Distant Chest Skin Metastasis in Squamous Cell Carcinoma of Gingivobuccal Sulcus: A Rare Case Report
Venkatesh Anehosur1, Sayali Kiran Desai2, Swetha Acharya3, Niranjan Kumar4
1 Professor, Department of Oral and Maxillofacial Surgery, SDM College of Dental Sciences and Hospital, Shri. Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
2 Resident, Department of Oral and Maxillofacial Surgery, SDM College of Dental Sciences and Hospital, Shri. Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
3 Associate Professor, Department of Oral Pathology, SDM Dental College and Hospital, Shri. Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
4 Vice-Chancellor, Professor and Head, Department of Plastic and Reconstructive Surgery, SDM College of Medical Sciences and Hospital, Shri. Dharmasthala Manjunatheshwara University, Dharwad, Karnataka, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Venkatesh Anehosur, Professor, Department of Oral and Maxillofacial Surgery, SDM Craniofacial Research Centre, SDM College of Dental Sciences and Hospital, Shri. Dharmasthala Manjunatheshwara University, Dharwad-580009, Karnataka, India.
E-mail: venkysdm@gmail.com
The sixth most common cancer in the world is head and neck squamous cell carcinoma with an annual estimated incidence of around 275,000 for oral carcinoma. India has been cited frequently as the country with the highest incidence in the world with over 100,000 cases noted every year. Recurrence of oral squamous cell carcinoma are commonly seen as locoregional failure which is at the primary site or in the neck lymph nodes. Distant metastasis incidence is very uncommon and they are reported in lung, liver, and spinal cord. There are few reported