Rare Presentation of Primary GI Lymphoma with Synchronous Lesions in a Retropositive Patient
Gursimran Singh1, P Ganesh2, S Shanmuganathan3
1 Postgraduate, Department of Medical Gastroenterology, Sri Ramachandra Medical College and Research Institute, Chennai, Tamil Nadu, India.
2 Professor and Head, Department of Medical Gastroenterology, Sri Ramachandra Medical College and Research Institute, Chennai, Tamil Nadu, India.
3 Professor, Department of Medical Gastroenterology, Sri Ramachandra Medical College and Research Institute, Chennai, Tamil Nadu, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Gursimran Singh, Prestige Bella Vista, Tower-8, Level-6, Flat No: 8072, Iyyapanthangal, Chennai-600056, Tamil Nadu, India.
E-mail: gursimran1725@yahoo.in
Non Hodgkin’s Lymphoma (NHL) with primary Gastrointestinal (GI) involvement occurs in 27-74% of all HIV infected persons. Most common site of involvement is stomach, small intestine and ileocaecal region. Synchronous GI lesions are quite rare presentations in lymphoma. There are very few case reports of synchronous lesions in lymphoma, but no reports of the same in primary GI lymphoma. These tumours behave aggressively and need early diagnosis and treatment.
Here, we present the case of a 48-year-old retropositive male patient, presenting with jaundice and significant weight loss. On CT whole abdomen, he was found to have gastric and colonic lesions with perigastric nodes and nodes at porta hepatis. Presence of B-cell type of lymphoma was diagnosed by histopathological examination and confirmed with immunohistochemistry. Chemotherapy was initiated and he responded to the same. This case is presented for its rarity, whereby synchronous gastric and colonic lymphomas are seen in retropositive patient.
Chemotherapy, Extranodal, Human immunodeficiency virus, Immunosuppression
Case Report
A 48-year-old gentleman, smoker and tobacco chewer presented with one month history of dark yellow coloured urine and yellowish discolouration of eyes. He also had watery diarrhoea not mixed with blood and mucous but associated with abdominal pain, localised to right iliac region. He had a significant weight loss of approximately 4 kg within that month. He did not have fever, bone pain or night sweats.
At the time of presentation, he was found to have pale skin with yellow eyes presenting as anaemia and jaundice. Blood pressure in right arm was 128/80 mmHg, Heart Rate was 80/minute and regular, Respiratory Rate 16/minute, Temperature was afebrile. Examination of abdomen revealed no organomegaly or palpable mass. No relevant family and social history was present.
Blood investigations revealed Hb- of 10 g/dL and raised ESR. He had a Total bilirubin of 2.91 mg/dL with Indirect bilirubin- 1.27 mg/dL. Liver function test revealed raised transaminases with SGOT- 115 U/L, SGPT- 98 U/L, Total protein- 8.2 g/dL, Albumin- 1.6 g/dL, Globulin- 6.6 g/dL and Alkaline phosphatase- 476 U/mL. He was tested positive for retrovirus with CD4 count of 287. He was also tested positive for Hepatitis B surface antigen. Stool examination was not contributory with no inflammatory cells. These findings were suggestive of retropositivity with acute hepatitis B.
