Anterior Abdominal Wall Lymphangioma in a Child: Challenging Reconstruction at an Unusual Location
Amit Kumar Sinha1, Amit Kumar2, Rashi3, Bindey Kumar4, Prem Kumar5
1 Assistant Professor, Department of Paediatric Surgery, AIIMS, Patna, Bihar, India.
2 Senior Resident, Department of Paediatric Surgery, AIIMS, Patna, Bihar, India.
3 Senior Resident, Department of Paediatric Surgery, AIIMS, Patna, Bihar, India.
4 Professor, Department of Radiology, AIIMS, Patna, Bihar, India.
5 Professor, Department of Paediatric Surgery, AIIMS, Patna, Bihar, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Amit Kumar Sinha, Phulwarisarif, Patna, Bihar, India.
E-mail: dr_amits@yahoo.com
Lymphangiomas are rare benign hamartoma resulting from sequestered lymphatic channels. They mostly occur at head, neck and axilla. Abdominal wall is an unusual location for the lesion. This is a report of abdominal wall lymphangioma in a three year eleven-month-old male child, having painless abdominal wall swelling, which presented a challenge at diagnosis as well as surgical reconstruction while treating the child. It is important to consider lymphangioma as an important differential diagnosis during evaluation of any abdominal wall swelling.
Lymphatic malformation, Sclerotherapy, Transillumination test
Case Report
A three year eleven-month-old male child, with an uneventful perinatal history, reported to the Outpatient Department with complaints of swelling over right side of abdominal wall since birth. The swelling was painless and gradually increasing in size. Apart from cosmetic disfigurement, patient was not having any discomfort. They consulted a local physician at village level who referred the patient to us.
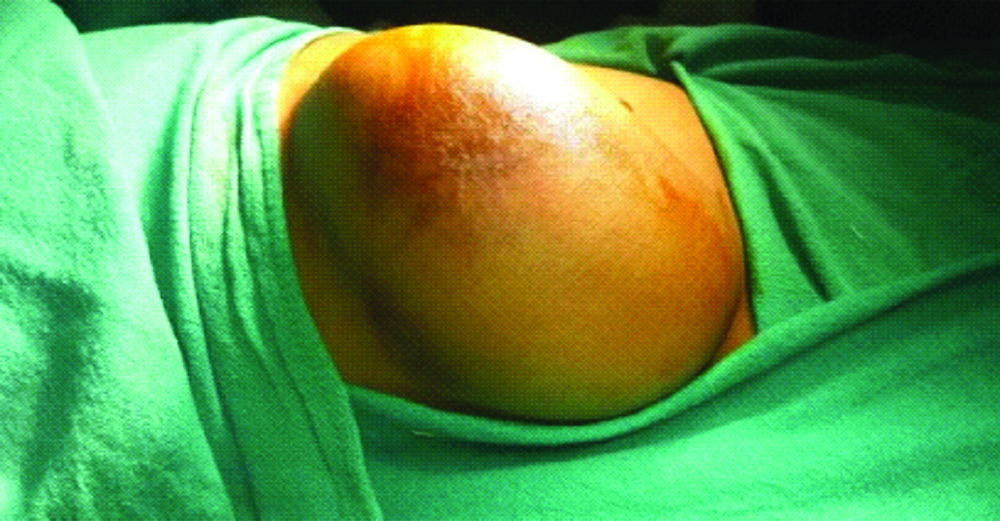
On examination, a solitary swelling of almost 15×12 cm in size, non-tender, and cystic in consistency extending over right hypochondrium and lumbar region was seen. The swelling was non-compressible, non-tender, slightly mobile, non-transilluminant and doubtfully originated from abdominal parietes. The skin over the swelling had venous prominence and a patch of discolouration [Table/Fig-1]. Bowel and bladder habits were regular.
Abdominal wall lymphangioma.
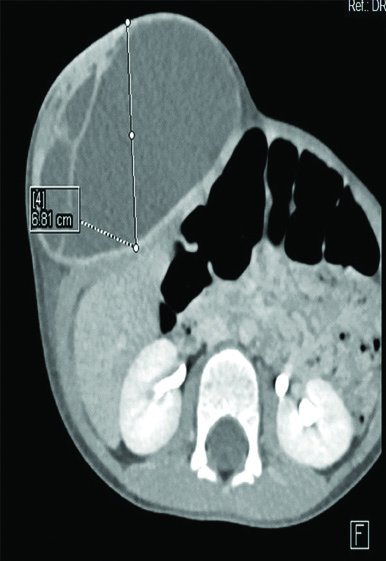
Initially, we were perplexed with the more usual diagnosis, like desmoid tumour and lipoma. Ultrasound scan suggested a lesion confined to abdominal parietes, possibly lymphangioma. CECT scan was done that revealed it to be a large ovoid cystic lesion in subcutaneous plane, opposite D11-L3 vertebral body with few septae within. The peripheral wall and internal septae of swelling showed contrast enhancement. There was no evidence of calcification or solid component or fat density seen within. Underlying muscles appeared normal and indented by the lesion [Table/Fig-2]. With provisional diagnosis of lymphangioma or antibioma, excision of mass was planned.
CECT scan showing cystic swelling with internal septation and contrast enhancement of cyst wall and septae.