cases of squamous cell carcinoma from oral cavity which has shown metastasis over the chest skin. This case report highlights a rare metastasis which, in spite of good locoregional control with surgery and adjunctive radiotherapy, resulted in a poor outcome.
Metastatic squamous cell carcinoma, Neck dissection, Pectoralis major myocutaneous flap
Case Report
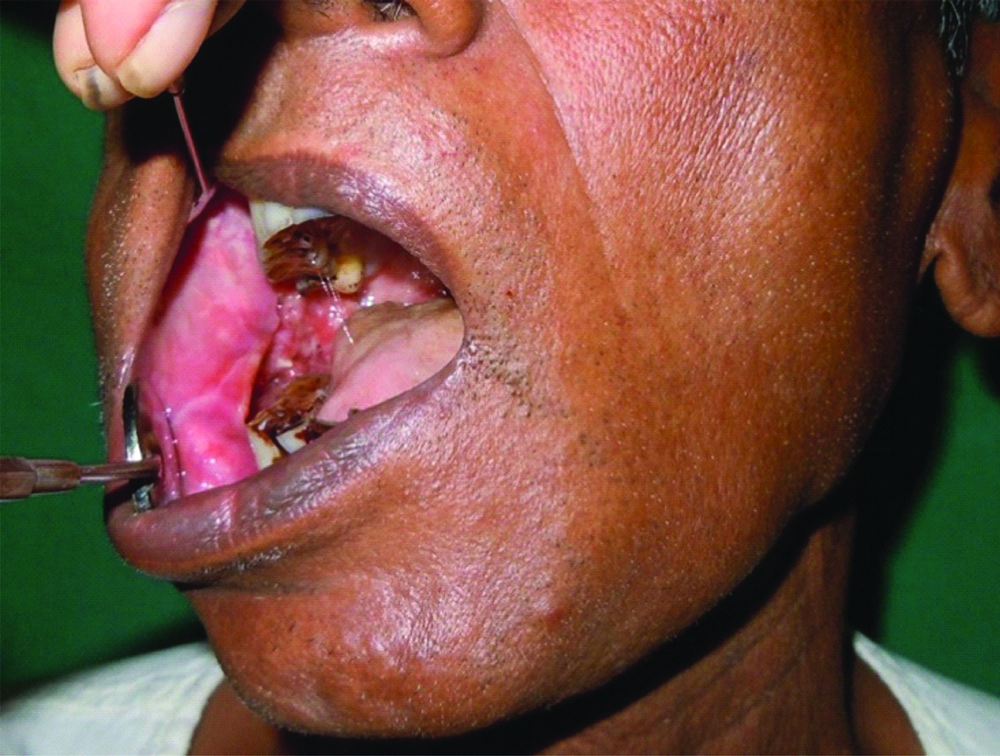
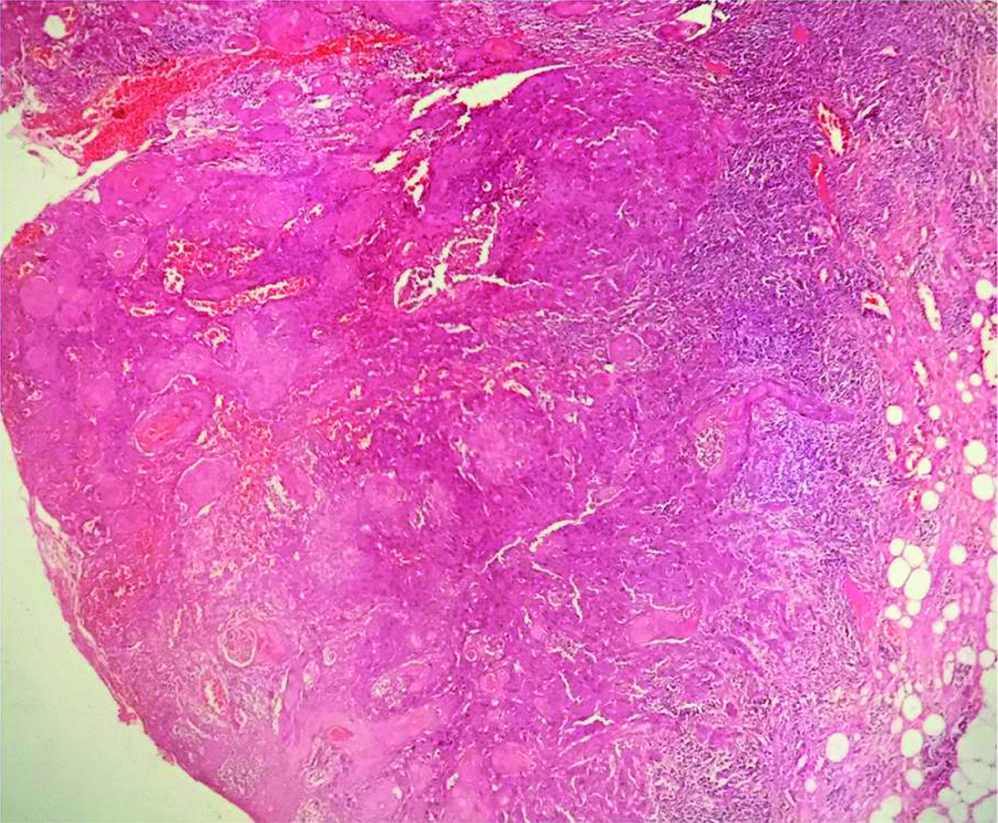
A 58-year-old male patient presented to the Department of Oral and Maxillofacial Surgery, SDM Craniofacial Centre with a chief complaint of pain and swelling in the right side of lower jaw since one month. History of chronic tobacco chewing 10 packs per day for 15 years was noted. History revealed a small growth in the right lower back tooth region which rapidly increased in size. Extraoral examination revealed a diffuse swelling on the right lower third of the face. Single Level IB lymph node was palpable and firm. Mouth opening was adequate with no other relevant abnormality of the Temporo Mandibular Joint (TMJ). On intraoral examination, proliferative growth was noted in the right alveolus extending into the right gingivo-buccal sulcus and retro-molar trigone region [Table/Fig-1]. Routine blood haematological investigations were carried out. Incisional biopsy was reported as well differentiated squamous cell carcinoma [Table/Fig-2]. Histopathologically, tumour cells proliferating into connective tissue stroma in the form of interconnecting islands and strands with keratin pearls were seen. Clinical TNM (Tumour, Node, Metastasis) staging was T4N1Mo. CT scan and ultrasonography of the lesion and the neck was done to determine the extent of the lesion.
