Tooth alignment plays a major role in facial esthetics; therefore, the demand for orthodontic treatment is increasing worldwide. By the increased demand of adult patients for orthodontic treatment, bonding of orthodontic brackets to dental restoration surfaces is now a common necessity [1] because most adult patients have composite resin, amalgam, gold, acrylic resin or ceramic restorations [2]. In the recent years, application of ceramic materials for dental restorations, such as fixed crowns, has greatly increased [1,3]. Ceramics used in dentistry include different types of glass ceramics, spinel, alumina, and zirconia ceramics [2]. Ceramic crowns for fixed partial dentures are made of full ceramic, feldspathic porcelain, porcelain fused to metal and recently zirconia [3]. Zirconia crowns are among the best types of dental crowns, which have high fracture resistance, excellent fit, optimal biocompatibility and a natural appearance. In contrast to porcelain fused to metal restorations, zirconia crowns do not cause marginal discoloration of the gingiva. Moreover, they can be used in patients who are allergic to metals [2]. Zirconia crowns are commonly used in areas where esthetics is as important as strength [4]. Orthodontic brackets can be bonded to materials other than the enamel using adhesives. However, bonding of orthodontic brackets to restorative materials such as ceramic crowns is still a challenge for orthodontists [5]. Considering the increasing demand for orthodontic treatment and presence of teeth with different types of restorations or crowns in the oral cavity, novel adhesives and techniques are required to enable a stronger bond between metal brackets and different crown types. Since zirconia is commonly used for the fabrication of dental crowns, it is important to assess the efficacy of different methods for zirconia surface treatment [6]. An optimal bonding agent should support and protect the brackets against masticatory and orthodontic forces and prevent their debonding during the course of treatment. It should also enable safe debonding and protect the tooth structure or the surface of restorative materials during debonding [7]. Surface treatment is among the most important factors in bracket bonding to surface of restorative materials. For ceramic surface treatment, mechanical methods such as increasing the surface roughness by a diamond bur and air abrasion with aluminium oxide or silica particles or chemical methods such as acid etching with/without the application of silane can be used [2].
Zirconia is increasingly used due to its optimally high strength and favourable biocompatibility. However, it lacks the glass phase and therefore, etching with hydrofluoric acid is not an option [8]. Bonding of zirconia to dental structure or synthetic materials is hardly possible due to its chemical non-reactivity and resistance to chemical agents such as acids, alkaline materials and organic and inorganic solvents [8]. Therefore, novel methods are required to enable more efficient bonding of zirconia to different substrates. A new type of adhesive known as universal adhesive or multipurpose adhesive is now available in the market that can be used in two-step etch and rinse or one-step self-etch mode. This property allows the clinicians to choose one of the two strategies according to their priorities and/or clinical status of patients. Universal adhesives can bond to materials such as resin, stainless steel, ceramic and zirconia [9]. According to the manufacturers’ claims and the findings of previous studies, universal adhesives have unique chemical composition and bonding mechanism, which enable efficient bonding to different substrates such as zirconia crowns [10]. Studies on surface treatment of zirconia crowns prior to bonding to orthodontic brackets are limited [11-13]. Thus, this in-vitro study aimed to assess the SBS of metal brackets bonded to zirconia surfaces with a universal adhesive following different surface treatments-sandblasting with 50 μm aluminium oxide particles and acid etching with 37% orthophosphoric acid.
Materials and Methods
This in-vitro experimental study was conducted in School of Dentistry, Kermanshah University of Medical Sciences from November 2017 to March 2018 and was approved by the Ethics Committee of Kermanshah University of Medical Sciences, Kermanshah, Iran. In this study, sample size was calculated considering alpha=99, power of 95%, standard deviation of SBS in the control and hydrofluoric acid groups to be 0.01 and 0.03 and the mean SBS of 0.012 and 0.099, respectively. Minimum sample size was calculated to be 6 in each group using PASS version 11. To increase the accuracy, 10 samples were included in each group (a total of 30 samples) [9]. Thirty non-glazed zirconia porcelain blocks measuring 1×1×1 cm were prepared by computer-aided design/computer-aided manufacturing technique. For this purpose, zirconia blanks (Zircon Z; Amann Girrbach) were used. The shrinkage of these blanks is separately calculated for each blank by the manufacturer, which is referred to as the f-factor or shrink factor. The computer calculated the dimensions and transferred them to the milling machine. The blanks were milled at a spindle speed of around 20,000 rpm. Next, they were removed from the machine, cleaned, dyed and dried under 70°C in infrared light. They were then transferred to zircon sintering slab [14]. The sintering process was started at 200°C. The temperature increased at a rate of 480°C/hour, held at 900°C for 30 minutes and then held at 1450°C for 2 hours followed by gradual cooling at a rate of -300°C/hour [14]. The samples were then randomly divided into three groups of 10 for different surface treatments as follows:
Control group: No zirconia surface treatment
Sandblasting group: Sandblasting of the zirconia surface with 50 μm aluminium oxide particles at 45° angle, 90 psi pressure and 10 mm distance from the surface for 2 seconds [Table/Fig-1a].
