Detection of Cup-like Blasts in Acute Myeloid Leukaemia
Supriya Mehrotra1, Priyanka Singh2, Rajesh Kumar Srivastava3, AK Kapoor4
1 Pathologist, Department of Pathology, RML Mehrotra Pathology Ltd., Lucknow, Uttar Pradesh, India.
2 Pathologist, Department of Pathology, RML Mehrotra Pathology Ltd., Lucknow, Uttar Pradesh, India.
3 Technologist, Department of Pathology, RML Mehrotra Pathology Ltd., Lucknow, Uttar Pradesh, India.
4 Pathologist, Department of Pathology, RML Mehrotra Pathology Ltd., Lucknow, Uttar Pradesh, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: AK Kapoor, Pathologist, Department of Pathology, RML Mehrotra Pathology Ltd., Nirala Nagar, Lucknow-226020, Uttar Pradesh, India.
E-mail: drashokkapoor2016@gmail.com
A 59-year-old female, presented with generalised weakness for several months. Her complete blood cell count revealed mild anaemia moderate thrombocytopenia with hyperleukocytosis. Differential leucocyte count revealed large number of blast cells (88%), with most of the blast cells having cup-like invaginations of nuclear membrane (giving it a fish-mouth-like nuclear appearance). Using a strict criteria of ≥10% blasts with nuclear invaginations and occupying ≥25% of nuclear area, most of immature cells were cup-like blasts. The blast cells had fine chromatin, high nuclear cytoplasmic ratio and 1 to 2 nucleoli. Cytoplasm of blast cells did not contain granules or Auer rods. Morphologically, the blast cells were suggestive of Acute Myeloid Leukaemia (AML) and the M1 subtype (FAB classification). Flow cytometric examination revealed moderate positivity for CD33 and mild positivity for CD11c, CD117 and CD45 antigens. Moderate expression of CD33 suggested aberrant expression on myeloid cells and Nucleophosmin (NPM 1) mutation.
