Introduction
Early Childhood Caries (ECC) affects a significant proportion of the child population worldwide. Salivary proteins contain a host defense system that plays a role in the caries pathogenesis.
Aim
To analyse the protein profile and salivary viscosity from ECC and caries-free subjects.
Materials and Methods
The number of subjects included in the study was 16 ECC and 16 caries-free. Salivary viscosity was assessed visually and salivary protein profile was analysed using SDS PAGE.
Results
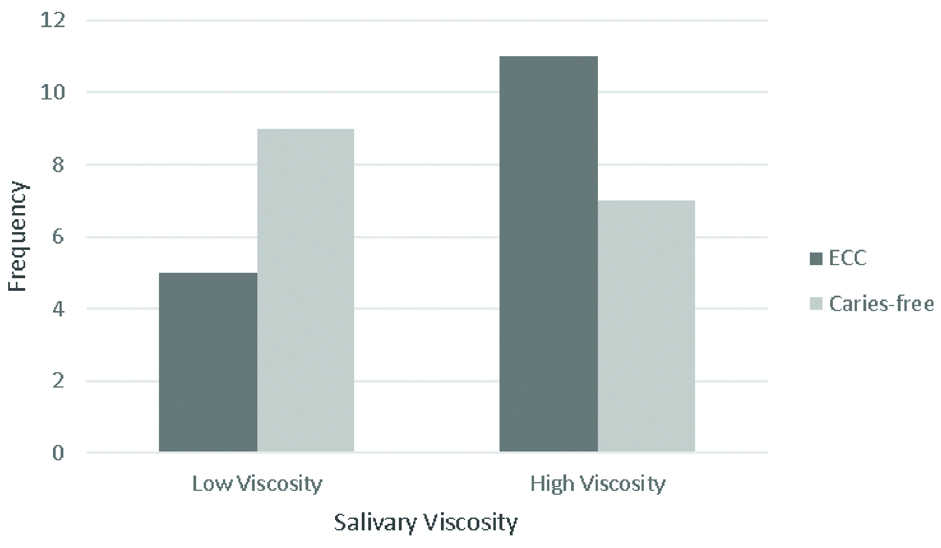
The five most prominent protein bands with molecular mass 15 kDa, 25 kDa, 60 kDa, 65 kDa and 95 kDa were found in ECC patients with the frequency of occurrences in order 8, 3, 16, 6, 2 and caries-free subjects 14, 5, 16, 16, 9. The saliva viscosity of ECC patient was higher than the caries-free subjects.
Conclusion
In ECC patients, the frequency of occurrences of salivary protein profile 15 kDa, 25 kDa, 65 kDa and 95 kDa were found less than in caries-free subjects. Meanwhile, protein profile 60 kDa has the same frequency of occurrence in ECC and caries-free subjects. Viscosity of ECC patient was higher than the caries-free subject.
Albumin, Amylase, Lactoferrin, Salivary Proline-Rich Proteins (PRPs), sIgA
Introduction
Early childhood caries is a condition in which there is presence of one or more decayed tooth with non-cavitated or cavitated lesions, missing due to caries, or filled tooth surfaces in any primary tooth of a child under the age of six years based on American Association of Paediatric Dentistry (AAPD) [1].
In most developed countries the prevalence rate of ECC is between 1 and 12% [2]. In less developed countries and among the disadvantaged groups in the developed countries, the prevalence has been reported to be as high as 70%. ECC has been found to be more prevalent in low socioeconomic groups [3,4]. Prevalence of ECC in DKI Jakarta and its surrounding area in 1988 was 85.17% and in 2001 was 81.2% [5]. As a result of the development of caries, it can cause pain and affect the ability to chew and eat, which can also cause iron deficiency due to malnutrition [6].
Dental caries is a chronic disease with complex and multifactorial causes resulting from an interaction between pathogenic microorganisms, carbohydrate fermentation substrate, host conditions and at a certain period [7]. One of the host factors in ECC is saliva which plays a crucial role in maintaining oral health and inhibiting the growth of pathogenic microorganisms.
Saliva has a physiochemical characteristic, one of which is viscosity-that affects performing one of saliva function which is self-cleansing that can reduce the risk of ECC [8]. There are specific defense proteins such as immunoglobulins, as well as many other proteins that work nonspecifically such as Proline Rich-Proteins (PRPs), mucin, agglutinin, lactoferrin, cystatin, lysozyme, etc., [9,10]. Protein helps saliva function in protecting and maintaining the oral health.