(a) Sandblasting of zirconia surface; (b) Acid etching of zirconia surface; (c) Curing of composite used to bond bracket to zirconia surface (control group); (d) A block in universal testing machine.
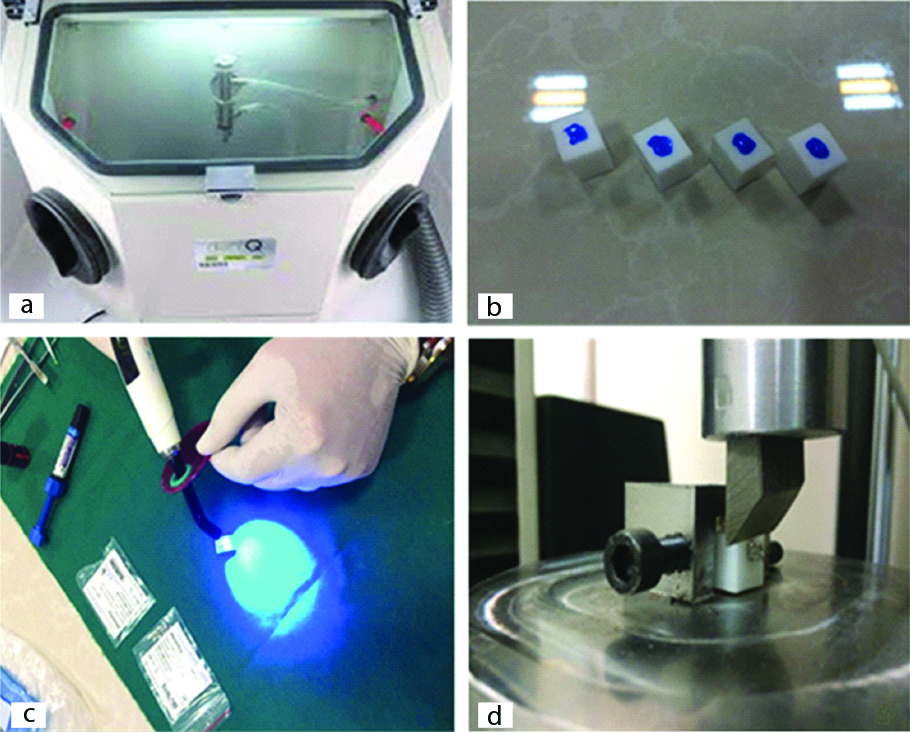
Acid etching group: Acid etching of the zirconia surface with 37% orthophosphoric acid (Ultra-etch) for one minute [Table/Fig-1b].
In all groups, Scotchbond Universal adhesive (3M, USA) was applied on the surface of blocks according to the manufacturer’s instructions and light-cured for 40 seconds using a LED light curing unit (LED curing light DB 686 Mocha) with a light intensity of 1200 mW/cm2. Next, metal orthodontic brackets (maxillary central incisor brackets) were bonded to zirconia blocks using Transbond XT orthodontic composite (3M Unitek). For this purpose, the entire bracket base was coated with composite, placed on the zirconia block with finger pressure, excess composite was removed by a dental explorer and the composite was then light-cured for a total of 40 seconds [Table/Fig-1c]. The samples were then subjected to shear load in a universal testing machine (SM5; Santam, Tehran, Iran) at a crosshead speed of 2 mm/minute [Table/Fig-1d]. The amount of load at the time of bracket debonding from the zirconia block was measured in Newton (N). The SBS was calculated in megapascals (MPa) by dividing the load in Newton by the bracket base surface area (5×2.5 mm2).
All samples were inspected under a stereomicroscope (SZX16, Olympus) at x20 magnification, and the mode of failure (bracket-zirconia) was determined. The mode of failure was determined according to the Adhesive Remnant Index (ARI) score by Artun and Bergland [15], which indicates the amount of composite remaining on the zirconia surface.