CD33++, Cyto MPO+, CD11c+, CD117+, CD45+, Cup-like myeloblasts
Case Report
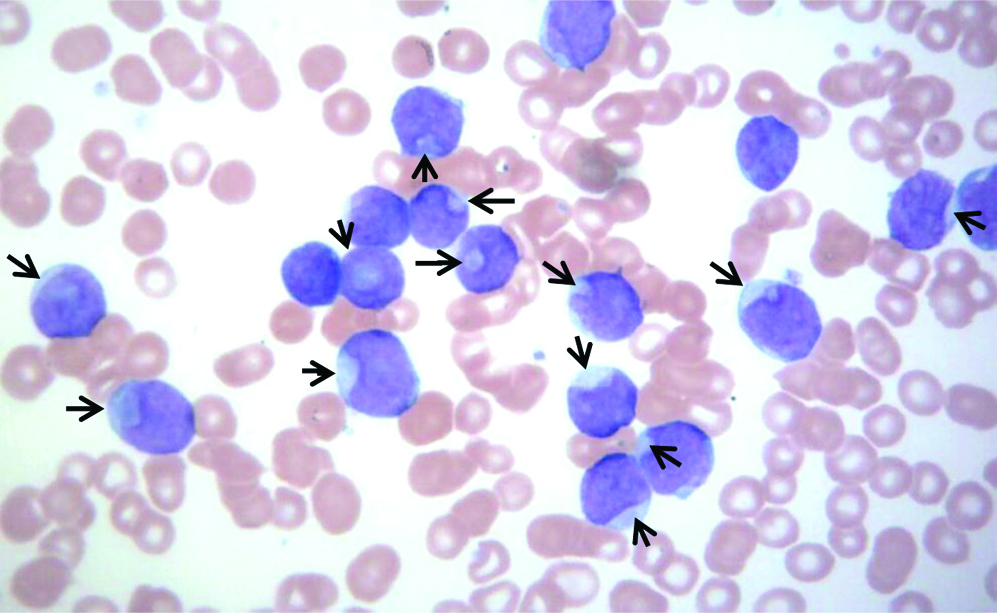
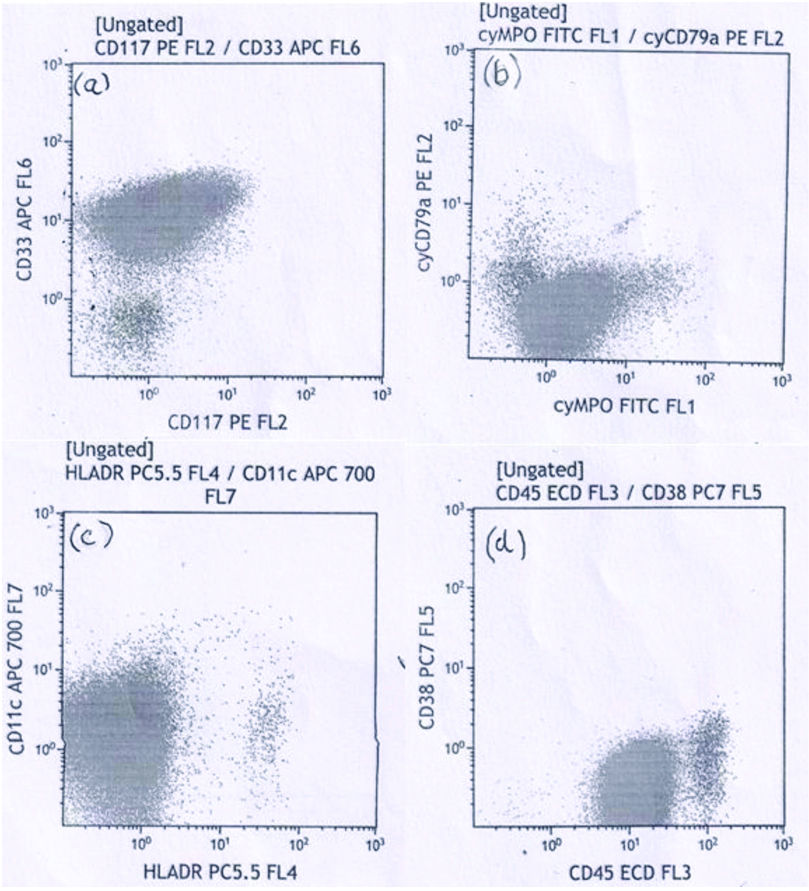
A 59-year-old female presented with chief complaint of generalised weakness for last several months. Relevant past medical history was not available. Haemoglobin concentration was 104 gm/L, platelets moderately reduced (51×109/L) and total WBC severely raised (179×109/L). Differential leucocyte count showed blast cells 88%, neutrophils 02%, lymphocytes 08% and monocytes 02%. Blast cells had high nuclear cytoplasmic ratio, the nucleus of the blasts had fine nuclear chromatin and 1 to 2 prominent nucleoli. Using a strict criteria of ≥10% blasts with nuclear invagination and occupying ≥25% of nuclear area, several of the immature cells were cup-like blasts-CLB [Table/Fig-1]. Neither Auer rods nor cytoplasmic granules were seen. These findings were suggestive of acute myeloid leukaemia (M1 in FAB classification). Flow cytometric examination revealed moderate positivity (2+) of blasts for CD33 antigen and dim expression (1+) of cytoplasmic myeloperoxidase (cyto MPO); CD11c, CD117 and CD45 antigens [Table/Fig-2a-d]. In addition, blast cells showed negative reaction with anti-CD34, anti-CD64, anti-CD13 and anti-CD36. Low-to-absent expression of HLA-DR antigen was seen on blast cells [Table/Fig-2c].