Differences in metaproteomic profiles of saliva and the salivary viscosity influence caries formation. This study aims to analyse the salivary protein profile by SDS-PAGE and salivary viscosity in children with ECC.
Materials and Methods
The sample size was determined by binomial proportions as this study was a cross-sectional. Total 32 saliva samples from children aged 3-5 years consisting of 16 ECC patients and 16 caries-free subjects were included. Samples were taken in Al-Multazam Kindergarten, Depok-West Java, with ethical approval from the Ethical Research Committee Faculty of Dentistry University of Indonesia (No:24/Ethical Approval/FKGUI/V/2017).
Sampling and Sample Preparation
Unstimulated saliva was taken using the sterile pipette and accommodated in the 1.5 mL microcentrifuge tube containing Phosphate Buffer Saline (PBS) and Phenylmethanesulfonylfluoride (PMSF) solution. Saliva samples were homogenised and then centrifuged for 10 minutes at 4°C with 12000 rpm. The salivary supernatant was taken and transferred to a new microcentrifuge tube. The concentration of the protein was measured using Bradford methods. Standard protein and saliva were added to 96 well plate as much as 10 μL. Each of the well containing standard protein and saliva was added 190 μL. Bradford reagents and then read by microplate reader with optical density 595 nm.
SDS Page
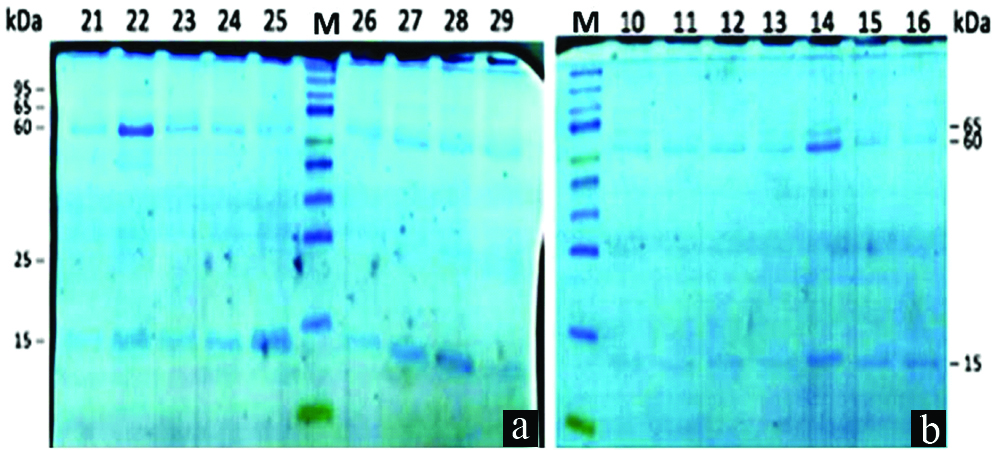
Saliva concentration was synchronised to 100 μg/mL based on Bradford for the SDS PAGE methods. Ten μL buffer sample was added to the synchronised saliva sample and then put on the thermoblock at 95°C for 5 minutes. Resolving gel and stacking gel were made until wells were formed. Then, 15 μL of sample and 5 μL of protein markers (PeqGold VI Prestained) were put in each well of stacking gel for electrophoresis procedure. Electrophoresis tank was connected with the power source with 150 V and 80 mA for 70 minutes [11].
After the electrophoresis procedure was done, the gel was taken and put in the container containing Page Blue as a staining solution. The container was put on the shaker at 60 rpm for overnight. Then, the gel was destained using aquades every 30 minutes. The result of SDS PAGE was read by the scanner, and each of protein bands was noted in table and charts.
Results
Protein Profile
Five most prominent protein band were found that are 15 kDa, 25 Da, 60 kDa, 65 kDa and 95 kDa.
As shown in [Table/Fig-1,2, and 3], there are differences in protein profile occurrences in ECC patients and caries-free subjects. Protein with molecular weight 15 kDa, 25 kDa, 65 kDa and 95 kDa were found more frequently in the caries-free subject compared to ECC patients.
Salivary protein profile in ECC patient and caries-free subjects.
