Introduction
Prostate Cancer (PCA) is the second most common cancer diagnosed in men worldwide. Prostate Specific Antigen (PSA) is the readily used biomarker for PCA screening, but lack of specificity limits its usefulness. Hence, role of Androgen receptor (AR) CAG repeat polymorphism and Cytochrome P450 3A5*3 gene polymorphism involved in the metabolism of testosterone, growth and differentiation of prostate gland were evaluated to assess their association with pathogenesis of prostate lesions.
Aim
To determine CAG repeats in AR gene and CYP3A5*3 gene polymorphism in South Indian men with PCA and Benign Prostate Hyperplasia (BPH).
Materials and Methods
Genomic DNA was isolated from 312 (100 men with PCA, 109 BPH patients and 103 controls). Formalin fixed paraffin embedded tissue/peripheral blood samples using salting out method were used. Polymerase Chain Reaction (PCR) procedure was carried out using specific primers for AR and CYP3A5*3 genes. Bands were visualised by electrophoresis on 2% ethidium bromide (EtBr) stained agarose gel. Odds ratios were calculated using MedCalcR version 18.2.1 and Chi-square analysis was carried out to determine the association among groups.
Results
AR gene with <22 CAG repeats and ‘GG’ genotype of CYP3A5*3 gene polymorphism were identified in 61 (61%), PCA patients, 11 (10%) BPH and 13 (13%) control individuals. A statistically significant association was observed between AR short repeats and CYP3A5*3 ‘GG’ genotype with PCA (p-value<0.05).
Conclusion
The results suggest that the best age for PCA screening is 48 years or above for early detection and prevention of this malignancy. A <22 CAG repeats in AR gene and ‘GG’ genotype of CYP3A5*3 (rs#776746) gene can be together considered as specific molecular marker for identifying men at a risk of developing PCA.
Introduction
PCA is the second most frequently diagnosed cancer among men worldwide and its incidence is higher in Western population compared to Asian population [1]. According to GLOBOCAN 2018, it is estimated that the incidence of PCA cases is almost 1.3 million with a mortality rate of 3,59,000 per 1,00,000 men in 2018 and it is the fifth leading cause of cancer related death in men [2]. Age and ethnicity play a key role in the incidence and mortality of PCA [3,4]. Over the past decade, there has been a significant decrease in the incidence of PCA in the developed countries due to enhanced awareness, broad implementation of prostate screening and the development of comprehensive cancer screening strategies [3]. However, in developing countries like India, due to lack of such screening strategies, the actual prevalence has yet been established.
Enlargement of the prostate gland also known as BPH is a common pathology affecting elderly men above 50 years of age that often progress to a characteristic pattern of growth to develop into PCA [5,6]. Apart from age, the major risk factors for prostate pathologies are ethnicity, family history and genetics [6]. It was known that inherited factors contribute about 5-9% to the aetiology of PCA [7]. Alterations in DNA sequence underline the development of PCA. Ample studies suggests that CAG repeat polymorphisms in AR gene and cytochrome P4503A5*3 (CYP3A5*3) gene polymorphism are involved in the metabolism of androgens such as testosterone, and influence the risk of PCA thus making them novel candidate genes for PCA risk assessment [4,8,9].
AR gene located on chromosome Xq11-12 encodes a four domains protein known to be involved in androgen signaling that regulates male secondary sexual characteristics [10]. AR binds to androgens, mainly testosterone and 5-α-dihydrotestosterone and plays a fundamental role in the growth, differentiation and maintenance of normal and malignant growth of the prostate [11,12]. AR gene has a triplet CAG repeat in its exon 1, which regulates the expression of AR. AR gene with shorter CAG repeats have higher transcription activity, and while high numbers of CAG repeats leads to decreased testosterone activity [10].
CYP3A5*3 gene located at 7q21-22 belongs to a multi-gene family of cytochrome P450. It is most abundantly expressed in liver, small intestine and to a lesser extent in normal prostate cells implying a more intense and localised influence on the prostate gland [13]. CYP3A5*3 is involved in drug metabolism, and oxidation and inactivation of testosterone to a less biologically active form like 2β, 6β, or 15β -hydroxytestosterone, which are readily eliminated from the body. CYP3A5*3 sequence variant 6986A>G affects the enzyme activity and increases the bioavailability of testosterone hence it serves as an important aetiological factor in development of PCA [13-15].