Clinical presentation of the primary lesion involving the gingivo-buccal sulcus.
Photomicrograph shows tumour cells proliferating into connective tissue stroma in the form of interconnecting islands and strands. (Incisional Biopsy, H&E,4X).
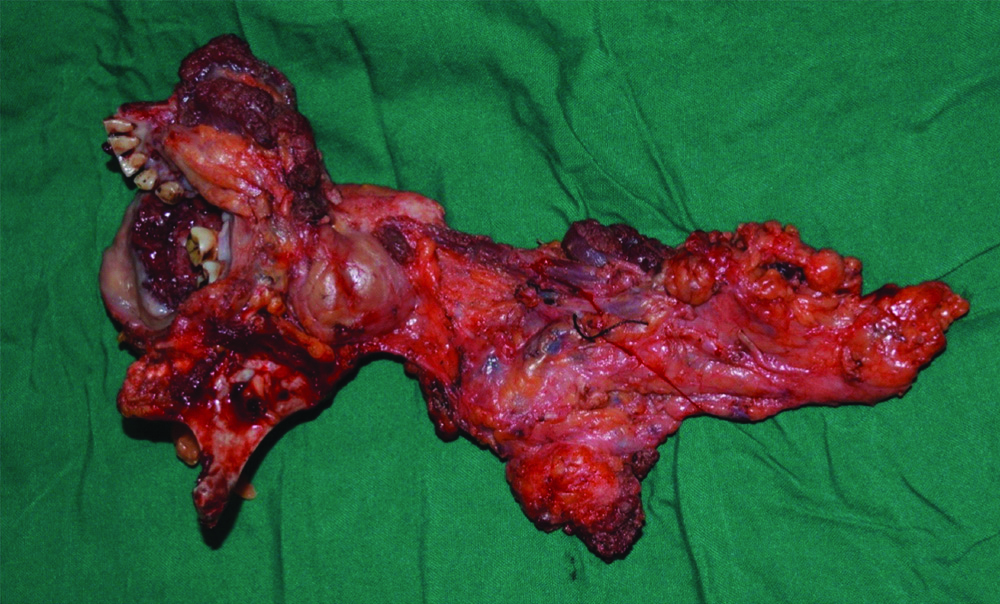
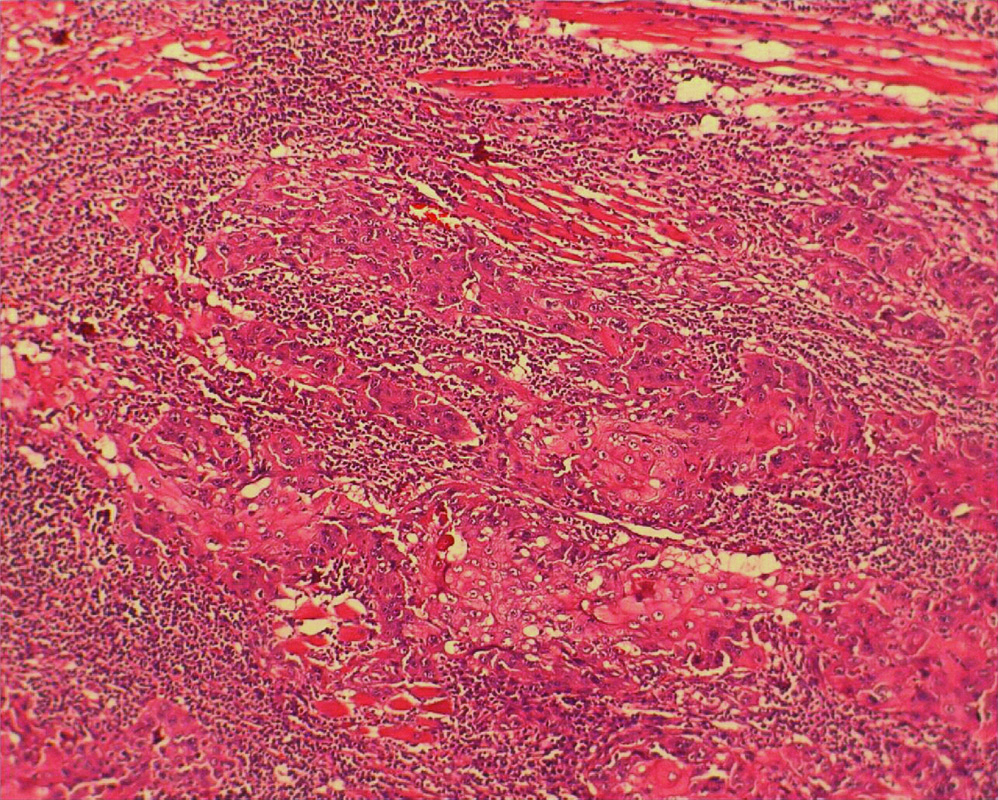
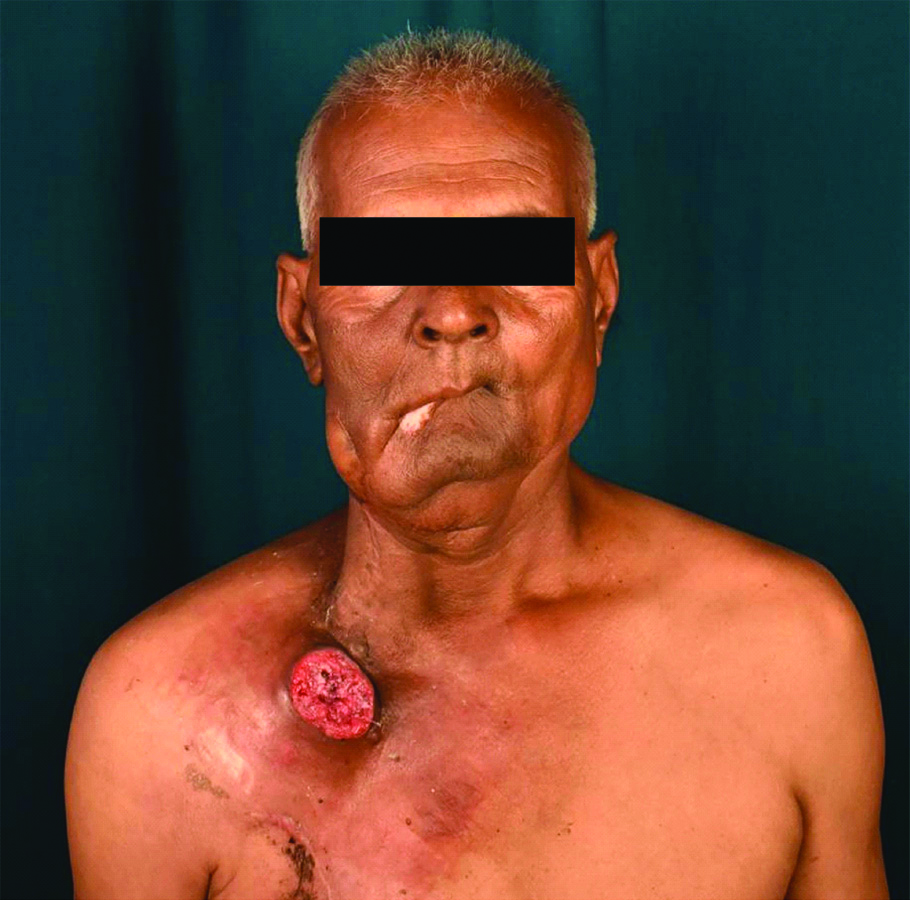
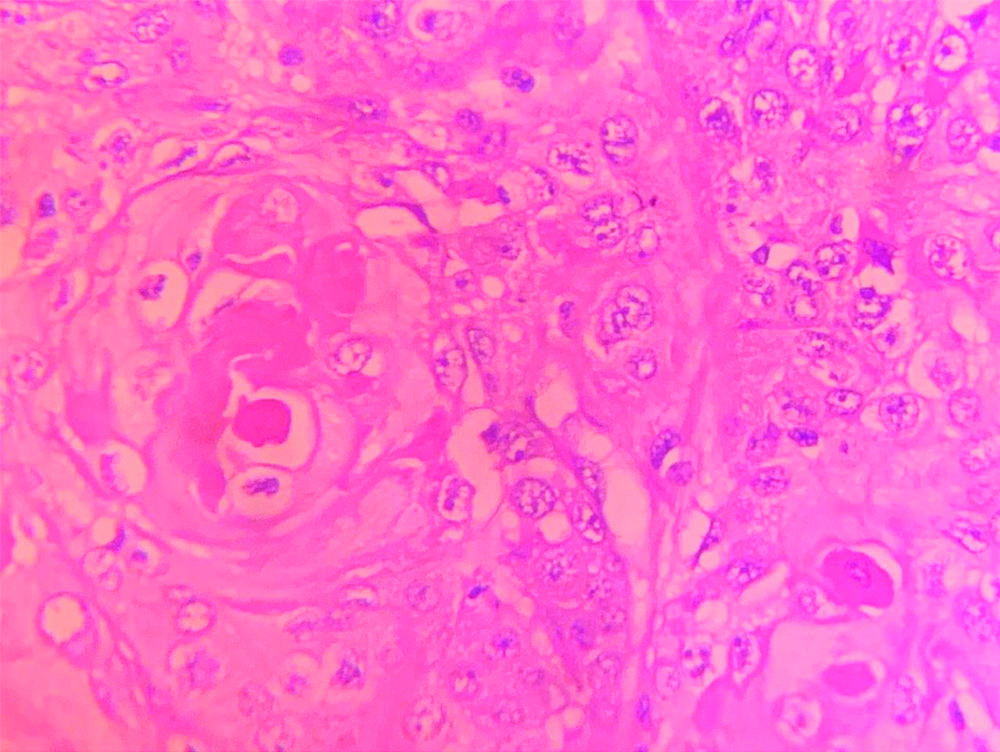
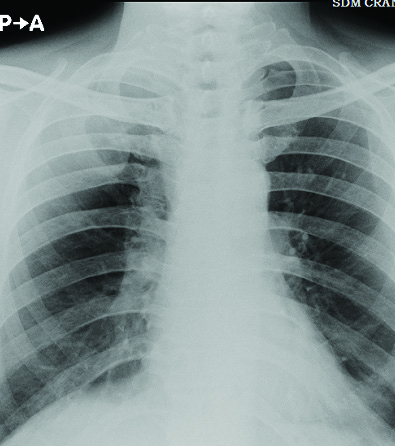
The patient underwent wide excision of the lesion, hemi-mandibulectomy, modified radical neck dissection type II (spinal accessory nerve and internal jugular vein sparing) and reconstruction of the defect was done using pectoralis major myocutaneous flap in July 2018 [Table/Fig-3]. The histopathological report was suggestive of pathological staging T4NoMo with perineural and perivascular invasion positive. Frozen section along with surgical margins were reported negative. Histopathological specimen showed parakeratinised dysplastic