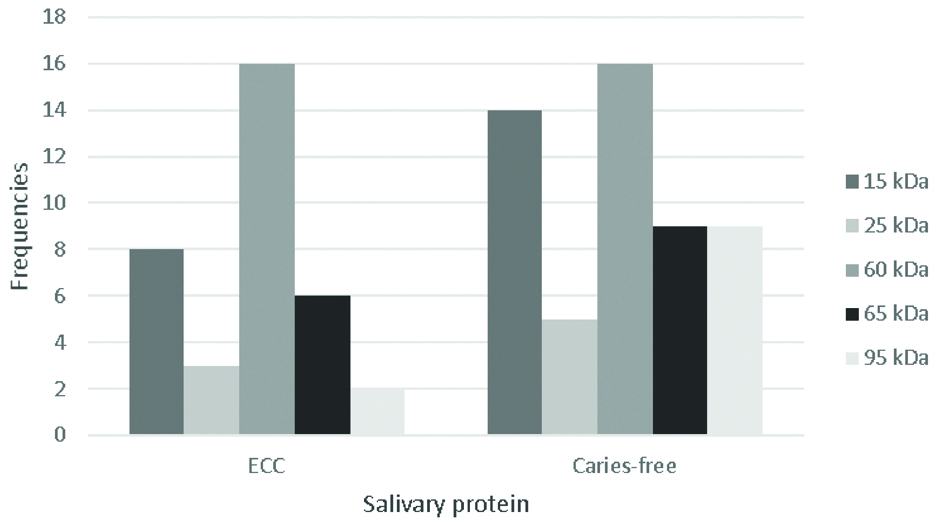
The summary of the protein bands expression from the saliva of caries-free and ECC subjects.
| 15 kDa | 25 kDa | 60 kDa | 65 kDa | 95 kDa |
|---|
| ECC | 8 | 3 | 16 | 6 | 2 |
| Caries-Free | 14 | 5 | 16 | 9 | 9 |
The representative data of SDS-PAGE protein bands of saliva from a) caries-free subject and b) ECC.
M: Marker
[Table/Fig-4] shows that on low viscosity, the frequency of ECC patients was lower (5) than the caries-free subjects (9). While on high viscosity, the frequency of ECC patients was higher (11) than the caries-free subjects (7).
The saliva’s viscosity in ECC patient and caries-free subject.
Discussion
The 15 kDa molecular weight protein was suspected as cystatin [12,13]. Cystatin is a natural inhibitor of cysteine proteinase that also acts as antiviral and antibacterial by assisting in remineralisation [14,15]. Cystatin can be found in pellicle, which allows the penetration of minerals into the enamel to increase the enamel crystalline growth [16]. In this study, the 15 kDa protein was found more on the caries-free subjects than ECC patients which is in accordance to the previous studies [17,18].
Salivary PRPs are classified as acidic, basic or glycosylated [14,15,19]. In this study, the protein with molecular weight of 25 kDa was suspected as basic PRPs (bPRPs) [12,18] and has the frequency of occurrence higher on caries-free subjects than ECC patients. However, the differences are not significant. The bPRPs are derived from parotid glands and can be found in oral epithelial cells [20]. The function of these bPRPs is not well known [14,15] but allegedly binding polyphenols which are acidic and toxic, thus preventing the spread of toxic effects in the oral cavity. This protein also has antimicrobial properties through microbial aggregates and prevent binding to the surface of the oral cavity [13,14,21]. The previous study found that there was a significant difference between bPRPs on ECC and caries-free group, which was higher on the caries-free group. Although it is said that the function of bPRPs is not known clearly, but based on the evidences from various studies indicates that bPRPs contribute in protecting the oral cavity from caries [15,21-23].
Protein with molecular weight 60 kDa was found 100% on ECC patient and caries-free subject. This protein was suspected as α-amylase which is the most commonly secreted protein and salivary enzyme, primarily through the parotid gland (80%). Amylase works by lubricating the food and turns them into bolus and breakdown the starch into maltose and oligosaccharides. Amylase can act as an antibacterial agent but its ability is limited [14,24,25]. Amylase does not bind to epithelial cells and exhibits very minimal activity and can be found in pellicle at low level [22,23,26]. Based on this theory, amylase activity does not affect the occurrence of caries. This finding is supported by the previous study that there were no significant differences in salivary amylase expression between caries groups and caries-free group [22,27-29].
