Introduction
Several variants in Subclavian Artery (SA) have been reported, yet its branching pattern has not been reappraised in the recent years.
Aim
To investigate the subclavian artery and its ramification to understand well its anatomy and surgical significance.
Materials and Methods
Thirty-two Subclavian arteries in 16 (8 male and 8 female) adult cadavers were dissected bilaterally and their origin, termination, course and branching pattern were investigated. The study was conducted in Sri Ramachandra Medical College and Research Institute, Porur, Chennai, India, from July 2012 to February 2014. Cadavers showing surgical intervention or any visible abnormalities in the region were excluded.
Results
The origin and termination of the Subclavian artery were normal throughout. The branching pattern could be classified into three types: Type I (20 arteries), Type II (11 arteries) and Type III (1 artery). The artery showed normal course in all cases except one with a transfixed course i.e., it perforated the Scalenus anterior muscle.
Conclusion
Comparison and analysis of the results with previous works showed that the origin, termination, course and branching pattern of the Subclavian artery exhibited conventionality. This information shall, thereby, contribute to literature and aid anatomists, head and neck surgeons, vascular surgeons and radiologists.
Introduction
The Subclavian Artery (SA) gains clinical significance due to its utilisation of cannulation during cardiopulmonary bypass in procedures involving the thoracic aorta and insertion for intra-aortic balloon pumps [1]. The part of the single trunked vessel of the upper extremity extending from its origin till the outer border of the first rib defines the SA. The first part of its course on either side differs in the length, direction and relations as the SA, on the right, originates from the Brachiocephalic Trunk, posterior to the sternoclavicular joint and from the aortic arch on the left side.
From a surgical perspective, it is studied as three divisions. The first segment on the right ascends a little above the clavicle, obliquely outwards, from its origin up to the inner border of the Scalenus Anterior Muscle (SAM). On the left, it ascends in a perpendicular fashion to the muscle; arising from the end of the Aortic Arch (AA), this vessel is longer and situated deeper in the chest [2].
The second part, very short, marks the highest point of the arch the artery describes, passing outwards, posterior to the SAM while the most superficial third portion lies between the SAM’s outer margin, associated superiorly to the clavicle, terminating at the lower border of the first rib, marking the beginning of the Axillary artery [2]. This most readily accessible portion of the SA is favoured for its ligation since it is located within the triangular space bound by the SAM in the front, the Omohyoid above and inferiorly by the clavicle [2]. On both sides, there is precision in similarity between the second and third portions. Occasionally, the SA is transfixed i.e., it perforates the SAM [2].
Of the four main branches of the SA, three originate from the first segment: the Vertebral Artery (VA) (the first and largest branch of the Subclavian) emerges from its upper and posterior part; the Thyroid axis {Thyrocervical Trunk (TCT)}, in front, appears as a short, thick trunk in close proximity to the inner margin of SAM; the Internal Mammary (Internal Thoracic) Artery (ITA) emerges below and opposite to the Thyroid axis, goes down to the anterior thoracic wall’s inner surface, traversing behind the clavicle. The TCT, immediately following its origination, ramifies into three arteries: the Inferior Thyroid artery (Inf.Thy.A), Suprascapular (also called Transverse Scapular) artery (SSA) and Transversalis colli {Transverse Cervical artery (TCA)}. Ascending Cervical artery (ACA) is the largest branch of the Inf.Thy.A. [2].
The Supreme Intercostal {Costocervical Trunk (CT)}, the fourth one, arises off the second portion, from its back and upper surface on the right, while arising to SAM’s inner margin on the left. Therefore, no branch is given off from the second portion of the artery on the left side [3].
The Superficial Cervical Artery (SCA) frequents as TCT’s separate branch and the Posterior or Dorsal Scapular Artery (DSA), from the third (rarely from the second) segment of the SA.
The branching pattern of the SA has been documented in various populations (Negro, White, Japanese, British) earlier [4-7]. This study records the same, after a considerable interval in time, in few cases in Southern India and is probably one of the first few studies in India, on the SA branching pattern. The classification in this study takes a lead from the prototype of TCT [7] suggested based on the study in the Japanese population.
Materials and Methods
This observational study was conducted from July 2012 to February 2014 at Sri Ramachandra Medical College and Research Institute, Porur, Chennai, India. Dissection was performed bilaterally in 16 (8 male and 8 female) adult cadavers and the Origin, Termination, Course and Branching pattern of the 32 SA were studied. The number of arteries of each type with the side and sex is specified. Ethical approval was obtained retrospectively (SRUPOC/2017/931).
Inclusion and Exclusion Criteria
The study was carried out in the cadavers utilised to teach the Medical students. However, cadavers showing surgical intervention or any visible abnormalities in the region were excluded.