Based on these findings, the present study was conducted to evaluate CAG repeats in AR gene and CYP3A5 gene 6986A>G variation in men with PCA and BPH to determine their association with cancer and benign prostate pathologies as compared to controls.
Materials and Methods
This case-control study was conducted at the Department of Genetics and Molecular Medicine, Kamineni Hospitals, Hyderabad, Telangana, India, from January 2014 to December 2016. Ethical approval was obtained from the Institutional Ethics Committee of Kamineni Hospitals (Registration Number: ECR/58/Inst/AP/2013) Hyderabad, India.
The sample size was calculated based on the formula n=z*sigma/5 by taking seven studies into consideration. The estimated sample size was 87 hence the study was carried out on 100 PCA patients and 109 BPH cases with 95% CI. Apparently healthy 103 men without any prostate problems visiting the Department for the Master Health Checkup (MHC) were recruited as controls. An informed verbal consent was obtained from the participants of the study.
PCA and BPH samples were collected from clinically identified men, who were radiologically diagnosed by Trans Rectal Ultrasonography (TRUS) or Trans Urethral Resection of Prostate (TURP) and confirmed histopathologically by salting out method collected from men above 40 years of age with a normal ultrasound of abdomen, PSA levels <4ng/mL and without any prostate related problems.
Isolation of Genomic DNA
Genomic DNA was extracted from 312 formalin fixed paraffin embedded tissue/peripheral blood samples by method, previously standardised by our group. The amount of DNA was quantified using NanoDropTM 2000/2000cc, (Thermo ScientificTM, model number: ND-2000) and stored at -20°C deep freezer until further analysis [16,17].
AR Gene CAG Repeats and CYP3A5*3 Gene 6986A>G Polymorphism Genotyping
A three-step PCR procedure was carried out in a gradient PCR thermal cycler, (Applied Biosystems 9902 Veriti) using AR and CYP3A5*3 specific-gene primers purchased from BioArtis Life Sciences Private Limited, Hyderabad, India [Table/Fig-1] [18,19].
Forward and reverse primers for AR and CYP3A5*3 gene [18,19].
| Gene | Orientation | Primers (5’-3’) | Reference |
|---|
| AR gene | Forward | TGCGCGAAGTGATCCAGAAC | [18] |
| Reverse | CTTGGGGAGAACCATCCTCA |
| CYP3A5*3 | Forward | CATCAGTTAGTAGACAGATGA | [19] |
| Reverse | GGTCCAAACAGGGAAGAAATA |
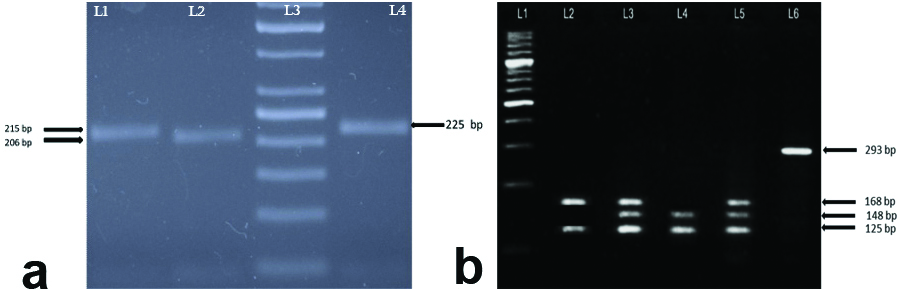
Restriction Fragment Length Polymorphism (RFLP), of CYP3A5*3 gene polymorphism was carried out using SsPI restriction enzyme (Thermo ScientificTM catalog number ER0771) [19]. Amplified PCR and restriction digestion products were separated on 2% agarose gel by electrophoresis [17]. Gel was photographed using UVITEC gel documentation system [Table/Fig-2].