The protein with 65 kDa molecular weight was determined as serum albumin [12,13], that acts as a protein carrier and plays a role in salivary buffer system [15,29-32]. Based on this study which supported by previous studies, it appears that the serum albumin protein contributes in preventing the occurrence of caries as it was found to be more abundant in the caries-free subjects compared to ECC patients.
Protein with molecular weight 95 kDa was found more frequently in the caries-free subjects than ECC patients. Protein with molecular weight 80 kDa, 83 kDa, and 97 kDa are reported as the secretory component, which are the transporters of IgA in epithelial cells [12,20,30,31,32]. With this same range, 95 kDa protein can be estimated as the secretory component in saliva. Secretory IgA (s-IgA) is the primary immunoglobulin of saliva that serves as the first-line defense of the oral cavity that acts through inhibition of glucosyltransferase activity (Gtf), prevents bacterial adhesion, inactivates enzymes and bacterial toxins and acts synergistically with other salivary components [33-36]. The s-IgA has a relationship with dental caries even though there are different opinions about it. According to Chawda JG et al., s-IgA was found more significant in the caries-free subjects by providing localised immune protection against cariogenic microorganisms [34]. The result of this study found that the frequency of occurrence of 95 kDa protein was higher in the caries-free subjects compared to ECC patients. However, Ranadheer E, et al., mentioned that there was an increase of s-IgA level in the subjects with active caries [32]. This is supported by the previous study that s-IgA is more prevalent in caries subject providing an indication of an immune response to protect the oral cavity against pathogenic microorganism [35].
In addition to the protein profile of saliva, we also assessed the viscosity of saliva. Viscosity defined as a condensed salivary state, which is the rheological property of a complex salivary fluid [33,36]. In this study, the ECC patients had a high viscosity which is thick and bubbly while the caries-free subjects had a low viscosity of clear and watery saliva. Increase in salivary viscosity might due to the decrease of water content in saliva [34] and may increase the risk of caries [14,16,36].
Limitation
There is some limitation of this study that needs further investigation such as determination of the proteins by Immunoblotting. The results of protein function analysis may have a benefit in future caries prevention.
Conclusion
The frequency of occurrences of the 15 kDa, 25 kDa, 65 kDa and 95 kDa protein profiles in the ECC patients was found lesser than in caries-free subjects. The 60 kDa protein profile has the same frequency in ECC patients and caries-free subjects. There is also a difference in the salivary viscosity which is high in ECC patients and low in caries-free subjects.
[1]. American Academy, Policy on Early Childhood Caries (ECC): classification, consequences, and preventive strategiesOral Heal Policies 2014 37(6):50-52. [Google Scholar]
[2]. Congiu G, Campus G, Luglie PF, Early childhood caries (ECC) prevalence and background factors: a reviewOral Health Prev Dent 2014 12(1):71-76. [Google Scholar]
[3]. Ismail AI, Lim S, Sohn W, Willem JM, Determinants of early childhood caries in low-income African American young childrenPediatr Dent 2008 30(4):289-96. [Google Scholar]
[4]. Vachirarojpisan T, Shinada K, Kawaguchi Y, Laungwechakan P, Somkote T, Detsomboonrat P, Early childhood caries in children aged 6-19 monthsCommunity Dent Oral Epidemiol 2004 32(2):133-42.10.1111/j.0301-5661.2004.00145.x15061862 [Google Scholar] [CrossRef] [PubMed]
