Response to Infliximab Biosimilar in a Case of Reactive Arthritis: Our Experience
Amruta Deepak Morey1, Bhushan Sevakram Madke2, Adarsh Lata Singh3, Sudhir Singh4, Sandeep Kulkarni5
1 Junior Resident, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
2 Professor and Head, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
3 Professor, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
4 Assistant Professor, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
5 Assistant Professor, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Bhushan Sevakram Madke, Professor and Head, Department of Dermatology, Venereology and Leprosy, Jawaharlal Nehru Medical College, Datta Meghe Institute of Medical Sciences, Wardha, Sawangi, Maharashtra, India.
E-mail: drbhushan81@gmail.com
Sir,
Reactive arthritis syndrome consists of a triad of asymmetric oligoarthritis, urethritis, and conjunctivitis with characteristic mucocutaneous lesions. The role of Tumour Necrosis Factor alpha (TNF α) in pathogenesis of various rheumatic diseases suggests that it may play a role in other autoimmune diseases such as Reactive arthritis [1]. Treatment includes NSAIDs, DMARDs (methotrexate or sulfasalazine), steroids and antibiotics. Biological and biosimilar drugs are new therapeutic agents used in management of spondylo-arthropathies. Considering paucity of data on use of Infliximab biosimilar, we hereby report successful use of Infliximab biosimilar in a case of reactive arthritis.
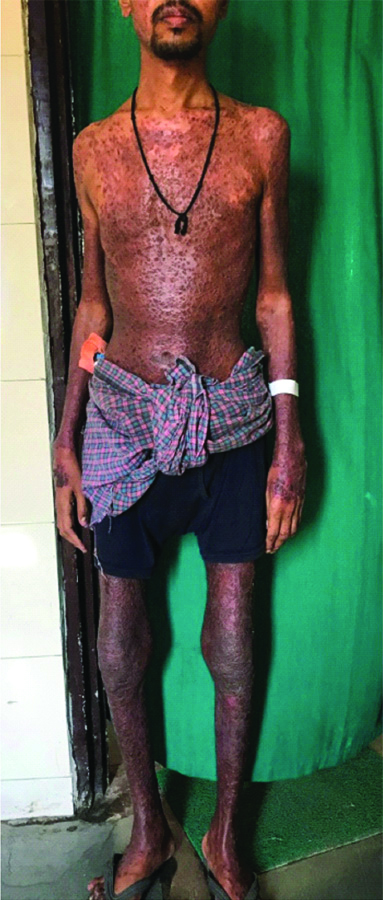
A 36-year-old unmarried male, teacher by profession presented to the OPD with diffuse, dusky, erythematous, scaly plaques involving trunk, extremities, scalp with disabling joint pain involving knees, shoulders and ankles with intermittent history of fever and myalgia for past ten years. The patient experienced partial remissions and exacerbations; however the condition worsened in the last six months. He was previously treated with oral methotrexate, acitretin, cyclosporine and sulfasalazine for varying duration in the past six years with minimal improvement of joint pain and skin lesions. Cutaneous examination showed crusted psoriasiform papules and plaques on different body parts [Table/Fig-1].
Clinical photograph showing characteristic crusted papules and plaques along with swollen knee joints taken at baseline before Infliximab infusion.