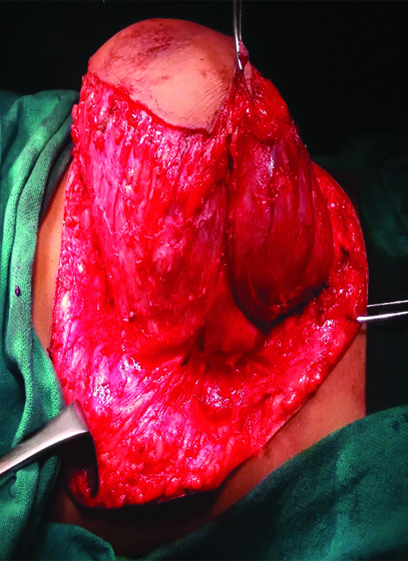
On exploration, the swelling was confined superficial to muscles, but the superficial muscle layer was densely adhered with it [Table/Fig-3]. By meticulous dissection, cystic swelling was removed without rupture. Superficial muscle layer was repaired. Specimen was sent for histo-pathological evaluation. Postoperative period was uneventful. Patient was discharged on 4th postoperative day. Histopathology of the specimen suggested lymphatic malformation of abdominal wall.
Abdominal wall lymphangioma on exploration showing densely adhered superficial muscle layer.
Discussion
Lymphangiomas are considered as benign tumours of abnormal lymphatic channel of embryonic origin. The sequestered lymphatics mostly fail to communicate with the surrounding lymphatics or venous system [1]. Most of these are present at birth and almost 90% are diagnosed before two years of age [2]. They cause aesthetic and pressure symptoms. According to the size of lymphatics involved, capillary, cavernous or cystic varieties of lymphatic malformations have been quoted. Common sites of occurrence include head and neck (75%), axilla (15%) and sometimes, chest wall, mediastinum, abdominal wall, retroperitoneal areas [3,4].
Abdominal wall is a rare location for lymphangiomas in a child. Common differential diagnosis include desmoid tumour, lipoma, abscess, haematoma, hydatid cyst etc. In this report, a three year eleven-month-old child had an abdominal wall lymphangioma, progressively increasing in size since birth and producing heaviness and cosmetic disfigurement.
Lymphangiomas are rare congenital malformations of the lymphatic system. These appear to be ubiquitous in terms of appearance in the body. By searching the available literature, abdominal wall lymphangioma is still unusual to find [5]. Diagnosis poses a challenge mostly due to this very reason and also occurrence of other more common lesions at the site [6]. Spontaneous or traumatic haemorrhage may conceal the characteristic transillumination test and explain the skin discolouration. Although radiological investigations like ultrasonic scan, computed tomography scan, magnetic resonance imaging help to delineate the lesion from surrounding structures, histopathology is confirmatory. Spontaneous regressions of lymphangiomas have been evidenced in head and neck, but not in the abdominal wall [7].
Lymphangiomas can be treated by injection sclerotherapy or surgery [7]. Bleomycin or OK-432 can be used as sclerosant depending upon number and size of individual cyst. Due to unavailbility of histological diagnosis in injection sclerotherapy method, surgery is still advocated as best modality to treat these lesions, though repair of the wall after excising a massive lesion remains a challenge [8]. Injection sclerotherapy can be considered in case of a recurrence [9]. However, the treatment planning may need to be standardised.
Conclusion(s)
Abdominal wall lymphangiomas should be considered in the differentials, while considering an abdominal wall swelling. With this paper, we hope to contribute a bit to the better understanding of this unusual location and presentation.
Author Declaration:
Financial or Other Competing Interests: None
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Jul 02, 2019
Manual Googling: Dec 18, 2019
iThenticate Software: Jan 11, 2020 (11%)
[1]. Vogel AM, Warner BW, Toward an understanding of lymphatic malformationsGastroenterology and Hepatology 2013 9(3):195-96. [Google Scholar]
[2]. Bhavsar T, Saeed-Vafa D, Harbison S, Innis S, Retroperitoneal cystic lymphangioma in an adult: A case report and review of the literatureWorld J Gastrointest Pathophysiol 2010 1(5):171-76.10.4291/wjgp.v1.i5.17121607159 [Google Scholar] [CrossRef] [PubMed]
[3]. Kosir MA, Sonnino RE, Gauderer MW, Pediatric abdominal lymphangiomas: A plea for early recognitionJ Pediatr Surg 1991 26(11):1309-13.10.1016/0022-3468(91)90607-U [Google Scholar] [CrossRef]
[4]. Bal HS, Jehangir S, Kurian JJ, A giant lymphangioma of the body wall in a child: A heavy companionBMJ Case Reports 2014 doi: 10:1136/bcr-2014-20621610.1136/bcr-2014-20621625427931 [Google Scholar] [CrossRef] [PubMed]
[5]. David EA, Marshall MB, Review of chest wall tumours: A diagnostic, therapeutic and reconstructive challengeSemin Plast Surg 2011 25(1):16-24.10.1055/s-0031-127516722294939 [Google Scholar] [CrossRef] [PubMed]
[6]. Ardenghy M, Miura Y, Kovach R, Hochberg J, Cystic hygroma of the chest wall: A rare conditionAnn Plast Surg 1996 37(2):211-13.10.1097/00000637-199608000-000188863986 [Google Scholar] [CrossRef] [PubMed]
[7]. Okazaki T, Iwatani S, Yanai T, Kobayashi H, Kato Y, Marusasa T, Treatment of lymphangiomas in children: Our experience of 128 casesJ Pediatric Surg 2007 42(2):386-89.10.1016/j.jpedsurg.2006.10.01217270554 [Google Scholar] [CrossRef] [PubMed]
[8]. Yang J, Xu CY, Giant lymphangiomas of the abdomen and thighEuropean Journal of Vascular and Endovascular Surgery 2016 52(5):68810.1016/j.ejvs.2016.08.00727595521 [Google Scholar] [CrossRef] [PubMed]
[9]. Galofre M, Judd ES, Perez PE, Harrison EG Jr, Results of surgical treatment of cystic hygromaSurg Gynecol Obstet 1962 115:319-26.10.1097/00006534-196212000-0002613896490 [Google Scholar] [CrossRef] [PubMed]