[5]. Sugito FS, Djoharnas H, Darwita RR, Breastfeeding and early childhood caries (ecc) severity of children under three-years-old in Dki JakartaMakara Seri Kesehatan 2008 12(2):86-91.10.7454/msk.v12i2.310 [Google Scholar] [CrossRef]
[6]. Fung MH, Wong MC, Lo EC, Chu C, Early childhood caries: a literature reviewJournal of Oral Hygiene & Health 2013 1(1):1-7. [Google Scholar]
[7]. Moslemi M, Sattari M, Kooshki F, Fotuhi F, Modarresi N, Khalili Sadrabad Z, Relationship of salivary lactoferrin and lysozyme concentrations with early childhood cariesJ Dent Res Dent Clin Dent Prospects 2015 9(2):109-14.10.15171/joddd.2015.02226236438 [Google Scholar] [CrossRef] [PubMed]
[8]. Chiappin S, Antonelli G, Gatti R, De Palo EF, Saliva specimen: A new laboratory tool for diagnostic and basic investigationClin Chim Acta 2007 383(1-2):30-40.10.1016/j.cca.2007.04.01117512510 [Google Scholar] [CrossRef] [PubMed]
[9]. Hajishengallis E, Parsaei Y, Klein MI, Koo H, Advances in the microbial etiology and pathogenesis of early childhood cariesMol Oral Microbiol 2017 32(1):24-34.10.1111/omi.1215226714612 [Google Scholar] [CrossRef] [PubMed]
[10]. Huang CM, Comparative proteomic analysis of whole human salivaArch Oral Biol 2004 49(12):951-62.10.1016/j.archoralbio.2004.06.00315485636 [Google Scholar] [CrossRef] [PubMed]
[11]. Bachtiar E, Coloe P, Smooker P, Construction and immunogenicity of Salmonella vaccine vector expressing HIV-1 antigen and MCP3Acta Microbiologica et Immunologica Hungarica 2009 56(4):403-15.10.1556/AMicr.56.2009.4.1020038492 [Google Scholar] [CrossRef] [PubMed]
[12]. Chevalier F, Hirtz C, Chay S, Cuisinier F, Sommerer N, Rossignol M, Proteomic Studies of Saliva: A Proposal for a Standardized Handling of Clinical SamplesClin Proteomics 2007 3(1-4):13-21.10.1007/s12014-007-9000-x [Google Scholar] [CrossRef]
[13]. Huq NL, Cross KJ, Ung M, Myroforidis H, Veith PD, Chen D, A Review of the Salivary Proteome and Peptidome and Saliva-derived Peptide TherapeuticsInt J Pept Res Ther 2007 13(4):547-64.10.1007/s10989-007-9109-9 [Google Scholar] [CrossRef]
[14]. Amado FML, Vitorino RMP, Domingues PMDN, Lobo MJC, Duarte JAR, Analysis of the Human Saliva ProteomeExpert Rev Proteomics 2005 2(4):521-39.10.1586/14789450.2.4.52116097886 [Google Scholar] [CrossRef] [PubMed]
[15]. Humphrey SP, Williamson RT, A review of saliva: Normal composition, flow, and functionJ Prosthet Dent 2001 85(2):162-69.10.1067/mpr.2001.11377811208206 [Google Scholar] [CrossRef] [PubMed]
[16]. Rudney JD, Staikov RK, Johnson JD, Potential biomarkers of human salivary function: A modified proteomic approachArch Oral Biol 2009 54(1):91-100.10.1016/j.archoralbio.2008.08.00718804197 [Google Scholar] [CrossRef] [PubMed]
[17]. Si Y, Ao S, Wang W, Chen F, Zheng S, Magnetic bead-based salivary peptidome profiling analysis for severe early childhood cariesCaries Res 2015 49(1):63-69.10.1159/00036086825547533 [Google Scholar] [CrossRef] [PubMed]
[18]. Dodds MWJ, Johnson DA, Yeh CK, Health benefits of saliva: a reviewJ Dent 2005 33(3 SPEC. ISS.):223-33.10.1016/j.jdent.2004.10.00915725522 [Google Scholar] [CrossRef] [PubMed]
[19]. Gibbins HL, Proctor GB, Yakubov GE, Wilson S, Carpenter GH, Concentration of salivary protective proteins within the bound oral mucosal pellicleOral Dis 2014 20(7):707-13.10.1111/odi.1219424205881 [Google Scholar] [CrossRef] [PubMed]
[20]. Levine M, Susceptibility to dental caries and the salivary proline-rich proteinsInt J Dent 2011 2011:95341210.1155/2011/95341222190937 [Google Scholar] [CrossRef] [PubMed]