Results
Origin and termination of the subclavian artery: The origin of the SA was observed to be conventional in all the specimens i.e., the Left SA took origin from the AA and the Right SA originated from the Brachiocephalic Trunk. All SA terminated at the outer border of the first rib.
Course of the subclavian artery: Normal course posterior to the SAM was observed in 31 SA (15 Left and 16 Right; 15 Male and 16 Female). One artery was transfixed (Left side, Male).
Branching Pattern of Subclavian Artery
The 32 arteries were classified based on the ramification of the Thyrocervical Trunk (TCT) [7] into 3 types:
Type I: Typical branching pattern of the TCT and hence, the SA;
Type II: The ACA directly arises from the TCT as one of its branch;
Type III: The TCT is absent and its branches directly make an appearance from the first segment of the Subclavian. One such artery was found [Table/Fig-1,2,3,4 and 5].
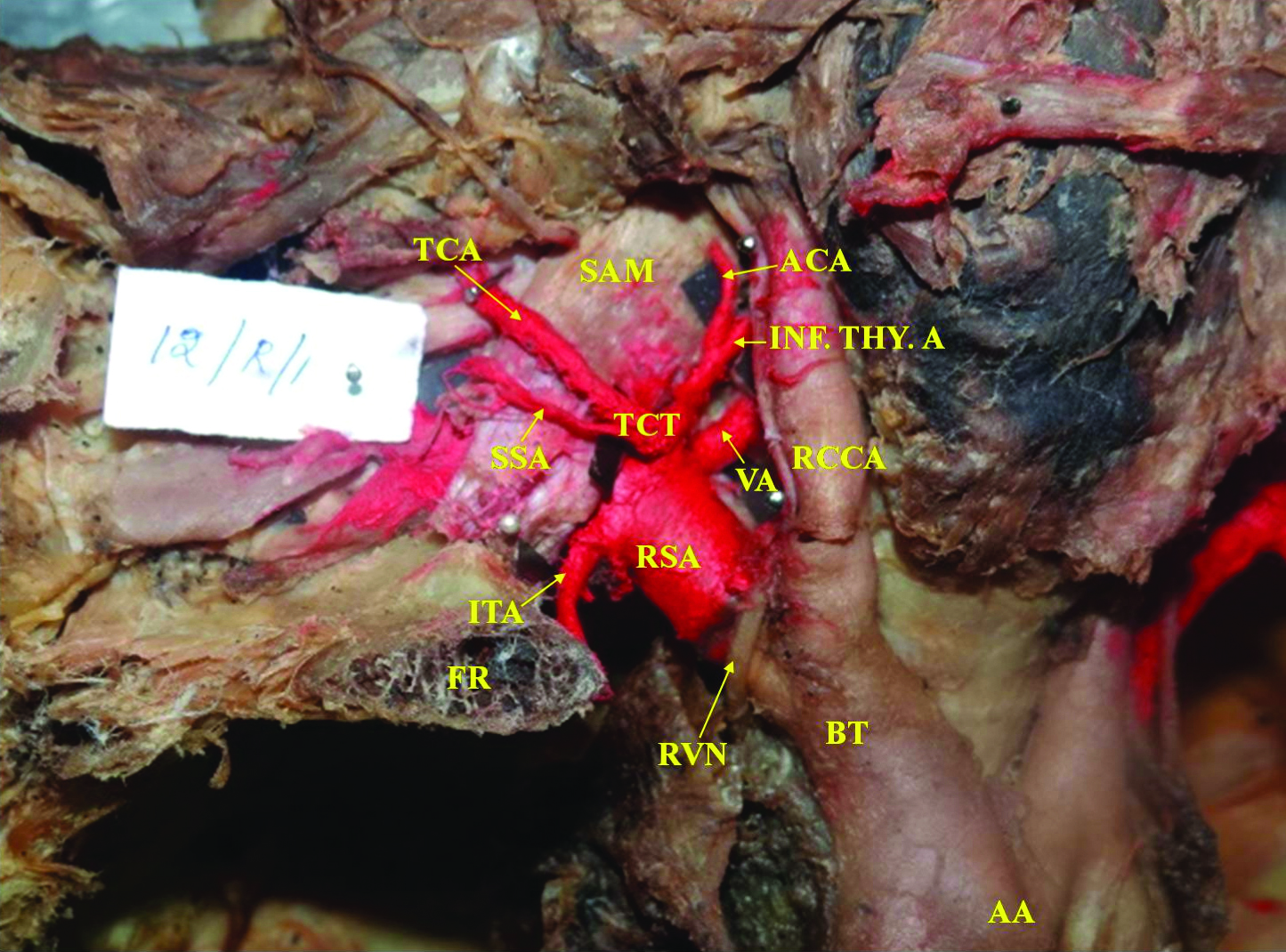
Branching Pattern Type I: Normal - Superficial Branches: RSA – Right Subclavian Artery, VA-Vertebral Artery, ITA-Internal Thoracic Artery, TCT- Thyrocervical trunk, INF.THY.A- Inferior Thyroid Artery, ACA- Ascending Cervical Artery, TCA-Transverse Cervical Artery, SSA-Suprascapular Artery, FR-First Rib, SAM-Scalenus Anterior Muscle, RVN-Right Vagus Nerve, BT-Brachiocephalic Trunk, RCCA- Right Common Carotid Artery, AA-Arch of Aorta
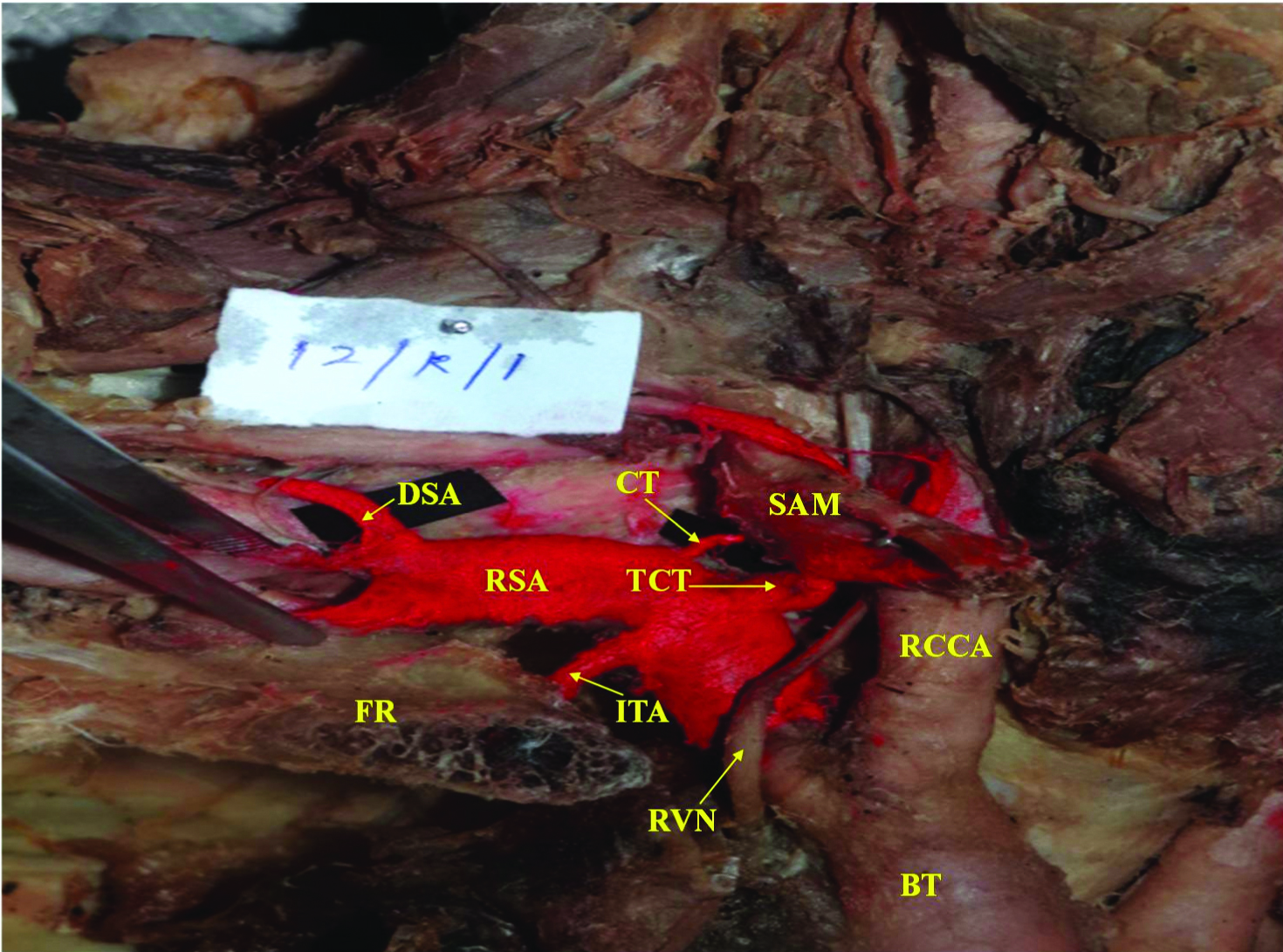
Branching Pattern Type I: Normal - Deep Branches: CT- Costocervical Trunk, DSA-Dorsal Scapular artery.