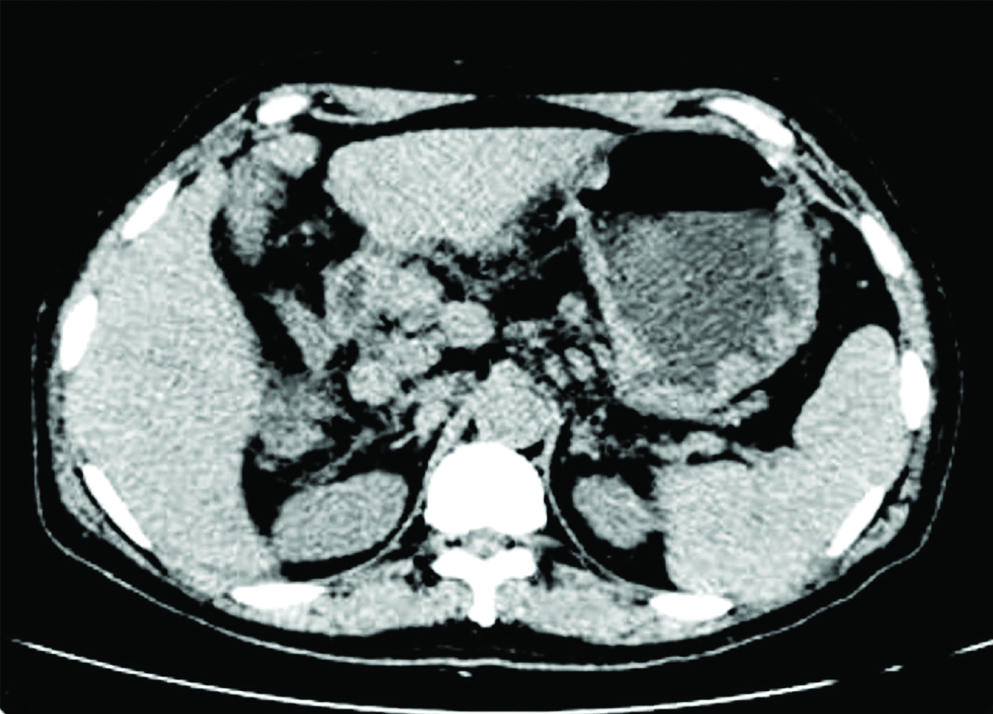
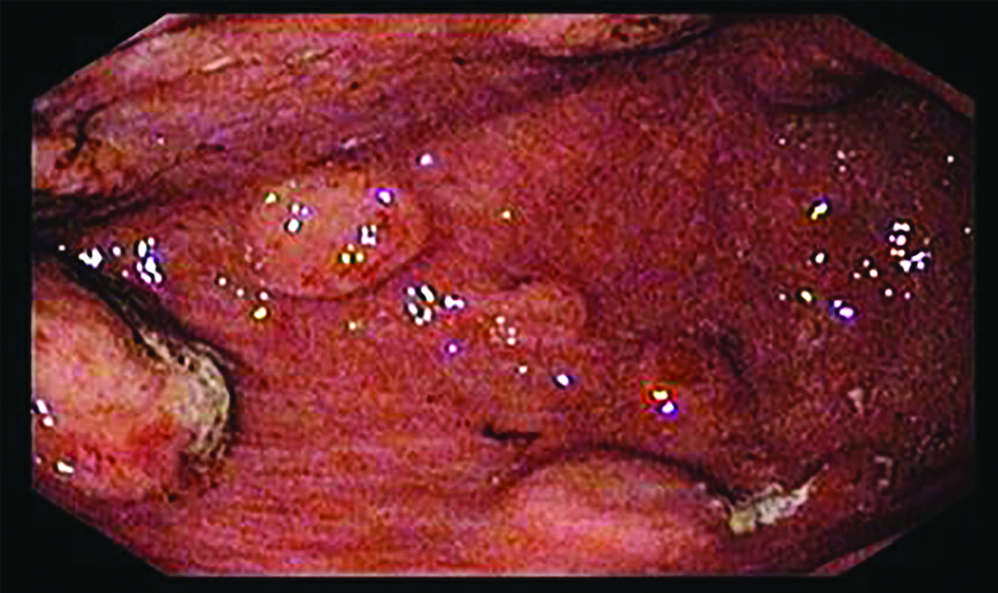
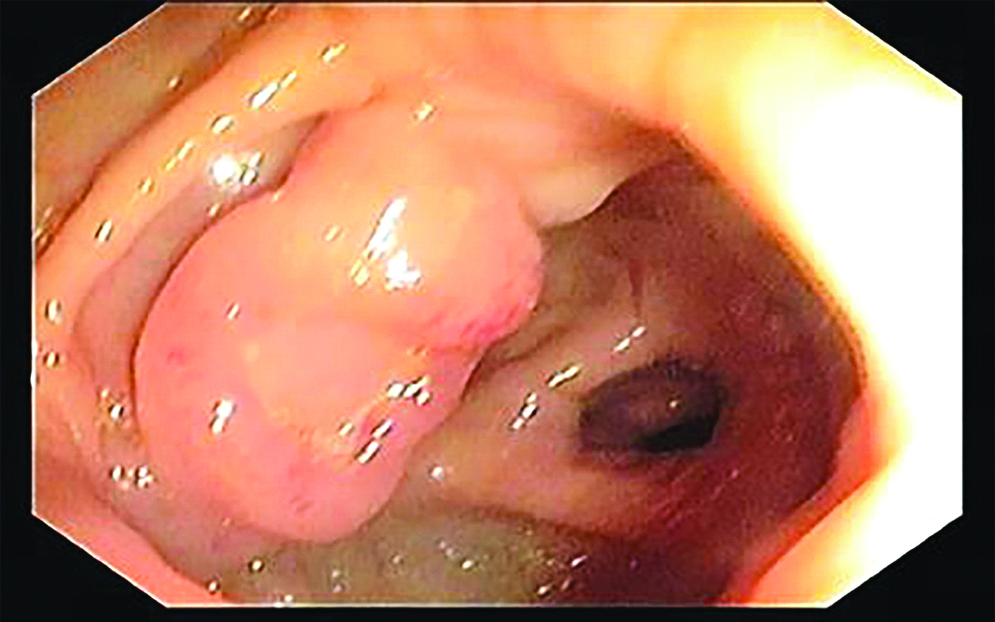
Initial evaluation with USG Abdomen showed multiple enlarged nodes at portahepatis. Hence we went ahead with CT Abdomen which showed few enlarged lymph nodes in portahepatis, largest measuring 1.3x0.9 cm. Rest of the organs were reported as normal including the liver and spleen [Table/Fig-1]. We proceeded with a gastroscopy which showed multiple nodules with central umblications in body and antrum of stomach [Table/Fig-2]. Colonoscopy was also done which showed nodular lesions of size 6cm in transverse colon and another lesion at the caecal pole [Table/Fig-3].
CT Abdomen showing multiple enlarged para aortic lymph, porto hepatic, mesentric nodes and gastric nodules.
Gastroscopy showing multiple nodules with central umblication of varying size ranging 5 mm to 2.0 cm.
Colonoscopy showing nodular lesions in transverse colon and caecum.
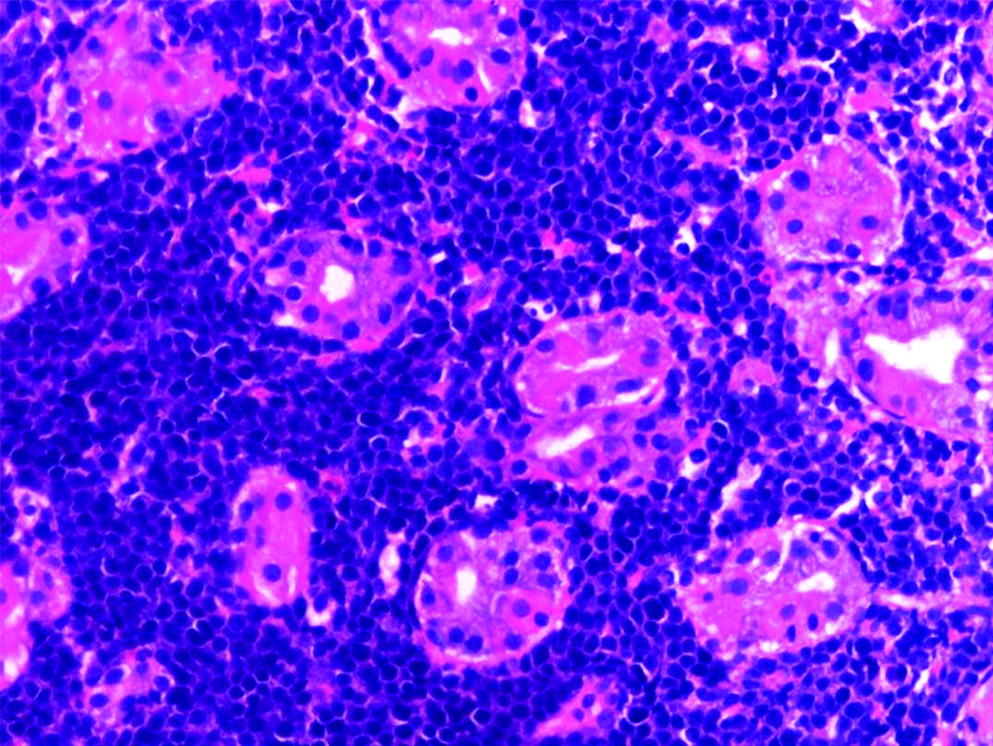
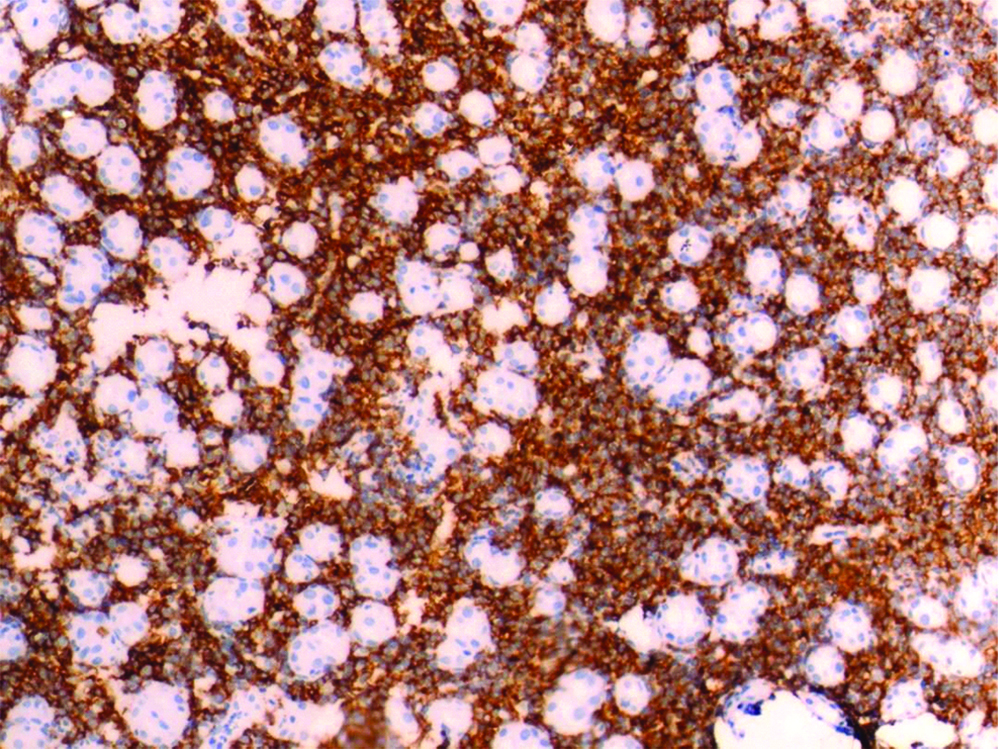