Score 1: All composite remaining on the zirconia surface
Score 2: Over 90% of composite remaining on the zirconia surface
Score 3: 10% to 90% of composite remaining on the zirconia surface
Score 4: Less than 10% of composite remaining on the zirconia surface
Score 5: No composite remaining on the zirconia surface
Two observers, a restorative dentist and an orthodontist, independently inspected the samples to determine the mode of failure. All samples were evaluated again after 2 weeks by the same observers. The inter-observer agreement was calculated using kappa statistics, and the kappa coefficient was found to be 0.92. According to the Cicchetti’s classification [16], this value indicated excellent agreement between the observers.
Statistical Analysis
Data were analysed using SPSS version 16 (SPSS Inc., IL, USA). According to the Shapiro-Wilk test, the SBS data were normally distributed. Thus, SBS data were analysed using ANOVA. However, the mode of failure, data were not normally distributed; thus, they were analysed using the Kruskal-Wallis test. Level of significance was set at 0.05.
Results
[Table/Fig-2] shows the mode of failure under a microscope and remaining composite on the surface of the bracket.
Mode of failure of bracket under a microscope. (a) No composite remaining on the bracket surface; (b) Less than 10% of composite remaining on the bracket surface; (c) 10% to 90% of composite remaining on the bracket surface; (d) Over 90% of composite remaining on the bracket surface; (e) All composite remaining on the bracket surface.

According to the Shapiro-Wilk test, the SBS values had normal distribution in the three groups. Thus, parametric ANOVA was used to compare the SBS among the three groups.
[Table/Fig-3] shows the mean, standard deviation, minimum and maximum SBS values in the three groups. The highest SBS was noted in sandblasting group (19.25±9.07 MPa). The SBS was 12.85±7.16 MPa in the control group, and the acid etching group showed the lowest SBS (9.89±9.28 MPa). ANOVA showed no significant difference in SBS among the three groups (F=3.131, df=2, p=0.06).
Comparison of SBS among the groups (n=10).
| Study group | Shear bond strength (MPa) | p-value |
|---|
| Minimum | Maximum | Mean±standard deviation | |
|---|
| Control | 4.51 | 28.45 | 12.85±7.16 | 0.06 |
| Sandblasting | 5.60 | 35.20 | 19.25±9.07 |
| Acid etching | 9.80 | 23.60 | 9.89±9.28 |
[Table/Fig-4] shows the frequency of modes of failure according to the ARI scores. Since the ARI score data were not normally distributed, the Kruskal-Wallis test was applied which showed that the three groups were not significantly different in terms of the mode of failure (chi-square=3.323, df=2, p=0.19). The mode of failure was mixed in all groups [Table/Fig-2].
Frequency of modes of failure in the three groups according to the ARI scores (n=10).
| Group | Score 1 | Score 2 | Score 3 | Score 4 | Score 5 | p-value |
|---|
| Control | 0 | 3 (30%) | 3 (30%) | 0 | 4 (40%) | 0.19 |
| Sandblasting | 0 | 3 (30%) | 1 (10%) | 0 | 6 (60%) |
| Acid etching | 1 (10%) | 6 (60%) | 0 | 0 | 3 (30%) |
Discussion
This study assessed the SBS of metal brackets bonded to zirconia with universal adhesive following different surface treatments. The results showed that the SBS values were 12.85±7.16, 19.25±9.07 and 9.89±9.28 MPa in the control, sandblasting and acid etching groups, respectively. No significant difference was noted among the three groups in SBS (p=0.06) or the ARI scores (p=0.19).
Several methods are used for treatment of zirconia surfaces such as mechanical surface roughening by diamond bur, abrasive discs or aluminium oxide particles and chemical preparation, which includes the use of orthophosphoric acid, hydrofluoric acid or silane. In the current study, zirconia surfaces were roughened by sandblasting with 50 μm aluminium oxide particles in one group, and 37% orthophosphoric acid was used for cleaning of the zirconia surface from debris and organic materials in another group. Kulunk S et al., evaluated the efficacy of five methods of air abrasion with 1-3 μm artificial diamond particles, 110 μm aluminium oxide particles, 60-80 μm cubic boron nitride particles, 30 μm silica-coated aluminium oxide particles and 30-50 μm artificial diamond particles [17]. They also had a control group (no air abrasion). They found that the size and type of particles used for air abrasion affected the bond strength of zirconia such that the highest and the lowest bond strength values were recorded in air abrasion with 30-50 μm artificial diamond particles and the control group, respectively. Also, higher bond strength values were noted in groups of 30-50 μm artificial diamond particles, 110 μm aluminium oxide particles and 60-80 μm cubic boron nitride particles. Previous studies used 125 μm [18], 110 μm [14,19], 50 μm [20] and 30 μm [21] aluminium oxide particles.