Amplification profile of PCR and restriction digested products of AR and CYP3A5*3 gene polymorphisms. a) AR gene CAG repeats. Lane 1: Band size 215 bp Allele 23 (A23) has 23 CAG repeats, Lane 2: 206 bp Allele 22 (A22) has 22 CAG repeats, Lane 3: 50bp DNA ladder, and Lane 4: 225 bp Allele 24 (A24) has 24 CAG repeats. b) CYP3A5*3 (rs#776746) gene polymorphism. Lane 1: 100bp DNA ladder, Lane 2: Homozygous GG genotype (168/125 bp), Lane 3 & Lane 5: Heterozygous AG genotype (168/148/125) bp), Lane 4: Homozygous AA genotype (148/125 bp) and Lane 6: Undigested PCR product size 293 bp.
Statistical Analysis
Odds Ratios (OR) and Chi-square analysis tests were used to determine association between groups with 95% Confidence Intervals (CIs) using MedCalcR statistical software version 18.2.1 [17].
Results
Baseline Characteristics of the Participants
The risk factor for onset of prostate pathologies and cancer increases with age. In the present study age range of participants was 40 to 87 years. The mean age of PCA patients was 69.46±9.25 years, while that of individuals with BPH was of 66.99±8.30 years Right, 72 (72%) PCA and 71 (65%) BPH cases were aged ≥65 years [Table/Fig-3].
Categorization of PCA, BPH and controls individuals on the basis of clinical parameters.
| Clinical parameters | Groups |
|---|
| PCA (n=100) | BPH (n=109) | Controls (n=103) |
|---|
| Age |
| <65 years | 28 (28%) | 38 (35%) | 87 (84%) |
| ≥65 years | 72 (72%) | 71 (65%) | 16 (16%) |
| PSA |
| ≤4 ng/mL | 6 (6%) | 21 (19%) | 91 (88%) |
| >4 ng/mL | 94 (94%) | 88 (81%) | 12 (12%) |
| PV |
| ≤25cc | 18 (18%) | 14 (13%) | 100 (97%) |
| >25cc | 82 (82%) | 95 (87%) | 3 (3%) |
| GS | (n=81) | NA | NA |
| <7 | 13 (16%) | | |
| ≥7 | 68 (84%) | | |
PCA: Prostate cancer; BPH: Benign prostate hyperplasia; GS: Gleason score; PCA: Prostate cancer; PSA: Prostate specific antigen; PV: Prostate volume; NA: Not applicable
Clinically, PSA levels ≤4 ng/mL is considered as normal by the National Institutes of Health, USA. In the present study, PSA levels ranged from 0.09 ng/mL to one of the cases showed 1230 ng/mL in cases and controls, with mean PSA 50.28±51.51 ng/mL, 20.48±19.06 ng/mL and 3.86±13.36 ng/mL in PCA, BPH and control groups, respectively. Majority of the PCA 94 (94%) and BPH 88 (81%) cases had PSA levels >4 ng/mL as opposed to 12 (12%) control individuals [Table/Fig-3].
Enlargement of prostate gland diagnosed by ultrasound is another marker for Prostate Cancer. In adult men, Prostate Volume (PV) depicts prostate size and PV ≤25cc is considered normal. In the present study, PV levels ranged from 10cc to 210cc, with mean PV of 45.33±27.50cc, 50.81±25.76cc and 18.02±3.51cc in PCA, BPH and control groups, respectively. Prostate enlargement was observed in 82 (82%) PCA and 95 (87%) BPH cases, while 100 (97%) control individuals had normal prostate size [Table/Fig-3].
Histopathological grading of the PCA was based on the Gleason Score (GS) that ranges from 5 to 10 and helps in assessing aggressiveness of the cancer [20]. Based on the International society of urological pathology (ISUP), GS is categorised as: (i) <7; and (ii) ≥7 and in the present study, 68 (84%) of the PCA cases had a GS of ≥7 which indicates aggressive state of disease [Table/Fig-3].
AR gene CAG Repeats
AR gene amplified PCR products had a band size ranging in length from 85bp (9 CAG repeats) to 300bp (32 CAG repeats) in cases and controls. A <22 CAG repeats corresponding to increased transcriptional activity was observed in 94 (94%) PCA cases, in contrast to only 20 (18%) BPH cases and 18 (17%) control individuals. The <22 CAG repeats of AR gene were found to be significantly associated with PCA (OR-73.9815, 95%CI- 28.0617 to 195.0436) (p-value <0.0001) [Table/Fig-4].