Peripheral blood smear of the patient. Arrow points towards the cups of blast cells; these cells were called as Cup-like blasts (CLB). About 70% cells were CLB. Another 18% cells were blasts without nuclear indentations (Leishman stain × 400).
a) Flow cytometry showed moderate (2+) expression of CD33 and dim (1+) expression of CD117; b) shows expression of Cytoplasmic MPO; c) Tumour cells showed mild reactivity (1+) with anti-CD11c. Low-to-absent expression (-) of HLA DR is seen; d) anti-CD45 antibody did not stain blast cells.
Discussion
Cup-like blasts (CLB) have been described in a subset of AML with NPM1 and FLT3-ITD mutations. A total of 6 cases of CLB-AML were encountered at Kyoto University Hospital [1]. Cup-like AML has been correlated with female preponderance, high myeloperoxidase positivity, low-or-absent CD34 and HLA-DR expression [2]. In addition, CLB-AML patients present with higher blast counts compared to non-CLB-AML [2]. Moreover, CLB-AML patients with NPM1 mutation may have excellent prognosis with survival of 60% patients around 5 years [1].
Most important finding of current case was the CLB in venous blood smear. Several cases of CLB with BCR-ABL1-positive AML associated with Nucleophosmin Mutation (NPM1) have been reported [1,2]. Rarely, CLB may be found in Philadelphia chromosome positive and FLT3-ITD negative ALL [3]. CLB may be found both in non-monocytic and monocytic cases of AML. M1 subtype is known to have better prognosis when compared with M3 to M5 subtypes. Furthermore, M1 subtype with or without CLB may not affect the prognosis of AML. In addition, CLB are known to be associated with high percentage of fms-like tyrosine kinase-3 gene (FLT3-ITD) and NPM1 mutations [2,4,5]. Identification of genetic defects may be important in diagnosis and treatment of AML. For example, inhibitors of a mutated fms-like FLT3 may be useful in the treatment of AML with CLB [2]. In addition, cup-like AML may be associated with Isocitrate Dehydrogenase 1 (IDH1) gene mutation. In NPM1-mutated AML, NPM protein is localised in the cytoplasm of leukaemic cells instead of nucleoli resulting in nuclear indentations of blast cells [2]. In addition, mitochondria are concentrated in the indentations [6]. On the contrary, in NPM1 unmutated AML, NPM protein is restricted to nucleus [2]. Poor prognostic impact of FLT-ITD has been recognised and NPM1 gene with relatively more favourable prognosis has been acknowledged [2]. Moderate (2+) expression of CD33 suggested aberrant expression on myeloid cells and nucleophosmin mutation. Weak or dim reaction (1+) with CD45 antigen further suggested origin of blast cells from leucocytes. Mild reaction with anti-CD117 suggested origin of blast cells from a haemopoietic progenitor cell. Weak reaction with anti-CD11c was also detected on blast cells; CD11c is found on NK cells, monocyte, macrophages and neutrophils. Detection of dim cytoplasmic MPO reactivity suggested weak granulocytic differentiation. In addition, cytoplasmic MPO reactivity may enhance chemosensitivity through generation of reactive oxygen species and nitration of proteins favouring prognosis. Another important feature of current case was the detection of hyperleukocytosis (leucocytes >100000 cells/mm3). Complications, for example, leukostasis, tumour cell lysis syndrome and disseminated intravascular coagulation may develop resulting in higher risk of death. Moreover, these patients have higher probability of relapse. Further, high leucocyte counts indicate high proliferation rate [7]. Due to early death, Hyperleukocytosis (HL) in AML might be considered as a medical emergency requiring early treatment with hydroxyurea and leukopharesis. In addition, platelets are maintained >50,000/mm3 by platelet transfusions. Another factor responsible for HL may be disruption of normal haematopoietic cell adhesion resulting in depressed affinity for bone marrow [7,8]. Further, other haematological problems in index case were moderate thrombocytopenia and moderate anaemia. Moderate anaemia might have resulted in generalised weakness. Cup-like morphology of myeloblasts in AML is rare and its association with NPM1 mutation may result in excellent outcome.