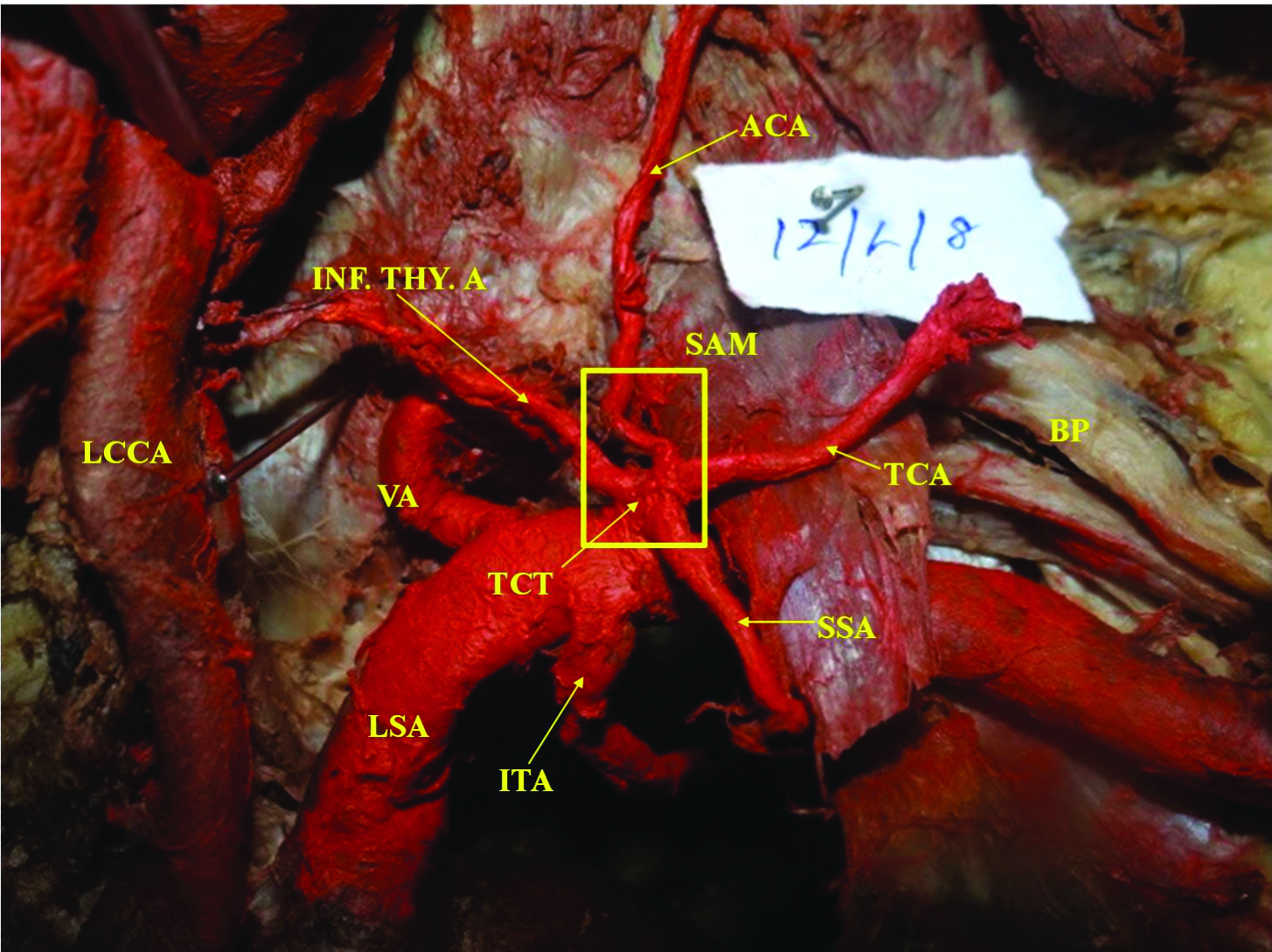
Branching Pattern Type II: ACA arises directly from TCT: BP- Brachial Plexus, LSA- Left Subclavian artery, LCCA- Left Common Carotid artery.