AR gene CAG repeats analysis in PCA, BPH and control groups.
| Groups | <22 CAG repeats | ≥22 CAG repeats | Categories, Odds ratio (OR), 95%CI | p-value |
|---|
| PCA (n=100) | 94 (94%) | 6 (6%) | PCA vs. controls OR- 73.9815, (95% CI- 28.0617 to 195.0436) | <0.0001* |
| BPH (n=109) | 20 (18%) | 89 (82%) | BPH vs. controls OR- 1.0612, (95%CI- 0.5255 to 2.1428) | 0.8524 |
| Controls (n=103) | 18 (17%) | 85 (83%) | PCA vs. BPH OR- 69.7167, (95%CI- 26.7668 to 181.5836) | <0.0001* |
OR (Odds ratios) *p-value <0.05 statistically significant. PCA: Prostate cancer; BPH: Benign prostate hyperplasia
CYP3A5*3 Gene 6986A>G Polymorphism (rs#776746)
The frequency of ‘G’ allele from control population was established as 0.54. In PCA cases, the frequency of ‘G’ allele was observed to be 0.77 and 0.66 in BPH cases [Table/Fig-5]. The risk ratio for the individuals with GG genotype was found to be seven fold higher when compared to AG and AA genotype in our study population. Dominant model of inheritance explains the ‘G’ allele association with PCA.
Genotype frequency of CYP3A5 gene in PCA, BPH and control groups.
| Groups | Allelic frequency | Allelic frequency ‘G’ vs. ‘A’ allele | p-value |
|---|
| G | A | Categories | Odds ratio, 95%CI |
|---|
| PCA (n=100) | 155 (0.77) | 45 (0.23) | i) PCA vs. controls | OR- 2.9479, 95%CI- 1.9170 to 4.5333 | <0.0001* |
| BPH (n=109) | 145 (0.66) | 73 (0.34) | ii) BPH vs. controls | OR- 1.7000, 95%CI- 1.1480 to 2.5175 | 0.0081 |
| Control (n=103) | 111 (0.54) | 95 (0.46) | iii) PCA vs. BPH | OR- 1.7341, 95% CI- 1.1224 to 2.6793 | 0.0131 |
| Groups | Genotype frequency | Dominant model | Odds ratio, 95% CI | p-value |
| AA | AG | GG | GG+AG vs. AA |
| PCA (n=100) | 8 (8%) | 29 (29%) | 63 (63%) | i) PCA vs. controls | OR- 7.9180, 95%CI- 3.4789 to 18.0217 | <0.0001* |
| BPH (n=109) | 27 (24.77%) | 19 (17.43%) | 63 (57.8%) | ii) BPH vs. controls | OR- 2.0911, 95%CI- 1.1636 to 3.7578 | 0.0136 |
| Control (n=103) | 42 (40.77%) | 11 (10.68%) | 50 (48.55%) | iii) PCA vs. BPH | OR- 3.7866, 95%CI- 1.6294 to 8.7998 | 0.0020 |
OR (Odds ratios) *p-value<0.05 statistically significant. PCA: Prostate cancer; BPH: Benign prostate hyperplasia
Assessment of Short CAG Repeats (<22) in AR Gene with ‘GG’ Genotype in CYP3A5 Gene Polymorphisms in Cases and Controls
In the present study, 61 (61%) PCA cases, 11 (10%) BPH cases and 13 (13%) control individuals had <22 CAG repeats and ‘GG’ genotype. Chi-square analysis showed that AR gene with <22 CAG repeat polymorphism and the GG genotype of CYP3A5*3 was found to be significantly associated with PCA (X2=50.105, 95%CI-35.4494 to 58.3715) (p-value<0.0001) [Table/Fig-6].
