Acute abdomen, Bladder carcinoma, Peritonitis, Squamous cell carcinoma, Uroperitoneum
Case Report
A 70-year-old male patient presented to the emergency with a history of gradual abdominal distension, nausea, vomiting, passage of loose stools with diffuse pain abdomen and somnolence for the past three days. There was no history of abdominal trauma or urinary complaints in the past. Patient was non-diabetic non-hypertensive with no significant past illnesses and no history of chronic medication.
On initial examination, patient was drowsy but was following commands. Detailed examination revealed adequate hydration, heart rate of 80/min, blood pressure 100/70 mm of Hg, and a respiratory rate of 26/min. Patient was afebrile; his chest auscultation revealed slight bibasal creptitations. He had a soft but distended abdomen with diffuse tenderness but no signs of peritonitis. Digital rectal examination was within normal limits.
He was managed conservatively with Ryle’s tube insertion, fluid resuscitation and all blood parameters were sent for examination. Foley catheterization yielded clear and adequate urine. A straight X-ray of abdomen in erect posture was done, the findings of which were non-specific. An ultrasonography of the abdomen on the next day revealed a large amount of intraperitoneal loculated fluid collection suggestive of complex ascites or intraabdominal abscess and a thickened urinary bladder with a 6x4.5 cm2 soft tissue mass arising from the posterosuperior aspect. Blood reports showed significantly raised urea and creatinine levels (urea 60 mg/dL, creatinine 2.6 mg/dL), hyponatremia (120 mmol/L), hyperkalemia (5.9 mmol/L) and a total count of 15000/cu mm. By the next day, the patient had developed frank peritonitis and surgical intervention was promptly planned.
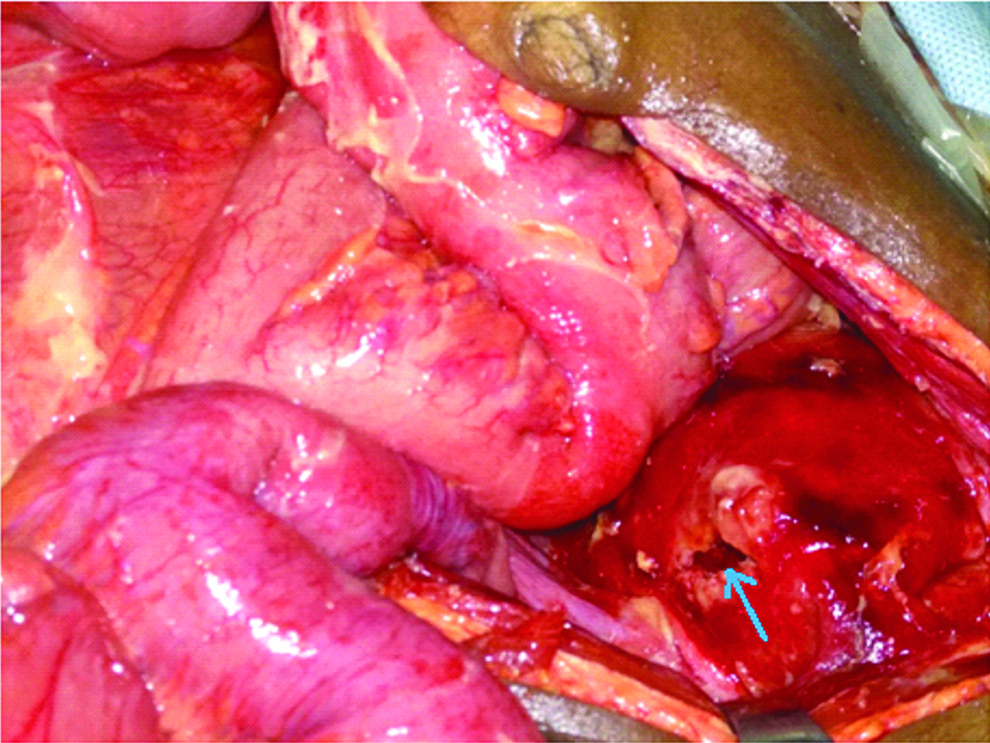
Exploratory laparotomy was undertaken through vertical midline incision. Abdominal cavity was noted to be full of clear straw colored liquid with loculated collection within gut loops. Gut loops were moderately distended with whitish inflammatory exudates on the surface. A thorough search for source of pathology revealed a hard mass involving whole of the urinary bladder, free from the adjacent structures and rectal surface. The mass had fungated through the right posterosuperior surface resulting in spontaneous intraperitoneal bladder perforation and uroascites [Table/Fig-1]. The rent was freshened, biopsies taken and thereafter repaired using 1-0 vicryl with interrupted sutures. Thorough peritoneal lavage was done, a drain was placed in the pelvis and abdomen was closed in layers. Specimens were sent for histopathology and the Foley catheter was replaced by a three way bladder wash catheter as slow postoperative lavage was planned to prevent blockade by pieces of necrotic soft tissue that had started evacuating into the urobag.