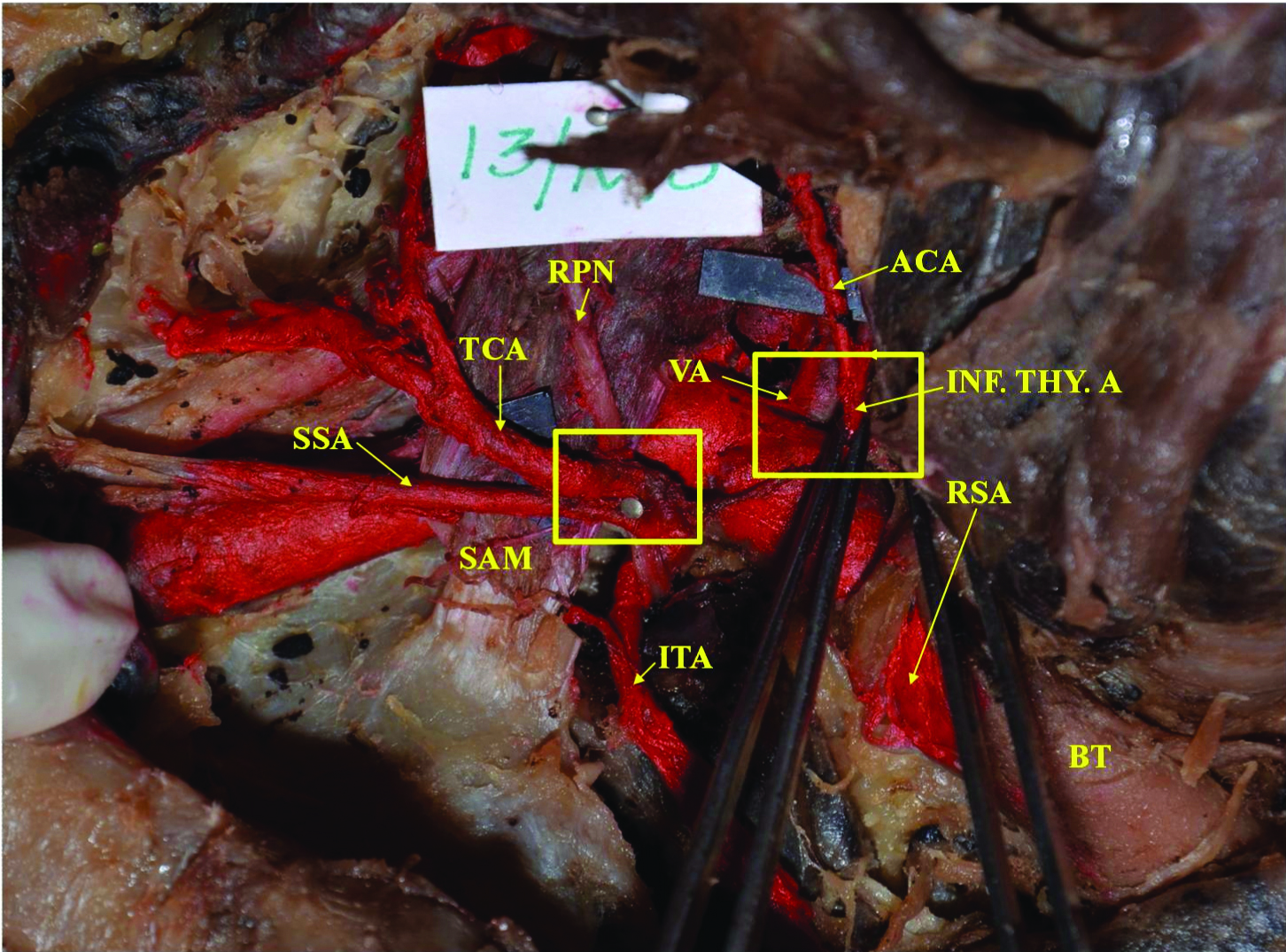
Branching Pattern Type III: Inf. Thy. A, TCA and SSA taking origin from the RSA in the absence of TCT: RPN-Right Phrenic Nerve.
Branching pattern of the Subclavian artery.
| Branching Pattern | No. of Arteries | Side | No. of Arteries | Sex | No. of Arteries |
|---|
| Type I | 20 | Left | 10 | Male | 12 |
| Right | 10 | Female | 8 |
| Type II | 11 | Left | 6 | Male | 3 |
| Right | 5 | Female | 8 |
| Type III | 1 | Left | 0 | Male | 1 |
| Right | 1 | Female | 0 |
Thirty-one VA (15 and 16 arteries on the left and right sides, respectively) originated from the first part of the Subclavian artery. The ITA proved to be one of the most constant branches arising from the first part (32 arteries) [Table/Fig-1].
Since the SA was classified based on the ramification of the TCT, more particularly, the ACA, their origin was keenly recorded. The TCT predominantly arose from the first part of the Subclavian (31 arteries) while its absence was found only once on the right side in a male [Table/Fig-4]. The Inf.Thy.A. was 100% incident among which 31 arteries arose from TCT and one artery arose directly from the Subclavian, in the absence of the TCT [Table/Fig-4]. The ACA arose from the Inf.Thy.A. in majority cases (21 arteries) and arose directly from the TCT in 11 cases [Table/Fig-3]. The TCA and SSA exhibited stereotypical origin from the TCT in 28 arteries and 29 arteries, respectively.