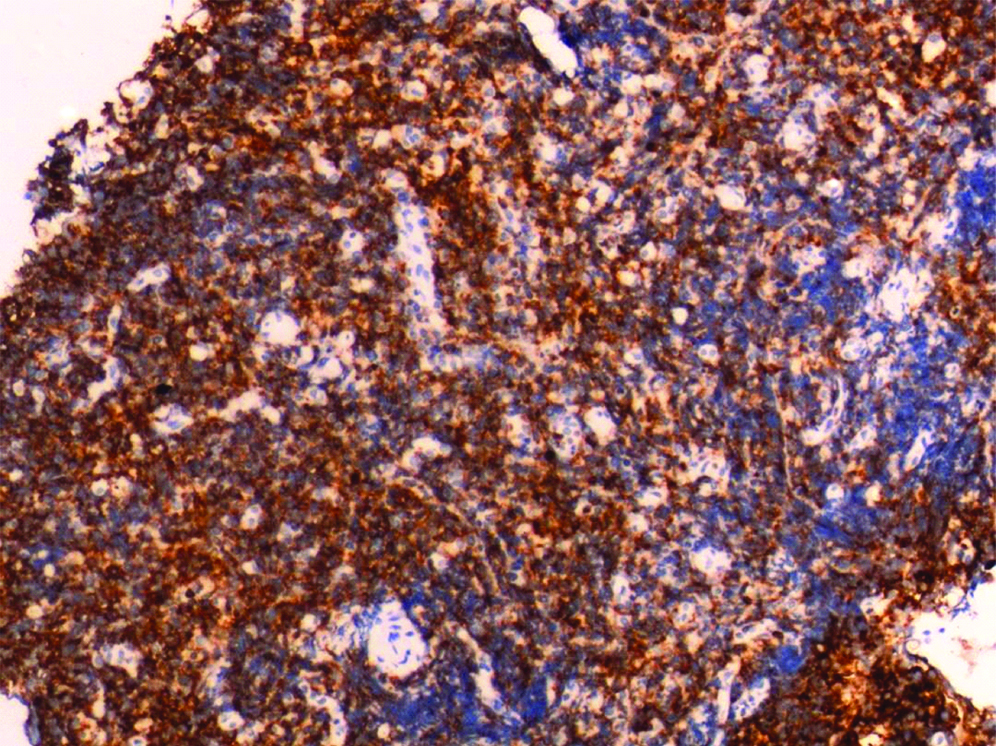
Histopathological examination revealed fragments of gastric mucosa with infiltration of lamina propria by sheets neoplastic cells having round to oval nuclei with condensed chromatin and scanty cytoplasm. Atypical mitosis and apoptotic cells are noted [Table/Fig-4]. Immunohistochemiostry showed positivity for CD- 20 confirming it to be B-Cell Lymphoma. IHC was also positive for Vimentin [Table/Fig-5] and CD-45 [Table/Fig-6]. Immunohistochemistry showed diffuse and strong staining for CD20+ and BCl-6 and negative staining for CD30 and MUM-1. Proliferation index was 80-90% as accessed by Ki67. Immunostainings for keratins (AE1-AE3), Epstein-Barr Virus (EBV), BCl-2 and cyclin-D1 were negative. In situ hybridization for EBV was negative. Similar findings were seen in colonic mucosa with infiltration of lamina propria by sheets of monomorphous population of cells with occasional mitosis and apoptotic bodies noted. Bone marrow biopsy was normal as was CT Thorax. All these investigations confirmed the presence of Primary GI