epithelium with invasion into the underlying connective tissue stroma. Tumour cells were well differentiated with dysplastic features. The dysplastic epithelial cells were seen in the form of islands of varying sizes with keratin pearl formation. Tumour cells showed infiltration into the muscle fibres and perineural and perivascular tissues. Lymph node specimen showed encapsulation, with increased vascularity and reactive changes [Table/Fig-4]. The patient was then referred for adjunctive radiation therapy and underwent 60 Gy in 30 cycles. The patient was then kept on regular follow-up, which was uneventful for the next three months. Postoperatively, in the fourth month, the patient reported with a complaint of pain and erythematous tender swelling from the right side of the chest and restricted shoulder movements. Clinical examination revealed, a solitary swelling measuring 2×2 cm, round in shape over the right side of the chest, inferior to the clavicle with pus discharge. The swelling was firm in consistency, tender and non-fluctuant on palpation [Table/Fig-5]. Contralateral and axillary lymph nodes were clinically non-palpable. Fine Needle Aspiration Cytology (FNAC) revealed presence of malignant cells. Incisional biopsy of the swelling was done which was suggestive of metastatic well differentiated squamous cell carcinoma. Histopathological examination revealed tumour cells arranged in lobules, nests and sheets with infiltration around the adnexal structures. The cells have moderate cytoplasm, large pleomorphic vesiculate nucleus with prominent nucleoli. Mitotic figures and keratin degradation were also seen [Table/Fig-6]. On the subsequent visit, an ulcer was noted over the same area with everted edges and an indurated base. Postero-anterior chest radiograph was done to rule out metastasis to the lung [Table/Fig-7]. Patient was then advised to undergo palliative chemotherapy and was later lost to follow-up after two months.