Association of <22 CAG repeats in AR gene and ‘GG’ genotype of CYP3A5 gene in PCA cases.
| AR, CYP3A5*3 | <22, GG | <22, AG | <22, AA | >22, GG | >22, AG | >22, AA |
|---|
| PCA (n=100) | 61 (61%) | 27 (27%) | 6 (6%) | 4 (4%) | 1 (1%) | 1 (1%) |
| BPH (n=109) | 11 (10%) | 5 (4%) | 4 (4%) | 52 (48%) | 14 (13%) | 23 (21%) |
| Control (n=103) | 13 (13%) | 0 (0%) | 5 (5%) | 37 (36%) | 11 (10%) | 37 (36%) |
| Short CAG repeats (<22) in AR gene and with ‘GG’ genotype in CYP3A5 gene in groups | Comparison of proportions in categories- 95%CI | p-value |
| PCA (n=100) <22, GG (n= 61) (61%) | i) PCA vs. controls- 50.105, 95%CI- 35.4494 to 58.3715 | <0.0001* |
| BPH (n=109) <22, GG (n=11) (11%) | ii) BPH vs. controls- 0.200, 95% CI- N6.9138 to 11.0918 | 0.4941 |
| Control (n=103) <22, GG (n=13) (13%) | iii) PCA vs. BPH- 57.095, 95%CI- 37.8113 to 60.0906 | <0.0001* |
Chi-square analysis *p-value<0.05 statistically significant. PCA: Prostate Cancer; BPH: Benign Prostate Hyperplasia
Discussion
PCA is a heterogeneous cancer in men that progresses gradually with age. Several candidate genes are associated with PCA risk, and in the present study genes linked with androgen metabolism leading to PCA cell growth and progression were evaluated. The mean age of men at the time of diagnosis was 69.46±9.25 years, which was similar to Finland, Philadelphia and Australian population where the mean age was 68.8 years, 62.9±8 years and 63 years, respectively [2,21,22]. Based on these values, age at diagnosis was considered as >65 years. Since the estimated age of screening is calculated as mean age ±2xSD, the men aged 48 years or above are recommended to screen for PCA. National cancer institute (NIH) recommends PCA screening in men >50 years of age and with elevated PSA levels [23,24].
In our study, both PCA and BPH patients had PSA levels >4 ng/mL indicating that PSA is not a specific marker for prostate malignancy. According to NIH, there is no specific normal or abnormal level of PSA, but a clinical value of ≤4 ng/mL is taken as normal and that of >4 ng/mL requires a follow-up biopsy [25]. PSA is the only marker currently available and routinely used in screening and diagnosis of prostate Pathologies, however, it lacks specificity, and cannot differentiate between prostatitis, benign and malignant tumours which leads to over diagnosis and overtreatment [26,27]. Therefore, a better and more specific molecular genetic marker for the assessment of PCA risk in men is the need of the hour.
CAG repeats in AR and CYP3A5*3 gene polymorphisms involved in testosterone metabolism were evaluated for PCA risk assessment. A meta-analysis study in Asian population, reported that short number of (<22) CAG repeats in AR gene and CYP3A5 6986A>G variation along with conventional risk factors are associated with increased risk of PCA [28]. Numerous studies report similar findings in Macedonian, Mexican, Indian and Taiwanese men [29-32].
A meta-analysis of worldwide population based sequencing on CYP3A5*3 revealed a Minor Allele Frequency (MAF) of 100% in South Asians, 98.6% in East Asians, 99.5% in Europeans, 41.2% in Africans and 96.7% in admixed Americans [33]. According to 1000G and GnoMAD database MAF of CYP3A5*3 the frequency of allele ‘G’ is 62.1% and 73.47% respectively. The ‘GG’ genotype of CYP3A5*3 gene was documented to play a central role in increasing the bioavailability of testosterone; thereby increasing the PCA cell growth and progression [15,34].
Previous literature reports a significant association of ‘GG’ genotype of CYP3A5*3 polymorphism with PCA and BPH in dominant model indicating a role of testosterone metabolism in prostate pathologies. In the present study, CYP3A5 gene polymorphism was found to be associated with increased risk of developing PCA and these findings were in agreement with various other studies [13,15,34-37].
Primarily used Androgen Deprivation Therapy (ADT) for treatment modality for PCA show a positive initial response but inevitably relapse to an androgen-independent phenotype, and ultimately fails to suppress the presence of androgens at the tissue level. Therefore, genotypic analysis of CAG repeats of AR and GG genotype of CYP3A5 gene polymorphisms can also help in development of personalised therapy in treatment and management of the disease [14,38,39].