Spontaneous rupture site on right posterosuperior aspect of urinary bladder (marked by blue arrow). Note presence of inflammatory exudates on gut wall serosa.
Postoperatively patient was initially doing well with occasional dyselectrolytemia and chest complications developing on postoperative day three with severe cough with expectoration. He was managed with iv Meropenem 1g TDS and iv Moxifloxacin 400 mg OD with nebulisation and other supportive care. Day 4 investigations revealed a deterioration in renal status and total count of 28000/cu.mmn. Persistent hyponatremia and hyperkalemia developed simultaneously and patient was managed accordingly. Abdominal drain output continued to be around 100 mL/day till day 3, catheter aspiration was done regularly to ensure lumen patency, but on day 4 output increased to 200 mL/day. After a catheter change was performed on day 4, the urinary output increased significantly with gradual reduction in drain output which allowed eventual removal on day 10.
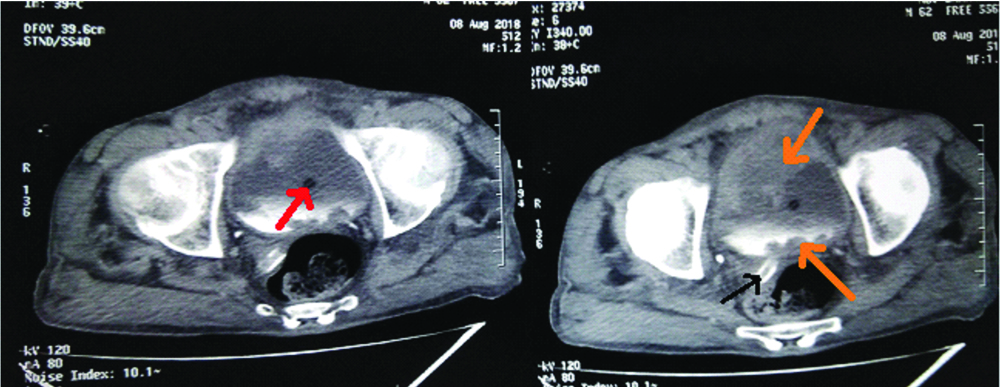
A postoperative contrast enhanced computed tomography scan was done on advice of the urosurgery department which showed a polypoid urinary bladder lesion-possibly, transitional cell cancer involving anterior and posterosuperior surface [Table/Fig-2,3]. Patient made a slow postoperative recovery and could be discharged in good health from our care two weeks after the surgery on referral to the Department of Urosurgery and Oncology with indwelling Foley catheter.
Postoperative computerised tomography of the abdomen-cross sectional cuts of pelvis showing polypoid bladder growth (orange arrows) protruding into bladder lumen containing contrast excreted in urine. Notably there is no active leak. Red arrow marks the catheter in situ; black arrow marks the intra abdominal drain.