[21]. Bhalla S, Tandon S, Satyamoorthy K, Salivary proteins and early childhood caries: A gel electrophoretic analysisContemp Clin Dent 2010 1(1):17-22.10.4103/0976-237X.6251522114372 [Google Scholar] [CrossRef] [PubMed]
[22]. Ayad M, Van Wuyckhuyse BC, Minaguchi K, Raubertas RF, Bedi GS, Billings RJ, The Association of Basic Proline-rich Peptides from Human Parotid Gland Secretions with Caries ExperienceJ Dent Res 2000 79(4):976-82.10.1177/0022034500079004140110831101 [Google Scholar] [CrossRef] [PubMed]
[23]. Dawes C, Pedersen AML, Villa A, Ekström J, Proctor GB, Vissink A, The functions of human saliva: A review sponsored by the World Workshop on Oral Medicine VIArch Oral Biol 2015 60(6):863-74.10.1016/j.archoralbio.2015.03.00425841068 [Google Scholar] [CrossRef] [PubMed]
[24]. Fejerskov O, Kidd E, Nyvad B, Baelum V, Dental Caries. 2nd ed 2008 Blackwell Munksgaard Ltd:201 [Google Scholar]
[25]. Gibbins HL, Yakubov GE, Proctor GB, Wilson S, Carpenter GH, What interactions drive the salivary mucosal pellicle formation?Colloids Surfaces B Biointerfaces 2014 120:184-92.Available from: http://dx.doi.org/10.1016/j.colsurfb.2014.05.02010.1016/j.colsurfb.2014.05.02024921197 [Google Scholar] [CrossRef] [PubMed]
[26]. de Farias DG, Bezerra ACB, Salivary antibodies, amylase, and protein from children with early childhood cariesClin Oral Investig 2003 7:154-57.10.1007/s00784-003-0222-712915963 [Google Scholar] [CrossRef] [PubMed]
[27]. de Almeida PDV, Grégio AMT, Machado MAN, de Lima AAS, Azevado LR, Saliva composition and functions : a comprehensive reviewJ Contemp Dent Pract 2008 9(3):72-80.10.5005/jcdp-9-3-72 [Google Scholar] [CrossRef]
[28]. Pfaffe T, Cooper-White J, Beyerlein P, Kostner K, Punyadeera C, Diagnostic Potential of Saliva: Current State and Future ApplicationsClin Chem 2011 57(5):675-87.10.1373/clinchem.2010.15376721383043 [Google Scholar] [CrossRef] [PubMed]
[29]. Carpenter GH, The secretion, components, and properties of salivaAnnu Rev Food Sci Technol 2013 4(1):267-76.10.1146/annurev-food-030212-18270023464573 [Google Scholar] [CrossRef] [PubMed]
[30]. Morzel M, Palicki O, Chabanet C, Lucchi G, Ducoroy P, Chambon C, Saliva electrophoretic protein profiles in infants: Changes with age and impact of teeth eruption and diet transitionArch Oral Biol 2011 56(7):634-42.10.1016/j.archoralbio.2010.12.01521429473 [Google Scholar] [CrossRef] [PubMed]
[31]. Hashizume LN, Schwertner C, Moreira MJS, Coitinho AS, Faccini LS, Salivary secretory IgA concentration and dental caries in children with Down syndromeSpec Care Dent 2017 37(3):115-19.10.1111/scd.1222228263433 [Google Scholar] [CrossRef] [PubMed]
[32]. Ranadheer E, Reddy Nv, Nayak U, Rao VaP, The relationship between salivary IgA levels and dental caries in childrenJ Indian Soc Pedod Prev Dent 2011 29(2):10610.4103/0970-4388.8468121911947 [Google Scholar] [CrossRef] [PubMed]
[33]. Fidalgo TKDS, Freitas-Fernandes LB, Ammari M, Mattos CT, De Souza IPR, Maia LC, The relationship between unspecific s-IgA and dental caries: A systematic review and meta-analysisJ Dent 2014 42(11):1372-81.10.1016/j.jdent.2014.07.01125042566 [Google Scholar] [CrossRef] [PubMed]
[34]. Chawda JG, Chaduvula N, Patel HR, Jain SS, Lala AK, Salivary SIgA and dental caries activityIndian Pediatr 2011 48(9):719-21.10.1007/s13312-011-0113-y21992904 [Google Scholar] [CrossRef] [PubMed]
[35]. Ahmed Yas B, Jafer N, Salivary viscosity in relation to oral health status among a group of 20-22-year-old dental studentsIraqi J Comm Med 2013 (3):219-24. [Google Scholar]
[36]. Walsh LJ, Clinical aspects of salivary biology for the dental clinicianInt Dent South Africa (Australasian Ed 2007 2(3):16-30. [Google Scholar]