Limitation
Sample size was relatively small and study population was restricted to patients visiting the hospital. Further cohort studies on different ethnic groups and larger sample size are required for better understanding of the disease.
Conclusion
AR gene with <22 CAG repeats and GG genotype of CYP3A5*3 gene polymorphisms together can be used as a specific molecular marker for screening, diagnosis and management of PCA. Men over the age of 48 years, with short (<22) CAG repeats in AR gene and ‘GG’ genotype in CYP3A5*3 gene polymorphism are at an increased risk of developing PCA and require regular surveillance for risk assessment.
[1]. Mathew A, George PS, KM JK, Vasudevan D, James FV, Transition of cancer in populations in IndiaCancer Epidemiology 2019 58(1):111-20.10.1016/j.canep.2018.12.00330537646 [Google Scholar] [CrossRef] [PubMed]
[2]. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countriesCA: A Cancer Journal for Clinicians 2018 68(6):394-424.10.3322/caac.2149230207593 [Google Scholar] [CrossRef] [PubMed]
[3]. Kimura T, Egawa S, Epidemiology of prostate cancer in Asian countriesInternational Journal of Urology 2018 25(6):524-31.10.1111/iju.1359329740894 [Google Scholar] [CrossRef] [PubMed]
[4]. Merriel SW, Funston G, Hamilton W, Prostate cancer in primary careAdvances in Therapy 2018 35(9):1285-94.10.1007/s12325-018-0766-130097885 [Google Scholar] [CrossRef] [PubMed]
[5]. Udensi UK, Tchounwou PB, Oxidative stress in prostate hyperplasia and carcinogenesisJournal of Experimental & Clinical Cancer Research 2016 35(1):13910.1186/s13046-016-0418-827609145 [Google Scholar] [CrossRef] [PubMed]
[6]. Zhang SJ, Qian HN, Zhao Y, Sun K, Wang HQ, Liang GQ, Relationship between age and prostate sizeAsian Journal of Andrology 2013 15(1):11610.1038/aja.2012.12723223031 [Google Scholar] [CrossRef] [PubMed]
[7]. Hodson R, 4 BIG QUESTIONSNature 2015 528(7582):S13710.1038/528S137a26672789 [Google Scholar] [CrossRef] [PubMed]
[8]. Leskela S, Honrado E, Montero-Conde C, Landa I, Cascón A, Letón R, Cytochrome P450 3A5 is highly expressed in normal prostate cells but absent in prostate cancerEndocrine-Related Cancer 2007 14(3):645-54.10.1677/ERC-07-007817914095 [Google Scholar] [CrossRef] [PubMed]
[9]. Plummer SJ, Conti DV, Paris PL, Curran AP, Casey G, Witte JS, CYP3A4 and CYP3A5 genotypes, haplotypes, and risk of prostate cancerCancer Epidemiology and Prevention Biomarkers 2003 12(9):928-32. [Google Scholar]
[10]. Fujita K, Nonomura N, Role of androgen receptor in prostate cancer: A reviewThe World Journal of Men’s Health 2018 :3610.5534/wjmh.18004030209899 [Google Scholar] [CrossRef] [PubMed]
[11]. Husain I, Shukla S, Soni P, Husain N, Role of androgen receptor in prostatic neoplasia versus hyperplasiaJournal of Cancer Research and Therapeutics 2016 12(1):11210.4103/0973-1482.15142927072221 [Google Scholar] [CrossRef] [PubMed]
[12]. Mitsiades N, Sung CC, Schultz N, Danila DC, He B, Eedunuri VK, Distinct patterns of dysregulated expression of enzymes involved in androgen synthesis and metabolism in metastatic prostate cancer tumorsCancer Research 2012 72(23):6142-52.10.1158/0008-5472.CAN-12-133522971343 [Google Scholar] [CrossRef] [PubMed]
[13]. Vaarala MH, Mattila H, Ohtonen P, Tammela TL, Paavonen TK, Schleutker J, The interaction of CYP3A5 polymorphisms along the androgen metabolism pathway in prostate cancerInternational Journal of Cancer 2008 122(11):2511-16.10.1002/ijc.2342518306354 [Google Scholar] [CrossRef] [PubMed]