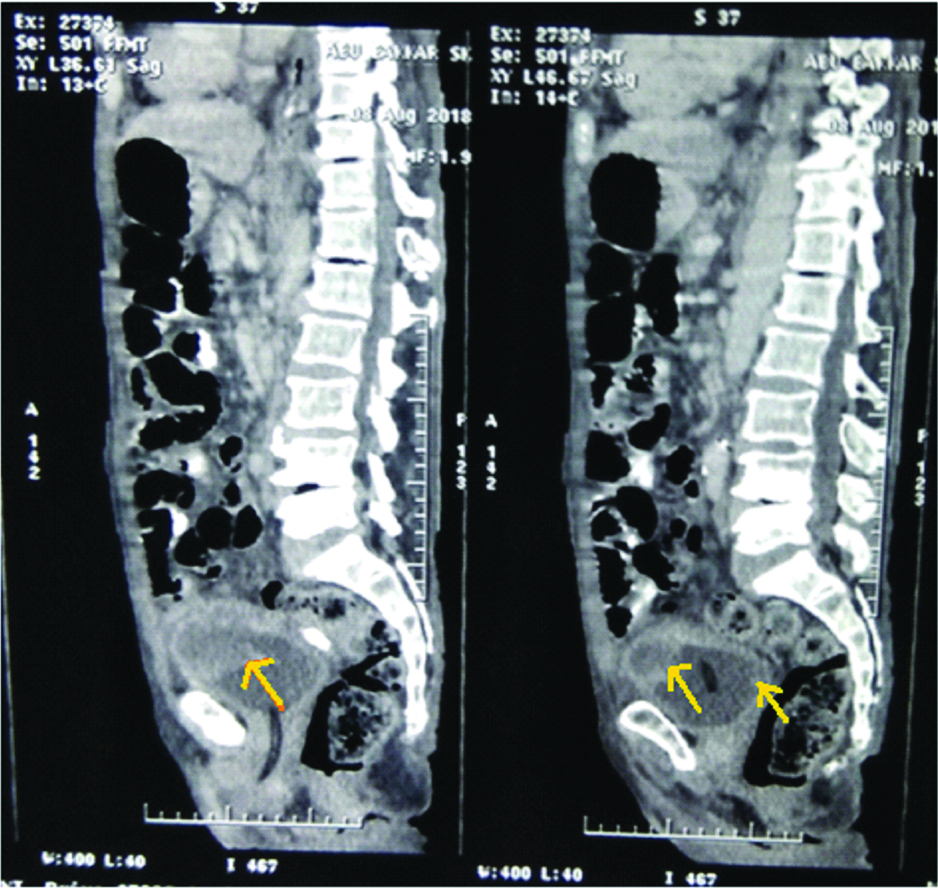
Postoperative comuterised tomography of abdomen-saggital sections showing bladder growth in anterior and posterior superior surfaces (yellow arrows). The Foley catheter is visible in the urethra in the initial cut and in the bladder lumen in the next cut.
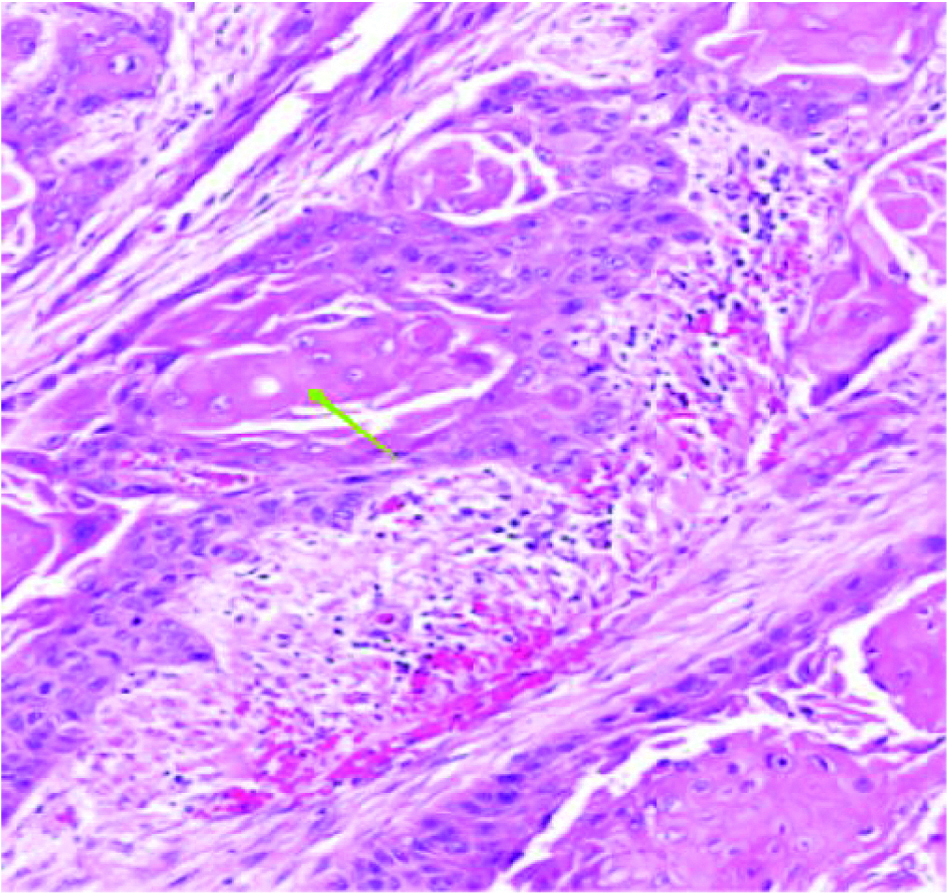
At follow-up, histopathological examination of the bladder biopsies showed presence of squamous cell cancer [Table/Fig-4].
histopathological examination slide of biopsy specimen obtained from bladder rupture site (HE stain, 10X magnification) showing keratin deposition (green arrow) suggestive of squamous cell carcinoma.