Lymphoma (B-Cell type). Patient was started on chemotherapy comprising R-CHOP regimen. He was also started on HAART treatment. After nine months, patient had no complains of diarrhoea and abdominal pain. Colour of skin had changed from pale to his actual skin tone and dark yellow coloured urine had turned light yellow. A relook colonoscopy after one year showed resolution of the nodule. A repeat scan after one year showed persistence of few para aortic and peri gastric lymph with reduced size. Patient was continued on chemotherapy. He was also given antivirals, Tab.Entecavir 0.5 mg OD for hepatitis B infection. He remains on follow-up and has shown weight gain from 55 kg at time of presentation to 64 kg after one year.
HPE 400X showed gastric mucosa with infiltration of lamina propria by sheets of neoplastic cells having round to oval nuclei with condensed chromatin and scanty cytoplasm. Atypical mitosis and apoptotic cells are noted.
IHC showing strong membrane positivity for CD-20 confirming it to be B-Cell lymphoma. Lesional cells are also showing strong diffuse cytoplasmic positivity for Vimentin (Vimentin is cytoplasmic and CD 20 is nuclear).
IHC showing positivity for CD-45.
Discussion
HIV infected patients are at increased risk of developing malignancies including lymphoma but more commonly Kaposi sarcoma. Up to 80% of the HIV-associated NHL arise systemically with primary GI lymphoma being rare. The definition of a primary GI lymphoma was given by Dawson et al., the five components include [1]: a) absence of peripheral lymphadenopathy at the time of presentation; b) lack of enlarged mediastinal lymph nodes; c) normal total and differential white blood cell count; d) predominance of bowel lesion at the time of laparotomy with only lymph nodes obviously affected in the immediate vicinity; and e) no lymphomatous involvement of liver and spleen. All these criteria were fulfilled in our case justifying the diagnosis of primary GI lymphoma. The most frequent site of involvement is the stomach (60%-70%) followed by the small intestine (20-30%) as reported in a number of studies [2-5].