[14]. Mitra R, Gorjala P, Goodman OB, CYP3A5 positively regulates androgen receptor signaling in prostate cancer cellsPotential for Therapeutic Targeting 2017 :3130-30.10.1158/1538-7445.AM2017-3130 [Google Scholar] [CrossRef]
[15]. Zhenhua L, Tsuchiya N, Narita S, Inoue T, Horikawa Y, Kakinuma H, CYP3A5 gene polymorphism and risk of prostate cancer in a Japanese populationCancer Letters 2005 225(2):237-43.10.1016/j.canlet.2005.03.00915876487 [Google Scholar] [CrossRef] [PubMed]
[16]. Shetty PJ, Pasupuleti N, Chava S, Nasaruddin K, Hasan Q, Altered transcription and expression of PTEN in breast tumors: is it regulated by hypermethylation?Breast Disease 2012 33(1):27-33.10.3233/BD-2010-031221846942 [Google Scholar] [CrossRef] [PubMed]
[17]. Poornima S, Subramanyam K, Khan IA, Hasan Q, The insertion and deletion (I28005D) polymorphism of the angiotensin I converting enzyme gene is a risk factor for osteoarthritis in an Asian Indian populationJournal of the Renin-Angiotensin-Aldosterone System 2015 16(4):1281-87.10.1177/147032031454740325178458 [Google Scholar] [CrossRef] [PubMed]
[18]. Shaik NA, Govindan S, Kodati V, Rao KP, Hasan Q, Polymorphic (CAG) n repeats in the androgen receptor gene: A risk marker for endometriosis and uterine leiomyomasHematology/Oncology and Stem Cell Therapy 2009 2(1):289-93.10.1016/S1658-3876(09)50040-4 [Google Scholar] [PubMed]
[19]. Rao P, Influence of gene polymorphism on the pharamacokinetics of calcineurin inhibitors: In renal transplant patients from IndiaInternational Research Journal of Pharmacy and Pharmacology 2013 3(1):09-15. [Google Scholar]
[20]. Chen N, Zhou Q, The evolving Gleason grading systemChin J Cancer Res 2016 28(1):58-64. [Google Scholar]
[21]. Zeigler-Johnson C, Friebel T, Walker AH, Wang Y, Spangler E, Panossian S, CYP3A4, CYP3A5, and CYP3A43 genotypes and haplotypes in the etiology and severity of prostate cancerCancer Research 2004 64(22):8461-67.10.1158/0008-5472.CAN-04-165115548719 [Google Scholar] [CrossRef] [PubMed]
[22]. Kirkman M, Young K, Evans S, Millar J, Fisher J, Mazza D, Men’s perceptions of prostate cancer diagnosis and care: insights from qualitative interviews in Victoria, AustraliaBMC Cancer 2017 17(1):70410.1186/s12885-017-3699-129078768 [Google Scholar] [CrossRef] [PubMed]
[23]. Ulmert D, Cronin AM, Björk T, O’Brien MF, Scardino PT, Eastham JA, Prostate-specific antigen at or before age 50 as a predictor of advanced prostate cancer diagnosed up to 25 years later: A case-control studyBMC Medicine 2008 6(1):610.1186/1741-7015-6-618279502 [Google Scholar] [CrossRef] [PubMed]
[24]. Carlsson S, Assel M, Ulmert D, Gerdtsson A, Hugosson J, Vickers A, Screening for prostate cancer starting at age 50-54 years. A population-based cohort studyEuropean Urology 2017 71(1):46-52.10.1016/j.eururo.2016.03.02627084245 [Google Scholar] [CrossRef] [PubMed]
[25]. Chen SL, Fann JC, Sipeky C, Yang TK, Chiu SY, Yen AM, Risk prediction of prostate cancer with Single Nucleotide Polymorphisms (SNPs) and Prostate-Specific Antigen (PSA)The Journal of Urology 2018 24 [Google Scholar]
[26]. Lakes J, Arsov C, PSA-Screening und molekulare markerDer Urologe 2019 14:01-06.10.1007/s00120-019-0900-y30874831 [Google Scholar] [CrossRef] [PubMed]