Conclusion
Present case deals with haematological findings of a patient with generalised weakness. Total WBC count was severely raised. Differential leucocyte count revealed 88% blast cells. Nuclei of blast cells had fish-mouth-like indentations. Flow cytometry revealed moderate positivity (2+) of blast cells for CD33 antigen and negative reaction with CD34 and HLA-DR. Blast cells had fine chromatin and 1 to 2 nucleoli. She was diagnosed as a case of AML with peculiar nuclear morphology. Blasts with cup-like nuclear appearance have been rarely reported. However, their response to treatment as well as prognosis is similar to the case of AML with conventional round nuclear appearance. Patient responded satisfactorily to cytarabine, daunorubicin and hydroxyurea therapy.
Author Declaration:
Financial or Other Competing Interests: No
Was informed consent obtained from the subjects involved in the study? Yes
For any images presented appropriate consent has been obtained from the subjects. Yes
PLAGIARISM CHECKING METHODS: [Jain H et al.]
Plagiarism X-checker: May 01, 2019
Manual Googling: May 22, 2019
iThenticate Software: Sep 20, 2019 (9%)
[1]. Jost E, Herwartz R, Ho-BM Vankann L, Fuchs R, Cup-like blasts in acute myeloid leukemiaAmer J Hematol 2015 90(9):847-48.10.1002/ajh.2395425615838 [Google Scholar] [CrossRef] [PubMed]
[2]. Kamoda Y, Shimomura D, Tsuda K, Hayashida M, Fukutsuka K, Izumi K, Acute myeloid leukemia with “cup-like” nuclear morphology, highlighting the electron microscopic featuresTenri Medical Bulletin 2017 20(1):26-37.10.12936/tenrikiyo.20-004 [Google Scholar] [CrossRef]
[3]. Chen W, Konoplev S, Medeiros LJ, Koeppen H, Leventaki V, Vadhan-Raj S, Cuplike nuclei (prominent nuclear invaginations) in acue myeloid leukemia are highly associated with FLT-ITD and NPM1 mutationsCancer 2009 115(23):5481-89.10.1002/cncr.2461019672946 [Google Scholar] [CrossRef] [PubMed]
[4]. Kim MJ, Ahn S, Jeong SH, Jang JH, Han JH, Choi JR, Minor BCR-ABL1-positive acute myeloid leukemia associated with the NPM1 mutation and FLT3 internal Tandem DuplicationAnn Lab Med 2016 36:263-65.10.3343/alm.2016.36.3.26326915617 [Google Scholar] [CrossRef] [PubMed]
[5]. Mehtap O, Atesoglu EB, Gonullu E, Keski H, Hachanefioglu A, Are cup-like blasts specific to AML patients with FLT3 ITD and a normal karyotype? An ALL case report and review of literatureTurk J Hematol 2011 28:142-45.10.5152/tjh.2011.3227264130 [Google Scholar] [CrossRef] [PubMed]
[6]. Sasine JP, Lee TD, Xian RR, Schiller GJ, Acute myeloid leukemia with cup-like nuclear inclusions, and FLT3-ITD and NPM1 mutationsJournal of Leukemia 2017 5:110.4172/2329-6917.1000i101 [Google Scholar] [CrossRef]
[7]. Jain P, Vega-Vazquez F, Faderl S, “Cup-like” blasts and NPM1 and FLT3 (ITD) mutations in acute myeloid leukemia (AML)Int J Hematol 2013 98:310.1007/s12185-013-1371-323690292 [Google Scholar] [CrossRef] [PubMed]
[8]. Rolling C, Ehninger G, How I treat hyperleukocytosis in acute myeloid leukemiaBlood 2015 125(21):3246-52.10.1182/blood-2014-10-55150725778528 [Google Scholar] [CrossRef] [PubMed]