Resected primary specimen with neck dissection in toto.
Photomicrograph shows tumour cells in the form of thin strands and cords infiltrating and destructing the muscle (Excisional Biopsy, H&E,20x).
Clinical presentation of distant metastasis over chest skin.
Histopathological image showing tumour cells with keratin degradation. (H&E,40x).
Posteroanterior chest radiograph showing no involvement of lung fields.
Discussion
Oral squamous cell carcinoma is a multifactorial disease and major public health problem in Southeast Asia including India. In India, it tops the cancer types both in incidence and mortality. Advanced stage of the disease is present in 60-80% of the patients. Smokeless tobacco in the form of pan, gutka and supari are the major aetiological factors. The five-year overall survival rate is 70.4%. Human Papilloma Virus is also known to cause genetic damage to the mucosal cells in the form of accumulated mutations leading to rapid and uncontrolled proliferation of cells and metastasis to regional lymph nodes and distant sites [1].
Advances in the treatment of head and neck cancer have resulted in the improvement of locoregional control and increased disease-free survival and overall survival. Most common distant metastatic sites are lung, bone, and liver [2,3]. The uncommon metastasis sites are to kidney, diaphragm, heart, brain, peritoneum. The risk of distant metastasis increases in advanced disease with tumour burden causing multiple neck lymph nodes to be positive. Other factors which influences the distant metastasis are tumour thickness and extracapsular spread. We have reported a case of chest skin metastasis of the donor site flap after primary treatment of squamous cell carcinoma of the gingivo-buccal sulcus.
Distant metastasis from the oral cavity is uncommon. The incidence of distant metastasis for oral squamous cell carcinoma is 15.4% [4]. Out of all patients with distant metastasis, 70% are diagnosed within a year of treatment [4]. Distant metastasis have found to have a dismal prognosis and after curative treatment, the median overall survival is 10 months for metastatic squamous cell carcinoma [4]. The most common site for metastasis are the lungs followed by bone and liver and non-regional lymph nodes. Skin metastasis is uncommon, occurring in only 1% to 2% of the cases. The first case was reported in 1985 [5]. However, the onset is rapid when compared to other sites. Skin of the head, neck and chest are common sites for involvement. The exact mechanism is not completely understood. However, associated risk factors include previous surgery, irradiation, cervical lymph node metastases and extra nodal extension all of which cause aberrant lymphatic drainage. Cervical and thoracic skin metastases may represent locoregional spread through the lymphatic system [4]. There is disruption of the lymphatic drainage which resulted in lymphatic dissemination of malignant cells below the clavicle. But skin metastases occurring at distant sites arise through hematogenous spread [6]. There is a hypothesis that distant metastasis in patients with good locoregional control may occur due to subclinical deposits of the tumour that have already occurred at distant sites prior to locoregional management which manifested in the post-treatment period [7]. This hypothesis has led to setting up of extensive pre-treatment investigative protocols like whole body scans, PET and SPECT scans, which are not feasible in all the regions of world.