[27]. Wang G, Zhao D, Spring DJ, DePinho RA, Genetics and biology of prostate cancerGenes & Development 2018 32(17-18):1105-40.10.1101/gad.315739.11830181359 [Google Scholar] [CrossRef] [PubMed]
[28]. Qin Z, Li X, Han P, Zheng Y, Liu H, Tang J, Association between polymorphic CAG repeat lengths in the androgen receptor gene and susceptibility to prostate cancer: A systematic review and meta-analysisMedicine 2017 96(25)10.1097/MD.000000000000725828640128 [Google Scholar] [CrossRef] [PubMed]
[29]. Madjunkova S, Eftimov A, Georgiev V, Petrovski D, Dimovski A, Plaseska-Karanfilska D, CAG repeat number in the androgen receptor gene and prostate cancerBalkan Journal of Medical Genetics 2012 15(1):31-36.10.2478/v10034-012-0005-z3776652 [Google Scholar] [CrossRef] [PubMed]
[30]. Gómez R, Torres-Sánchez L, Camacho-Mejorado R, Burguete-García AI, Vázquez-Salas RA, Martínez-Nava GA, Androgen receptor CAG polymorphism and sporadic and early-onset prostate cancer among Mexican menJournal of Human Genetics 2016 61(9):78110.1038/jhg.2016.4927193223 [Google Scholar] [CrossRef] [PubMed]
[31]. Mishra DK, Thangara K, Mandhani A, Kumar A, Mittal RD, Is reduced CAG repeat length in androgen receptor gene associated with risk of prostate cancer in Indian population?Clinical Genetics 2005 68(1):55-60.10.1111/j.1399-0004.2005.00450.x15952987 [Google Scholar] [CrossRef] [PubMed]
[32]. Huang SP, Chou YH, Chang WS, Wu MT, Yu CC, Wu T, Androgen receptor gene polymorphism and prostate cancer in TaiwanJournal of the Formosan Medical Association= Taiwan yi zhi 2003 102(10):680-86. [Google Scholar]
[33]. Zhou Y, Ingelman-Sundberg M, Lauschke VM, Worldwide distribution of cytochrome P450 alleles: A meta-analysis of population-scale sequencing projectsClinical Pharmacology & Therapeutics 2017 102(4):688-700.10.1002/cpt.69028378927 [Google Scholar] [CrossRef] [PubMed]
[34]. Liang Y, Han W, Yan H, Mao Q, Association of CYP3A5* 3 polymorphisms and prostate cancer risk: A meta-analysisJournal of Cancer Research and Therapeutics 2018 14(9):46310.4103/0976-7800.1791734832898 [Google Scholar] [CrossRef] [PubMed]
[35]. Moilanen AM, Hakkola J, Vaarala MH, Kauppila S, Hirvikoski P, Characterization of androgen-regulated expression of CYP3A5 in human prostateCarcinogenesis 2006 28(5):916-21.10.1093/carcin/bgl22217116727 [Google Scholar] [CrossRef] [PubMed]
[36]. Sribudiani Y, Marwan DW, Aulanni’am A, Widodo MA, Purnomo BB, Panigoro R, Germline mutations and polymorphisms of androgen receptor in prostate cancer patients: frequency and results of in silico analysisAsian Pacific Journal of Cancer Prevention 2018 19(8):2241 [Google Scholar]
[37]. Zeegers MP, Kiemeney LA, Nieder AM, Ostrer H, How strong is the association between CAG and GGN repeat length polymorphisms in the androgen receptor gene and prostate cancer risk?Cancer Epidemiology and Prevention Biomarkers 2004 13(11):1765-71. [Google Scholar]
[38]. Crawford ED, Heidenreich A, Lawrentschuk N, Tombal B, Pompeo AC, Mendoza-Valdes A, Androgen-targeted therapy in men with prostate cancer: evolving practice and future considerationsProstate Cancer and Prostatic Diseases 2018 21:110.1038/s41391-018-0079-030131604 [Google Scholar] [CrossRef] [PubMed]
[39]. Armstrong CM, Gao AC, Current strategies for targeting the activity of androgen receptor variantsAsian Journal of Urology 2018 6(1):42-49.10.1016/j.ajur.2018.07.00330775247 [Google Scholar] [CrossRef] [PubMed]