Universal Bonding (3M) was used in all samples in our study and the results showed that the SBS of all samples in all groups was acceptable irrespective of the type of surface treatment. According to the current findings, the highest SBS was noted in the sandblasted group (19.25±9.07 MPa). The SBS was 12.85±7.16 MPa in the control group, and the orthophosphoric acid etching group showed the lowest SBS value (9.89±9.28 MPa) and no significant difference in SBS among the three groups was observed. Similarly, Yassaei S et al., indicated that surface treatment with sandblasting yielded the highest SBS (7.81±1.02 MPa) of zirconia to orthodontic brackets while the lowest bond strength was noted in samples etched with hydrofluoric acid (5.84±0.78 MPa) [11]. However, it should be noted that Yassaei S et al., performed thermocycling, while thermocycling was not performed in our study [11]. Kim J et al., evaluated the effect of different surface treatments of zirconia for bracket bonding and reported that sandblasting yielded the highest bond strength of bracket to zirconia [22]. Their findings were in agreement with our results. Bavbek NC et al., demonstrated that air abrasion by CoJet followed by the application of universal primer improved the microshear bond strength of orthodontic resin to glazed or polished zirconia compared to the control (no air abrasion) samples [23]. Hosseini MH et al., evaluated the SBS of bracket to zirconia after surface treatment with Er:YAG laser, sandblasting and silane application [24]. They showed that sandblasted samples yielded the highest SBS, which was in line with the present study results. Another study also confirmed the optimal efficacy of mechanical surface treatments of zirconia in increasing the bond strength of resin cement to zirconia surfaces [25]. Kim JH et al., indicated the superior efficacy of primer application compared to air abrasion [26]. The current results and those of Yassaei S et al., showed the lowest bond strength in acid etched group [11]. The low bond strength of acid etched group may be due to the fact that zirconia cannot be etched due to its highly crystalline structure [21]. Lower bond strength in the etching group can be due to the effect of etchant on the zirconia surface, which may enhance chemical bonding to universal adhesives. However, since etching with phosphoric acid does not significantly damage the zirconia surface molecules; this reduction in bond strength was not considerable and did not reach statistical significance in our study. But, surface treatment with orthophosphoric acid yielded the lowest bond strength. However, it was clinically acceptable because bond strength in the range of 6-8 MPa is sufficiently high for orthodontic bracket bonding. Evidence shows that 37% phosphoric acid can be used for ceramic surface treatment as an alternative to hydrofluoric acid [27]. The current study showed no significant difference in ARI scores among the three groups. However, the area of the zirconia surface without composite coating in sandblasting group (ARI score 5 in 60% of the samples) was greater than that in the control (40%) and acid-etching (30%) groups. In bracket debonding, bond failure at the adhesive-tooth interface with no residue remaining on the tooth or crown surface is clinically favourable. Debonding at the bracket-adhesive interface or within the adhesive material, which results in residual adhesive remaining on the tooth surface, is important for prevention of enamel fracture [28]. Lee YJ et al., measured the bond strength of ceramic brackets to zirconia using different zirconia and universal adhesive primers [29]. The ARI score zero (no composite remaining on the zirconia surface) had the highest frequency, which was in agreement with our findings (since ARI score 5 is indicative of no composite remaining on the zirconia surface which had the highest frequency in our study).
Limitation
Our study had some limitations. In-vitro design was the limitation of this study. Thus, generalisation of results to the clinical setting should be done with caution. Also, thermocycling can help in better simulation of clinical settings in in-vitro studies, which was not performed in this study. Future studies are required on the efficacy of different types of adhesives and sandblasting with different sizes of aluminium oxide particles.
Conclusion
Surface treatment with sandblasting and etching with orthophosphoric acid had no significant effect on SBS of zirconia to metal brackets. Application of Scotchbond Universal adhesive in this study provided acceptably high SBS in all groups, irrespective of the type of surface treatment.
Fundings
This study was funded by the vice-chancellor of Research and Technology of Kermanshah University of Medical Sciences.