The CT arose from the second part of the SA in all cases; in 24 arteries, it did not exhibit immediate bifurcation into its branches at the point of origin. The DSA arose from the 3rd part in 21 cases.
Discussion
Earlier studies on the SA were carried out in different populations and involved a higher number of arteries in comparison to the present study [Table/Fig-6]. The sides, in the current study, showed the equal occurrence of the predominant branching pattern of SA (Type I) on either side in the present study unlike the patterns reported earlier [5,6]. The individual branches of SA, in addition to the SA branching pattern, were also classified and compared with earlier studies on different populations [5-7].
Branching pattern of SA – A Comparison [5-7].
| Authors | N (SA) | Population | Brief outcome |
|---|
| Bean RB, [5] | 129 | Negro and White | 5 Types of SA branching TCT branched into SSA, TCA and Inf.Thy.A. on the left. ‘Bunched’ origin of SA branches seen on the left; branches directly arose from SA with adequate spacing on the right
|
| De Garis CF [6] | 278 | Negro and White | Two normal patterns of SA in reference to the TCT or its components Comparison of the sides showed insignificant or infrequent occurrence of either of the patterns
|
| Takafuji T et al., [7] | 144 | Japanese | Type with common trunk formed by the ACA and the Inf.Thy.A containing branches other than ITA, appeared with the highest frequency (82.6%).
|
| Present study | 32 | Indian | Predominant type – 20 SA (62.5%) with TCT arising from the first part of SA, from which the Inf.Thy.A, TCA and SSA take origin; ACA branches from the Inf. Thy.A. Equal occurrence on each side
|
The normal three branched AA was reported in about 92.72% of hearts [8] and 63.5% AA [9] studied, suggesting conventional origin of Left SA from the AA. An amount of 59 out of 60 SA showed origin from the Brachiocephalic trunk and AA on the right and left sides, respectively [10]. The SA was found to have the normal pattern of origin and termination on both sides in the present study. This being the case with previous studies too [4-7], these parameters were not greatly emphasised. Extensive use of musculocutaneous flap grafts consisting of the upper and middle portions of the trapezius in plastic and orthopaedic surgery enhances the value of the study of the SA [11].
A total of 31 arteries recorded a normal course (posterior to the SAM) and one transfixed on the left side in a male cadaver similar to a previous record [8]. Studying the relation between the SA and the SAM [7] revealed that 142 SA traversed posterior to the muscle, one was transfixed and another traversed anterior to it. Consulting ontogeny, the authors [7] considered that the primordium of the SAM envelops the artery anteroposteriorly, thus, the transfixed should be the archetype; the disappearance of the posterior bundle of the muscle fibres gives rise to the normal course. Serizawa M reported a transfixed SA [12]. Hypertrophy of SAM [13] or Cervical rib may lead to Scalenus anticus syndrome which is resolved by partial or complete removal of the cervical rib or sectioning the SAM which would require exposing the Innominate artery or SA and their branches [4].
The branching of the SA had been worked upon [7] to understand its morphology as a prerequisite for diagnosis of pathogenic changes in the upper limb by advanced radiologic techniques. While extensive works [5-7] have documented even minor variations in the constituent branches of SA, the current study does not focus them, the emphasis being on the ramification. A 27% of the cases {22% (left) and 5% (right)} in Bean RB, study showed ramification similar to this study’s Type I (20 arteries (62.5%); 10 arteries each on the left and right; 8 female and 12 male) [Table/Fig-1] with the presence of a TCT, arising from the first part of Subclavian, from which the Inf.Thy.A, TCA and SSA take origin; ACA branches from the Inf.Thy.A [Table/Fig-1]. The VA and ITA arise from the first part and the CT arises from the second [5]. This is in accordance with the common description of SA [2], Type A [6] and Type I-A(1) [7] from older studies. Bunched origin (branches from the first part of SA arise closely) [5] of the SA branches was seen in most cases. Type II pattern of branching (11 SA) saw the ACA arising directly from the TCT like a variant spotted earlier [6]. The present Type III pattern features the TCT’s branches arising directly from the SA in the absence of a common trunk while the ACA arose from the Inf.Thy.A. This closely resembles SA Type I-F(4) [7] with less ‘bunching’ [5] in earlier accounts of SA.