Kroll SS et al., reported one incidence of tumour dissemination to the donor site following a study on a large series of 617 reconstructions to rule out hypothesis of surgical stress causing immunosuppression and leading distant metastasis [8]. The biology of cancer metastasis regards to the tumour seeding and soil is not proven with clinical evidence [9]. In present case, we were not able to conclude the reasons for this distant metastasis as one of the possibility of tumour seeding through the glove since the resection and reconstruction surgeons were different. Other rare sites for metastatic spread include the pleura, adrenal gland, soft tissues, pancreas, brain and spleen. In a study by Berger DS and Fletcher GH, the survival was found to be three months following the involvement of skin [10]. In this case too, the patient’s general condition deteriorated rapidly and succumbed to the disease within two months.
Conclusion(s)
This case report adds to the already reported cases of distant metastasis which will increase to the knowledge of surgeons about possibility of such rare expression of the oral squamous cell carcinoma in the post-operative period.
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Sep 26, 2019
Manual Googling: Oct 30, 2019
iThenticate Software: Jan 23, 2020 (14%)
[1]. Jayade BV, Bhat HKH, Kundalswamy G, Anehosur V, Kumar N, Gunari PP, Dismal outcome of oral squamous cell carcinoma after successful loco-regional control: Distant metastasis to rare sitesJ Oral Maxillofac Surg Med Pathol 2014 26(3):324-30.10.1016/j.ajoms.2012.12.010 [Google Scholar] [CrossRef]
[2]. Ferlito A, Shaha AR, Silver CE, Rinaldo A, Mondin V, Incidence and sites of distant metastases from head and neck cancerORL J Otorhinolaryngol Relat Spec 2001 63(4):202-07.10.1159/00005574011408812 [Google Scholar] [CrossRef] [PubMed]
[3]. Sakamoto Y, Matsushita Y, Yamada S, Yanamoto S, Shiraishi T, Asahina I, Risk factors of distant metastasis in patients with squamous cell carcinoma of the oral cavityOral Surg Oral Med Oral Pathol Oral Radiol 2016 121(5):474-80.10.1016/j.oooo.2015.11.02227068308 [Google Scholar] [CrossRef] [PubMed]
[4]. Duprez F, Berwouts D, Neve WD, Bonte K, Boterberg T, Deron P, Distant metastases in head and neck cancerHead and Neck 2017 39(9):1733-43.10.1002/hed.2468728650113 [Google Scholar] [CrossRef] [PubMed]
[5]. Schultz BM, Schwartz RA, Hypopharyngeal squamous cell carcinoma metastatic to skinJ Am Acad Dermatol 1985 12(1 Pt 2):169-72.10.1016/S0190-9622(85)80009-8 [Google Scholar] [CrossRef]
[6]. Pitman KT, Johnson JT, Skin metastases from head and neck squamous cell carcinoma: Incidence and impactHead Neck 1999 21(6):560-65.10.1002/(SICI)1097-0347(199909)21:6<560::AID-HED10>3.0.CO;2-Q [Google Scholar] [CrossRef]
[7]. Suzuki H, Hasegawa Y, Terada A, Hyodo I, Nakashima T, Nishio M, FDG- PET predicts survival and distant metastasis in oral squamous cell carcinomaOral Oncol 2009 45(7):569-73.10.1016/j.oraloncology.2008.07.00918804407 [Google Scholar] [CrossRef] [PubMed]
[8]. Kroll SS, Tavollali M, Castello-Sendra J, Pollock RE, Risk of dissemination of cancer to flap donor sites during immediate reconstructive surgeryAnn Plast Surg 1994 33(6):573-75.10.1097/00000637-199412000-000017880044 [Google Scholar] [CrossRef] [PubMed]
[9]. Fidler IJ, Balch CM, The biology of cancer metastases and implications for therapyCurr Probl Surg 1987 24(3):129-209.10.1016/0011-3840(87)90002-5 [Google Scholar] [CrossRef]
[10]. Berger DS, Fletcher GH, Distant metastases following local control of squamous-cell carcinoma of the nasopharynx, tonsillar fossa, and base of the tongueRadiology 1971 100(1):141-43.10.1148/100.1.1415147022 [Google Scholar] [CrossRef] [PubMed]