Among the histological types of primary GI lymphoma, B-cell lymphoma is far more frequent (85.5%) than T-cell lymphoma. Diffuse Large B-cell Lymphoma (DLBCL) is the the most frequent immunophenotype of primary GI lymphoma [5,6]. Gastric lymphomas can be MALT lymphoma or DLBCL and present as ulcers, polypoidal mass, thickened fold, mucosal nodularity or linitis plastica. Colonic lymphomas are predominantly of T-cell lymphoma type with rare occurrence of DLBCL and present as polypoidal mass, ulcer, mucosal nodularity cavitary mass, mucosal thickening and strictures the sub mucosal location and innocuous appearance of the lesion in endoscopy makes the diagnosis more difficult and diagnosis by endoscopic biopsy has been reported in 51-55% of cases the rest requiring surgical confirmation [7]. In our case diagnosis was done by endoscopic biopsy, histopathological examination and immunohistochemistry of the same.
Staging of the primary GI-DLBCL is performed by consideration of the imaging studies, as well as bone marrow aspiration and biopsy. According to the Lugano staging, DLBCL stage I is limited to the GI tract, stage II is extended to the abdomen, stage IIE is defined as adjacent tissue involvement by serosa penetration, and stage IV disseminates to extra-nodal regions or is concomitant supradiaphragmatic nodal involvement [8].
The choice of treatment for DLBCL is chemotherapy which is further adjusted as per age and international prognostic index regimen of choice is CHOP with rituximab being added for CD 20 positive cases [9]. The International Prognostic Index (IPI) is calculated using the following factors: age >60 years, increased serum LDH, performance status ≥2, more than one extra-nodal involvement excluding the stomach (as in this case), and stage III–IV of the disease [10]. Li X et al., demonstrated treatment response, and elevated serum LDH level as independent prognostic factors of survival for DLBCL [11]. Nakamura S et al., identified that an earlier stage, younger age, gastric localization, B-cell phenotype, and absence of B symptoms were independent prognostic factors for better optimum survival for primary GI lymphoma [2]. Dual modality of treatment with surgery and chemotherapy offers the best survival opportunity for patients in early stage [2,12]. There are many studies of primary GI lymphoma which report a male predominance and frequent occurrence in 6th decade [3-5,13]. But there are very few case reports of synchronous primary GI lymphoma in literature. Corti M et al., reported a case of Burkitts lymphoma of the duodenum who presented with abdominal pain and "B" symptoms but did not respond to treatment [14]. Sujata SG et al., has reported primary colonic lymphoma that responded to surgery and chemotherapy [15]. This is the first case report of synchronous primary GI lymphoma with multiple lesions involving both stomach and colon.
Conclusion
Primary GI lymphoma with synchronous lesions in stomach and colon is an extremely rare presentation but diagnosis can be made by endoscopic biopsy requiring a high index of suspicion. The most common site of primary GI lymphoma is the stomach even in immunocompromised patients. Although dual therapy gives best outcomes in primary GI lymphoma, this case demonstrates that chemotherapy is equally effective when synchronous lesions occur.