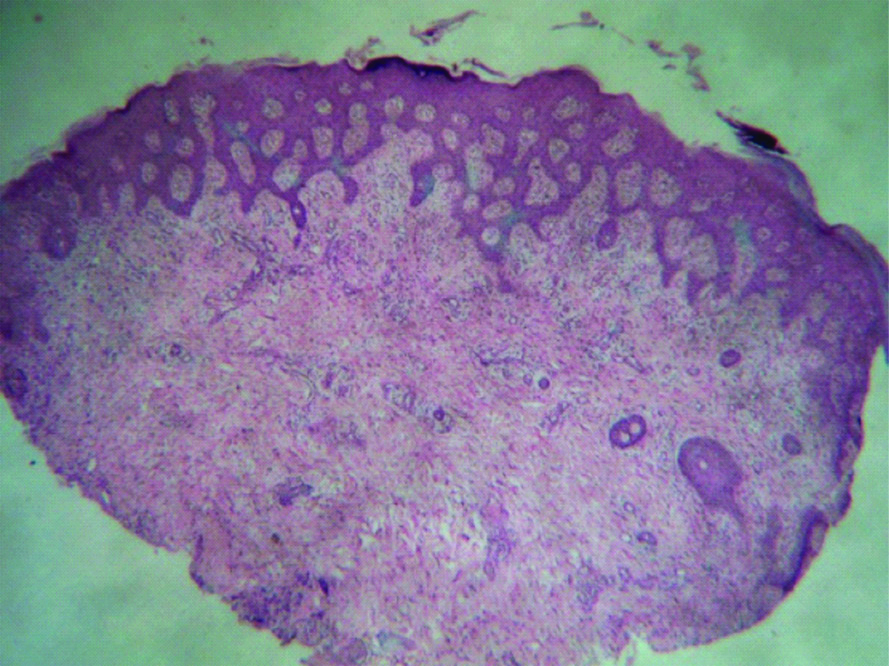
Complete blood count revealed leucocytosis-16500/cumm, granulocytosis (80%), raised ESR (80 mm). C-reactive protein titre was raised (48 mg/dL), serology for RA factor, hepatitis B, C virus and HIV were non-reactive. Tuberculin skin test was negative. Skin biopsy showed features of psoriasiform reaction pattern with neutrophil collection in stratum corneum [Table/Fig-2].
H&E (4x) section showing psoriasiform hyperplasia of epidermis with mild perivascular lymphocytic infiltrate.
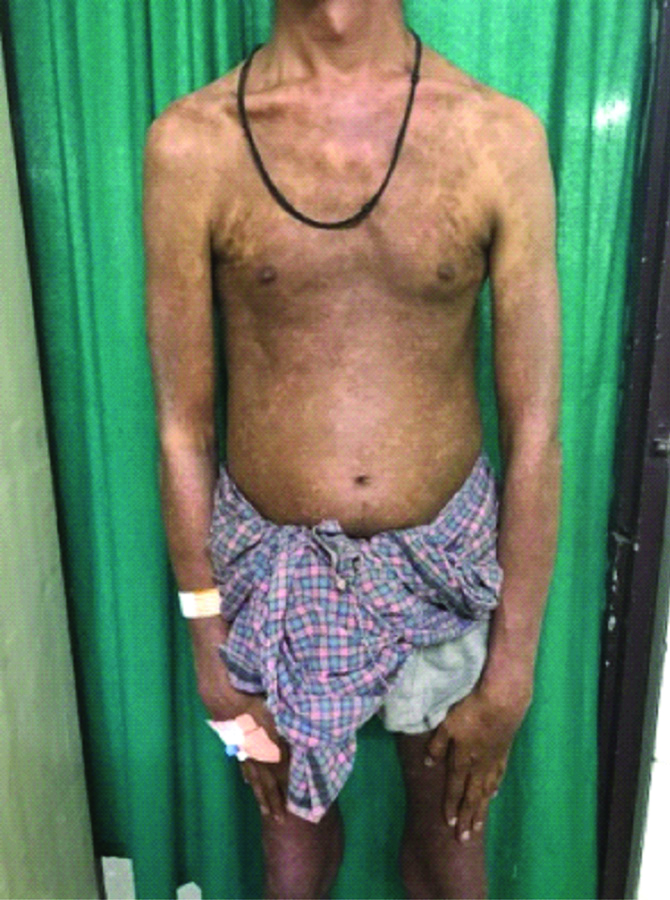
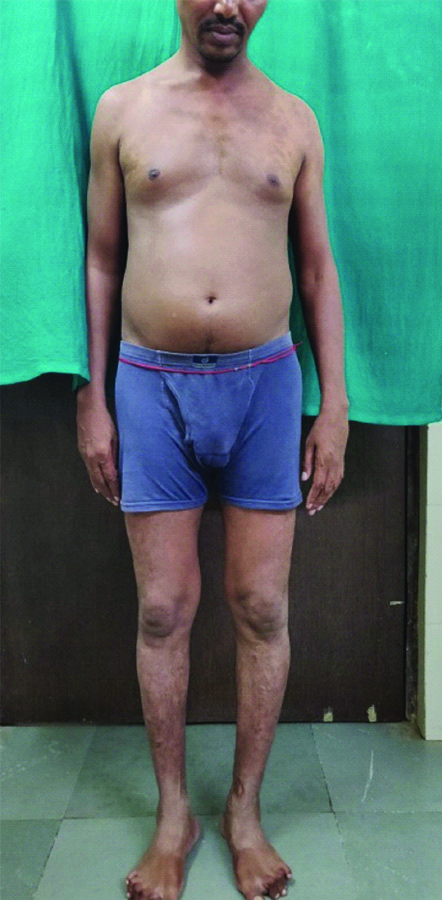
Considering recalcitrant nature of the condition, we planned to give Infliximab biosimilar to our patient. Patient was administered 100mg of Infliximab biosimilar @ 3mg/kg body weight under intensive care in June 2018. On day 7 post-infusion, patient noticed significant improvement in joint pain and resolution of skin lesions. Patient received second dose in August 2018 [Table/Fig-3] and third dose in October 2018 [Table/Fig-4]. After receiving three doses, patient reported 90% improvement of skin lesions and complete subsidence of joint pain and was able to perform full range of joint mobility.