The definition of TCT was clarified based on observations and development as a common trunk consisting only of ACA and the Inf.Thy.A [7]: All of the combinations of other arteries with this fundamental arterial trunk be termed the TCT, and forms in which ITA and ACA arise independently, or either one is absent be excluded from the definition of TCT [7]. The current study classified TCT considering this lead. The TCT pattern with that of previous studies has been compared [Table/Fig-7]. The supposedly typical pattern of branching of the TCT was found to occur in only 29% of 544 bodies in a British series [14]. The arrangement of TCT as seen in Type I SA in this study was stated as normal [2,5,15]. Besides recording an absence in over 17% of the cases, its constancy in location makes it a useful anatomic and surgical landmark [4]. The absence of the trunk featured the Inf.Thy.A. occupying its place at the origin on the Subclavian (similar to Type b 16.12% [4] and Type III SA in this study). This could be equated to Type a TCT in Japanese [7] [Table/Fig-7]. Inf.Thy.A. occurred as observed in Bean RB [5] SA (Types I and II (35% and 32% subjects, respectively) arising as a branch of the TCT and giving rise to ACA. This most surgically significant branch of the TCT [4] showed perfect constancy as its branch (80.25%) in Type A of a study [4] and in the current study (97%; 16 on the left, 15 right; 15 in male and 16 female). The ACA arose as a fairly constant small branch of the Inf.Thy.A in 72.33% (Type a) cases [4] in accordance with earlier calculations [5] similar to Type I SA in this study. ACA arose from the TCT (11 cases- Type II SA) [Table/Fig-3]; 10.68% (Type b) [4], 30 and 99 such cases per thousand have been recorded by earlier authors [5,14].
Comparison of the branching pattern of TCT with previous studies [4-7].
| Study | No. of specimen | Authors’ Type of TCT | Present Study: Type of TCT (No. of TCT) | Findings |
|---|
| Bean RB, [5] | 27% of 258 SA | Type II | Type I (20) | Normal |
| De Garis CF, [6] | 85/278 SA | Type A | Type I (20) | Normal |
| Daseler EH et al., [4] | 83/770 sides | Type d (10.78%) | Type I (20) | Normal |
| 131/770 sides | Type b (17%) | Type III (1) | Absence of common trunk |
| Takafuji T et al., [7] | 31.9% | Type I-c | Type I (20) | Normal |
| Modification of Type I-c | Type II (11) | ACA arises from TCT directly |
A comparison of the occurrence of the other constituent branches of the SA can be found in [Table/Fig-8] [4,5,6,7,14,16]. VA and ITA originated from the first part of SA in majority cases. Quain R, gave the most frequent origin of TCA from the TCT to divide into the posterior scapular and superficial cervical branches [15]. TCA and SSA show higher occurrence with its origin from the TCT in comparison to earlier studies [4,5,7]; this could be due to the less number of cases available in the present study. Nevertheless, SSA’s origin from TCT is found to be frequent in Asians [7], hence, may be expected to be so in Indians.
Comparison of the origin of other constituent branches of SA [4,5,6,7,14,16].
| S.No | Artery | Study | Type/No. of specimen | Population | Findings |
|---|
| 1. | VA | Daseler EH et al., [4] | 83.12% | - | Arises from first part of SA |
| Takafuji T et al., [7] | 94.5% | Japanese |
| Present study | 96.8% (31 cases) | Indians |
| 2. | ITA | Bean RB, [5] | 80% | Negroes and Whites | Arises from first part of SA |
| Daseler EH et al., [4] | 79.19% | - |
| Takafuji T et al., [7] | 87.5% | Japanese |
| Present study | 100% (32 cases) | Indians |
| 3. | CT | Bean RB, [5] | 90% | Negroes and Whites | Arises from first part of SA | Left |
| 9% | Arises from second part of SA | Right |
| 1% | Arises from third part of SA | Right |
| De Garis CF, [6] | 112 cases | Negroes and Whites | Arises from first part of SA |
| 119 cases | Arises from second part of SA |
| Takafuji T et al., [7] | 1-a: 22.9%;(33 cases) | Japanese | Arises from first part of SA | More on the left |
| 2-a: 42.4%;(61 cases) | Arises from second part of SA | More on the right |
| Present study | 100% (32 cases) | Indians | Arises from second part of SA |
| 4. | TCA | Bean RB, [5] | 39% | Negroes and Whites | With TCT or alone | Arises from second part of SA | More on the right |
| 36% | Arises from first part of SA |
| Thomson A [14] | - | English | From TCT |
| Rohlich K [16] | Europeans | From SA |
| Present study | 87.5% (28 cases) | Indians | From TCT, first part of SA |
| 5. | SSA | Bean RB, [5] | 34%(Type II SA) | Negroes and Whites | From TCT |
| Takafuji T et al., [7] | 38.2% (Type b) | Japanese; frequent in Asians | Frequent from TCT |
| Present study | 90.6% | Indians | From TCT |
| 6. | DSA | Bean RB, [5] | - | Negroes and whites | DSA present when SSA absent |
| Present study | 21 cases | Indians | Arises from third part of SA |
The origin of the CT, dividing almost immediately into its constituent branches [5], should be either from the first and second parts of the SA or from the junction of these two regions contiguous to the medial border of SAM opined an author [6]; this opinion that balances the contrast in reports of the earlier works [2,5] and the authors’ [6] findings also substantiate the current observation of CT arising from the second part of SA in all cases [Table/Fig-2,8].