Discussion
Most cases of bladder rupture presenting to the emergency are traumatic in origin and found in association with blunt or penetrating injuries of the abdomen and perineum with a surprisingly high mortality of 12-22%. Separately, extra-peritoneal ruptures constitute 62% of cases, intra-peritoneal ruptures constitute 25%, and they are found to be combined in 12% cases [1]. Eighty three percent of all bladder ruptures have been found to be accompanied by a pelvic fracture [2].
Spontaneous intraperitoneal bladder rupture is an uncommon event with subtle presentations. The incidence of spontaneous bladder rupture has been reported to be 1:126000; 79% of all cases are reported in men, with an overall mortality of 47% [3].
Pathophysiology of rupture has its origin either in overdistension and increased bladder pressure or weakening of bladder wall (either by inflammation or malignant infiltration). The former can be caused by urethral strictures, bladder outlet obstruction, neuropathic bladder, continuous bladder irrigation or alcohol binge drinking with nonvoiding due to alcohol stupor. Other causes of atraumatic bladder rupture include chronic inflammation like tuberculosis, malignancy, cystitis, long standing indwelling catheter and radiotherapy for pelvic malignancies [4,5].
This rare life-threatening event is often complicated by the fact that the presentations maybe vague and varied which inevitably leads to delayed diagnosis and intervention in a patient who is probably already in an advanced stage of the disease. The possible pathogenesis of bladder rupture in bladder cancer is precipitation of perforation on the weakened body wall by the tumour. The most frequent location for intraperitoneal perforation was the dome or the posterior wall of the bladder [6].
A majority of such patients will be presenting with diffuse abdominal pain with poor localizing signs, distension and passage of low volumes of urine (that maybe blood-tinged). There is invariably uroascites and peritonitis develops if the diagnosis is delayed. In this case, the patient had vague abdominal complaints with a picture of hyponatremia and pseudo renal failure initially, with peritonitis from the uroascites setting in soon afterwards.
Reverse autodialysis of urine across the peritoneum leads to an increase in serum urea and creatinine with severe dyselectrolytemia mimicking a picture of acute renal failure. This is often further complicated by episodes of vomiting, diarrhea and the ensuing intravenous fluid resuscitation. In this scenario, clear urine in the urobag and the negative history of abdominal trauma confounded the diagnosis and we worked with a differential diagnosis of a developing subacute intestinal obstruction.
For diagnosis, both conventional retrograde cystography and CT cystography are currently the golden standard; on ultrasonography or conventional CT the diagnosis is often missed [7]. A perforation sealed by bowel loop may lead to a false negative finding.
When diagnosed, extraperitoneal ruptures can be managed conservatively with a Foley catheter [8]. Surgical intervention is planned for intraperitoneal ruptures. Operative treatment includes drainage of urine from the peritoneal cavity, closing the rupture and instituting good vesicle drainage with placement of an intra abdominal drain in the pelvis for anticipated failure of repair. Early diagnosis and prompt surgical treatment decreases the morbidity and mortality associated with this condition A simple bedside paracentesis may be done and plasma and ascitic fluid levels of urea creatinine if measured can lead one to a diagnosis of uroascites.
A report by Hayashi W et al., concluded that the possibility of intraperitoneal bladder rupture should be considered if mesothelial cells are seen in the patient’s urine [9].
The association of SRUB with pelvic fractures and been described by Baxter NN et al., who stated in a recent article that women who undergo radiation therapy to the pelvis for cancers of the cervix, rectum, or anus are at an increased risk for pelvic fractures [10].
Basavaraj DR et al., recommend that conservative management of spontaneous perforation of the bladder following radiotherapy, with antibiotic and prolonged catheterization is a better option than surgical intervention [11].
The prognosis of spontaneous bladders rupture due to carcinoma is very poor. Most of patients died within months. The mortality rate can range from 25% to 80% depending on the time of diagnosis [6].