Clinical photograph showing partial resolution of skin lesions after two months of first infusion.
Clinical photograph showing near total resolution of skin lesions along with resolution of swelling over knee joints after four months of first infusion.
Infliximab is a chimeric monoclonal antibody having action against inflammatory mediator TNF α. TNF-α being a pro-inflammatory cytokine, induces release of interleukin 1, 6 and enhances leukocytic migration. Elevated serum levels of TNF-α have been reported in patients with reactive arthritis [2]. Infliximab binds to and inhibits the binding of cytokine TNF-α to its receptors thereby neutralising its action [3].
Some studies in support of use of Infliximab are summarised in [Table/Fig-5] [1,3-5].
Findings of present study and published studies [1,3-5].
| Author | Intervention | Outcome |
|---|
| Gill H and Majithia V [1] | A patient with joints and skin complaints was given Infliximab 200 mg IV at 0, 2, 6, and 14 weeks | Complete resolution of arthritis and skin lesions within six weeks. |
| Gaylis N [3] | Infliximab 300 mg IV at weeks 0,2,6 and every 6 to 7 weeks thereafter in an HIV positive patient with Reiter’s syndrome | All complaints resolved within six months and patient’s viral titre remained below 400 Qn:US copies/mL as he continued anti-retroviral therapy. |
| Seppanen KO et al., [4] | Two patients with reactive arthritis were given Infliximab IV at 0,2,6 weeks. | Good response in acute phase. |
| Meyer A et al., [5] | Ten refractory cases of reactive arthritis received TNFá therapy | Nine patients showed rapid improvement in joints and skin manifestations with steroid sparing effect. |
| Present case | A patient with joint pain and psoriasiform rash was given infliximab 100 mg IV in two doses at an interval of one month | Complete resolution of skin lesions and significant improvement in joint pain and mobility. |
The promising and rapid results of Infliximab biosimilar as seen in our patient emphasises its exploration for wider use in the treatment of refractory cases of reactive arthritis.
Author Declaration:
Financial or Other Competing Interests: No
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
Plagiarism Checking Methods: [Jain H et al.]
Plagiarism X-checker: Sep 27, 2019
Manual Googling: Nov 08, 2019
iThenticate Software: Nov 20, 2019 (9%)
[1]. Gill H, Majithia V, Successful use of infliximab in the treatment of Reiter’s syndrome: A case report and discussionClin Rheumatol 2007 27(1):121-23.10.1007/s10067-007-0692-017643185 [Google Scholar] [CrossRef] [PubMed]
[2]. Carter JD, Treating reactive arthritis: Insights for the clinicianTher Adv Musculoskelet Dis 2010 2(1):45-54.10.1177/1759720X0935750822870437 [Google Scholar] [CrossRef] [PubMed]
[3]. Gaylis N, Infliximab in the treatment of an HIV positive patient with Reiter’s syndromeJ Rheumatol 2003 30(2):407-11. [Google Scholar]
[4]. Seppänen KO, Niinisalo H, Korpilähde T, Virolainen J, Treatment of reactive arthritis with infliximabScand J Rheumatol 2003 32(2):122-24.10.1080/0300974031000015712737333 [Google Scholar] [CrossRef] [PubMed]
[5]. Meyer A, Chatelus E, Wendling D, Berthelot J-M, Dernis E, Houvenagel E, Safety and efficacy of anti-tumor necrosis factor α therapy in ten patients with recent-onset refractory reactive arthritisArthritis Rheum 2011 63(5):1274-80.10.1002/art.3027221538314 [Google Scholar] [CrossRef] [PubMed]