DSA is generally not dealt too extensively as it seldom follows strict occurrence. According to popular opinion, DSA is present when the SCA, a branch of TCA, is absent. On the contrary, Bean RB was of the opinion that DSA is present when the SSA is absent [Table/Fig-8] [5].
Limitation
The study results cannot be attributed to the Indian or Southern Indian population as the sample size of 32 arteries is less to provide statistical implication. This warrants population-based studies of the Subclavian Artery (SA) with adequate sample size.
Conclusion
All the specimens of Subclavian artery recorded normal origin and termination. The branches of the Subclavian were predominantly constant retaining conventional pattern of branching. Aberrations in AA, aneurysms, stenosis, dissection of subclavian artery and its major branches are seen in clinical practice. Hence, studying the anatomy of subclavian artery along with surgical and developmental implications is of importance. Anatomists, radiologists, vascular surgeons, neurologists will benefit from the knowledge gained.
[1]. Mehrdad J, Sadeghi Y, A case report of variation in subclavian artery branchesJ Med Sci 2007 7(3):457-59.10.3923/jms.2007.457.459 [Google Scholar] [CrossRef]
[2]. Gray H, Gray’s Anatomy - The Classic First EditionIn: Subclavian Arteries 1858 John W. Parker & SonThe Promotional Reprint Company; 1991. Pp. 339-348 [Google Scholar]
[3]. Hollinshead WH, Anatomy for Surgeons: The Head and Neck in Arteries 1982 Vol.13rdPhiladelphiaHarper & Row Publisher:452-458. [Google Scholar]
[4]. Daseler EH, Anson BJ, Surgical anatomy of the subclavian artery and its branchesSurg Gynecol Obs 1959 108(2):149-74. [Google Scholar]
[5]. Bean RB, A composite study of the subclavian artery in manAm J Anat 1905 IV(3):303-28.10.1002/aja.1000040303 [Google Scholar] [CrossRef]
[6]. De Garis CF, Patterns of branching of the subclavian artery in the white and negro stocksAm J Phys Anthropol 1924 VII(1):95-107.10.1002/ajpa.1330070116 [Google Scholar] [CrossRef]
[7]. Takafuji T, Sato Y, Study on the subclavian artery and its branches in Japanese adultsOkajimas Folia Anat Jpn 1991 68(2–3):171-86.10.2535/ofaj1936.68.2-3_1711758682 [Google Scholar] [CrossRef] [PubMed]
[8]. Rekha P, Senthil Kumar S, A study on branching pattern of human aortic arch and its variations in South Indian populationJ Morphol Sci 2013 30(1):11-15.10.5402/2013/82896925938106 [Google Scholar] [CrossRef] [PubMed]
[9]. Budhiraja V, Rastogi R, Jain V, Bankwar V, Raghuwanshi S, Anatomical variations in the branching pattern of human aortic arch: a cadaveric study from central IndiaISRN Anat 2013 2013:82896910.5402/2013/82896925938106 [Google Scholar] [CrossRef] [PubMed]
[10]. Poonam Jyoti N, Singla RK, Sharma T, Anatomical considerations and clincial implications of subclavian arteryJ Evol Med Dent Sci 2013 2(29):5484-91.10.14260/jemds/1013 [Google Scholar] [CrossRef]
[11]. Demergasso F, Piazza MV, Trapezius myocutaneous flap in reconstructive surgey for head and neck cancer: An original techniqueAm J Surg 1979 138:533-43.10.1016/0002-9610(79)90414-8 [Google Scholar] [CrossRef]
[12]. Serizawa M, On the m. scalenus anterior in adult JapaneseJ Nippon Med Sch 1968 35:188-202.10.1272/jnms1923.35.188 [Google Scholar] [CrossRef]
[13]. Swank Roy L, Simeone FA, The scalenus anticus syndromeArch Neurol Psychiatry 1944 51(5):432-45.10.1001/archneurpsyc.1944.02290290019004 [Google Scholar] [CrossRef]
[14]. Thomson A, Second Annual report of the Committee of Collective Investigation of the Anatomical Society of Great Britain and Ireland for the year 1890-91J Anat Physiol 1891 26:76-93. [Google Scholar]
[15]. Quain R, The Anatomy of the Arteries of the Human Body 1844 LondonTaylor and Walton:153 [Google Scholar]
[16]. Röhlich K, Über den Truncus thyreocervicalis des MenschenAnat Anz 1941 90:129-48. [Google Scholar]