Squamous cell carcinoma of bladder is a rare pathological finding accounting for only 3-7% of bladder malignancies, and when found it is usually in the setting of chronic irritation of inflammation. A naturally high incidence is seen in Egypt with 80% of the squamous cell carcinoma being associated with schistosomiasis (bilharziasis). Non bilharzial squamous cell carcinoma of bladder occurs in association with chronic irritation due to bladder calculi, long standing indwelling catheter or chronically infected bladder diverticula. Presenting most commonly in the seventh decade, this type of carcinoma is often widespread at diagnosis with muscular invasion in more than 80% patients and metastases in about 10% patients [12].
Conclusion
In the relatively senior age group, one should maintain a high index of suspicion in patients who are relatively haemodynamically stable but present with an acute abdomen and in every patient with ascites, hyponatremia and biochemical evidence of renal failure, uroperitoneum should be considered as a differential diagnosis.
[1]. Santucci RA, Mcaninch JW, Bladder injuries: Evaluation and managementInt Braz J Urol 2000 26:408-14. [Google Scholar]
[2]. Chan DP, Abujudeh HH, Cushing GL Jr, Novelline RA, CT cystography with multiplanar reformation for suspected bladder rupture: Experience in 234 casesAJR Am J Roentgenol 2006 187:1296-302.10.2214/AJR.05.097117056919 [Google Scholar] [CrossRef] [PubMed]
[3]. Mukhopadhyay M, Jamadar NS, Das Chattopadhyay S, Karmakar NC, Bhattarai N, Mondal SK, Spontaneous rupture of the bladder: Report of two casesHellenic Journal of Surgery 2015 87(6):509-11.10.1007/s13126-015-0271-9 [Google Scholar] [CrossRef]
[4]. Ogawa S, Date T, Muraki O, Intraperitoneal urinary bladder perforation observed in a patient with an indwelling urethral catheterCase Rep Urol 2013 2013:76570410.1155/2013/76570424078894 [Google Scholar] [CrossRef] [PubMed]
[5]. Engin O, Yakan S, Yıldırım M, Kozacıoğlu Z, A rare cause of acute abdomen: bladder rupture due to catheterizationJ Clin Anal Med 2013 4:327-28.10.4328/JCAM.667 [Google Scholar] [CrossRef]
[6]. Addar MH, Stuart GC, Nation JG, Shumsky AG, Spontaneous rupture of the urinary bladder: a late complication of radiotherapy-case report and review of the literatureGynecol Oncol 1996 62:314-16.10.1006/gyno.1996.02348751568 [Google Scholar] [CrossRef] [PubMed]
[7]. Quagliano PV, Delair SM, Malhotra AK, Diagnosis of blunt bladder injury: A prospective comparative study of computed tomography cystography and conventional retrograde cystographyJ Trauma 2006 61(2):e410-21.Discussion 421e42210.1097/01.ta.0000229940.36556.bf16917459 [Google Scholar] [CrossRef] [PubMed]
[8]. Parker H, Hoonpongsimanont W, Vaca F, Lotfipour S, Spontaneous bladder rupture in association with alcoholic binge: A case report and review of the literatureJ Emerg Med 2009 37(4):e386-89.10.1016/j.jemermed.2007.03.04917976802 [Google Scholar] [CrossRef] [PubMed]
[9]. Hayashi W, Nishino T, Namie S, Obata Y, Furukawa M, Kohno S, Spontaneous bladder rupture diagnosis based on urinary appearance of mesothelial cells: A case reportJ Med Case Rep 2014 8:4610.1186/1752-1947-8-4624521453 [Google Scholar] [CrossRef] [PubMed]
[10]. Baxter NN, Habermann EB, Tepper JE, Durham SB, Virnig BA, Risk of pelvic fractures in older women following pelvic irradiationJAMA 2005 294:2587-93.10.1001/jama.294.20.258716304072 [Google Scholar] [CrossRef] [PubMed]
[11]. Basavaraj DR, Zachariah KK, Feggetter JG, Acute abdomen-remember spontaneous perforation of the urinary bladderJ R Coll Surg Edinb 2001 46:316-17. [Google Scholar]
[12]. Tekes A, Kamel IR, Chan TY, Schoenberg MP, Bluemke DA, MR imaging features of non-transitional cell carcinoma of the urinary bladder with pathological correlationAJR Am J Roentgenol 2003 180(3):779-84.10.2214/ajr.180.3.180077912591696 [Google Scholar] [CrossRef] [PubMed]