[1]. Dawson IM, Cornes JS, Morson BC, Primary malignant lymphoid tumours of the intestinal tract. Report of 37 cases with a study of factors influencing prognosisBr J Surg 1961 49:80-89.10.1002/bjs.1800492131913884035 [Google Scholar] [CrossRef] [PubMed]
[2]. Nakamura S, Matsumoto T, Iida M, Yao T, Tsuneyoshi M, Primary gastrointestinal lymphoma in Japan: A clinicopathologic analysis of 455 patients with special reference to its time trendsCancer 2003 97:2462-73.10.1002/cncr.1141512733145 [Google Scholar] [CrossRef] [PubMed]
[3]. Ghimire P, Wu GY, Zhu L, Primary gastrointestinal lymphomaWorld J Gastroenterol 2011 17(6):697-707.10.3748/wjg.v17.i6.69721390139 [Google Scholar] [CrossRef] [PubMed]
[4]. Shirsat HS, Vaiphei K, Primary gastrointestinal lymphomas- A study of 81 cases from a Tertiary Healthcare CentreIndian J Cancer 2014 51:290-92.10.4103/0019-509X.14677725494124 [Google Scholar] [CrossRef] [PubMed]
[5]. Juárez-Salcedo LM, Sokol L, Chavez JC, Dalia S, Primary gastric lymphoma, epidemiology, clinical diagnosis, and treatmentCancer Control 2018 25(1):107327481877825610.1177/107327481877825629779412 [Google Scholar] [CrossRef] [PubMed]
[6]. Matysiak-Budnik T, Jamet P, Fabiani B, Nion-Larmurier I, Marjanovic Z, Ruskoné-Fourmestraux A, Primary intestinal B-cell lymphoma: A prospective multicentre clinical study of 91 casesDig Liver Dis 2013 45:947-52.10.1016/j.dld.2013.05.00823816692 [Google Scholar] [CrossRef] [PubMed]
[7]. Ran W, Ouyang Q, Primary intestinal Non Hodgkin’s lymphoma: a retrospective study of 85 casesWorld J Gastroenterol 2011 17(41):4625-31.10.3748/wjg.v17.i41.462522147970 [Google Scholar] [CrossRef] [PubMed]
[8]. Selcukbiricik F, Tural D, Elicin O, Berk S, Ozguroglu M, Bese N, Primary gastric lymphoma: Conservative treatment modality is not inferior to surgery for early-stage diseaseISRN Oncol 2012 2012:95181610.5402/2012/95181622988526 [Google Scholar] [CrossRef] [PubMed]
[9]. Sarid N, Joffe E, Gibstein L, Avivi I, Polliack A, Perry C, Reduced-dose ICE chemotherapy +/- rituximab is a safe and effective salvage therapy for fit elderly patients with diffuse large B-cell lymphomaLeuk Lymphoma 2016 57(7):1633-39.10.3109/10428194.2015.110653226643787 [Google Scholar] [CrossRef] [PubMed]
[10]. Rotaru I, Gaman GD, Stanescu C, Gaman AM, Evaluation of parameters with potential prognosis impact in patients with primary gastric diffuse large B-cell lymphoma (PG-DLB- CL)Rom J Morphol Embryol 2014 55(1):15-21. [Google Scholar]
[11]. Li X, Shen W, Cao J, Wang J, Chen F, Wang C, Treatment of gastrointestinal diffuse large B cell lymphoma in China: A 10-year retrospective study of 114 casesAnn Hematol 2012 91:1721-29.10.1007/s00277-012-1507-122733613 [Google Scholar] [CrossRef] [PubMed]
[12]. Chen Y, Chen Y, Chen S, Wu L, Xu L, Lian G, Primary gastrointestinal lymphoma: A retrospective multicenter clinical study of 415 cases in Chinese province of Guangdong and a systematic review containing 5075 Chinese patientsMedicine (Baltimore) 2015 94:e211910.1097/MD.000000000000211926632732 [Google Scholar] [CrossRef] [PubMed]
[13]. Aledavood A, Nasiri MR, Memar B, Shahidsales S, Raziee HR, Ghafarzadegan K, Primary gastrointestinal lymphomaJ Res Med Sci 2012 17(5):487-90. [Google Scholar]
[14]. Corti M, Villafane MF, Souto L, Schtirbu R, Narbaitz M, Soler MD, Burkitt’s lymphoma of the duodenum in a patient with AIDSRev Soc Bras Med Trop 2007 40:338-40.10.1590/S0037-8682200700030001717653472 [Google Scholar] [CrossRef] [PubMed]
[15]. Sujata SG, Garima J, Non Hodgkin lymphoma of caecum-A case reportIosr-JDMS 2015 14(1):90-92. [Google Scholar]