspected hyperinfection, sputum was inspected and was found to be flooded with multiple larvae of Strongyloides stercoralis as evident in [Table/Fig-2].
Rhabiditiform larvae of Strongyloides sterocoralis in stool.
Multiple larvae in sputum.
The patient deteriorated and was intubated in view of dampening sensorium. Blood culture showed growth of Salmonella typhimurium susceptible to ciprofloxacin which the patient was already on, hence continued. Despite best efforts, the patient succumbed to illness after two days of management in the intensive care.
Discussion
Strongyloides stercoralis related severe clinical syndromes include hyperinfection syndrome and disseminated strongyloidiasis [1]. The female worms possess the ability to produce off-springs without copulation, a process termed parthenogenesis and can infect the same host over and over again without reaching the soil, a phenomenon called autoinfection wherein the rhabditiform larvae which are non-invasive and non-infective, mature in the small bowel of the host into the invasive and infective filariform larvae [2].
When autoinfection occurs in an accelerated manner- the quantitative load of the larvae increases exponentially and this syndrome is termed hyperinfection. Hyperinfection can be triggered by alteration of the individual’s immune status and can manifest with symptoms due to increased larval migration in the gastrointestinal and respiratory systems. High loads of larvae are usually detected in stool and sputum of patients with hyperinfection. Larvae migrate to organs beyond the autoinfection life cycle to produce dissemination [3].
Dissemination and hyperinfection are potentially fatal compounded by the existing risk factors. Gram negative septicaemia, due to translocation of the gut flora during the migration of Strongyloides larva is usually the cause of death [4]. Patients with immunocompromised states, living in areas endemic to Strongyloides stercoralis are vulnerable to the development of one of the above. The index case had evidence of strongyloidosis in a background of immunosuppression, induced by corticosteroids as well as gram negative septicaemia, due to Salmonella species. She indeed bore the risk factors for both the presentations, being from a Southern Asian country which could probably be endemic for strongyloidiasis, apart from the well-known African countries [5].
Risk factors also include infection with Human Immunodeficiency Virus (HIV), Human T-cell Lymphotropic Virus (HTLV), solid organ transplant recipient state, chemotherapy, malignancy, poorly controlled diabetes mellitus, alcoholism and corticosteroid therapy [4]. The index patient had features suggestive of hyperinfection -as evidenced by multiple, actively motile larvae in the stools and sputum with pertaining risk factors being corticosteroid intake and her origin from a probable endemic area. Dissemination cannot be ruled out in her, for evaluation of presence of larvae at sites not endogenous to the natural cycle could not be done due to rapid decline in her health status.
Mortality rate with disseminated disease is around 80% [6]. The fulminant course of the patient described may be a disseminated disease owing to the higher rates of mortality among patients with disseminated disease. The index patient did not have eosinophilia as would be expected in a hyperinfection or dissemination state, while she had neutrophilic leukocytosis incited by the gram negative bacteraemia and corticosteroid mediated suppression of eosinophils [7]. Indications for corticosteroid therapy are innumerable in our day to day practice which tends to increase the risk of such nematodal infection.
Screening for Strongyloides stercoralis, using a stool sample or multiple stool samples -usually three or more [8], increases the likelihood of identifying the larvae in patients from endemic areas. Patients at risk of harbouring the nematode with no identifiable larvae in the stools may be benefitted by screening with serological tests [9] before initiating corticosteroids but the practice has it’s limitation in resource limited settings. To employ these serological tests for screening all patients who are started on steroids for varied indications has still not gained consensus in India.
Perhaps, the incidence of Strongyloides stercoralis in the community is much more than what is actually seen in practice which could amount to the tip of the iceberg as supported by Olsen A et al., who also suggested that strongyloidiasis is an underdiagnosed entity [10].
The index case had a rapid clinical deterioration and succumbed to her illness giving us very little time to evaluate for secondary causes. However, a negative HIV serology, blood culture that showed evidence of Salmonella species and a sterile urine culture were in line with the present clinical judgement.
Conclusion(s)
There is a clear need for establishing guidelines or reaching a consensus about screening strategies and treatment of individuals who are being started on corticosteroid therapy. Dose, duration, type of corticosteroid use and their association with strongyloidiasis are unclear. Therefore, a high degree of suspicion is needed to identify such cases of systemic strongyloidiasis among the patients with immunosuppression.
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. NA
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Nov 05, 2019
Manual Googling: Dec 19, 2019
iThenticate Software: Jan 23, 2020 (9%)
[1]. Corti M, Strongyloides stercoralis in immunosuppressed patientsArchives of Clinical Infectious Diseases 2016 11(1):e2751010.5812/archcid.27510 [Google Scholar] [CrossRef]
[2]. Vasquez-Rios G, Pineda-Reyes R, Pineda-Reyes J, Marin R, Ruiz EF, Terashima A, Strongyloides stercoralis hyperinfection syndrome: A deeper understanding of a neglected diseaseJournal of Parasitic Diseases 2019 (2):167-75.10.1007/s12639-019-01090-x31263320 [Google Scholar] [CrossRef] [PubMed]
[3]. Keiser PB, Nutman TB, Strongyloides stercoralis in the immunocompromised populationClinical Microbiology Reviews 2004 17(1):208-17.10.1128/CMR.17.1.208-217.200414726461 [Google Scholar] [CrossRef] [PubMed]
[4]. Igra-Siegman Y, Kapila R, Sen P, Kaminski ZC, Louria DB, Syndrome of hyperinfection with Strongyloides stercoralisReviews of Infectious Diseases 1981 3(3):397-407.10.1093/clinids/3.3.3977025145 [Google Scholar] [CrossRef] [PubMed]
[5]. Schär F, Trostdorf U, Giardina F, Khieu V, Muth S, Marti H, Strongyloides stercoralis: Global distribution and risk factorsPLoS Neglected Tropical Diseases 2013 7(7):e228810.1371/journal.pntd.000228823875033 [Google Scholar] [CrossRef] [PubMed]
[6]. Mokhlesi B, Shulzhenko O, Garimella PS, Kuma L, Monti C, Pulmonary strongyloidiasis: The varied clinical presentationsClinical Pulmonary Medicine 2004 11(1):06-07.10.1097/01.cpm.0000107609.50629.6920111672 [Google Scholar] [CrossRef] [PubMed]
[7]. Winkler CF, Snider HL, Strongyloides stercoralis infestationArchives of Internal Medicine 1981 141(5):689-90.10.1001/archinte.1981.003400501350347224755 [Google Scholar] [CrossRef] [PubMed]
[8]. Tefé-Silva C, Machado ER, Faccioli LH, Ramos SG, Hyperinfection syndrome in StrongyloidiasisInCurrent Topics in Tropical Medicine 2012 Mar 16 IntechOpena (https://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0003018)10.5772/28542 [Google Scholar] [CrossRef]
[9]. Greaves D, Coggle S, Pollard C, Aliyu SH, Moore EM, Strongyloides stercoralis infectionBMJ 2013 347:f461010.1136/bmj.f461023900531 [Google Scholar] [CrossRef] [PubMed]
[10]. Olsen A, van Lieshout L, Marti H, Polderman T, Polman K, Steinmann P, Strongyloidiasis- The most neglected of the neglected tropical diseases?Transactions of the Royal Society of Tropical Medicine and Hygiene 2009 103(10):967-72.10.1016/j.trstmh.2009.02.01319328508 [Google Scholar] [CrossRef] [PubMed]