Breast cancer surgeries are often associated with moderate to severe intensity acute postoperative pain, in the first week, which is the chief culprit in culminating into chronic postsurgical pain [1-4].
Thoracic Paravertebral Block, an excellent analgesic modality for intra- and postoperative pain management, is used in thoracotomy, gastrectomy, open cholecystectomy operations with negligible opioid and Nonsteroidal Anti-Inflammatory Drugs (NSAIDS) like side effects [5-9]. Though general anaesthesia is gold standard for breast cancer surgeries, TPVB has emerged as an adjunct for analgesic modality for these patients.
In TPVB-bupivacaine, lignocaine, levobupivacaine, ropivacaine; any one or combination of these drugs have been used [10]. Many adjuvants have been tried along with the above mentioned local anaesthetic agents; like clonidine, epinephrine, dexamethasone, opioids, dexmedetomidine etc., [11-13].
Ropivacaine, an amino-amide local anaesthetic, is less cardiac and central nervous system toxic than other long acting local anaesthetics like bupivacaine [14]. Magnesium, a plentiful cation of human body and N-methyl D-aspartate (NMDA) receptor antagonist, is necessary for the presynaptic release of acetylcholine and mimics calcium-entry-blocking drugs [15,16].
Investigators have demonstrated that magnesium administration during GA reduces anaesthetic requirement and post operative analgesic consumption [17]. Magnesium has long been used for its analgesic, antihypertensive and anaesthetic sparing effects [18-21]. Despite its known benefits for pain control, magnesium has never been studied extensively for its effects as an adjuvant to anaesthetics during thoracic paravertebral block (TPVB).
In our study, we had added 1 mL of magnesium sulphate (50%) to local anaesthetic solution ropivacaine in the test group to compare the analgesia with placebo group (ropivacaine with normal saline).
The primary objective of this study was to compare the time of rescue analgesic administration with the placebo group. Secondarily, total dose of rescue analgesics, intraoperative fentanyl, propofol requirement, VAS score and side effects among two groups were compared.
Materials and Methods
After planning for this randomised controlled study, we received approval from Institutional Ethics Committee. The study was conducted in an operation theatre of a tertiary care centre (Government Medical College) from December 2014 to December 2015. Firstly all patients including other surgical patients had to undergo routine preanaesthetic check-up. Among them, female patients who were planned for unilateral breast cancer surgery under general anaesthesia in the age group 40-65 years and met the eligibility criteria (ASA physical status I and II, Mallampati grade I and II) were counselled regarding the study and those who gave the consent were included in the study.
Exclusion criteria were patients with known allergic to ropivacaine, magnesium sulphate; hepatorenal or cardiopulmonary abnormalities, alcohol addict or diabetes, neuropsychiatric or neuromuscular disorders, thrombocytopenia, coagulation or seizure disorders, anatomical anomalies of thoracic spine.
The patients were randomised using computer generated random number list and group allocation was done using sealed envelopes. These opaque envelopes had a paper slip inside them indicating either RM or RP group. On the outside patient’s registration number was mentioned. When the patient was received in the OT complex, the numbered envelope was handed over to a resident anaesthesiologist who was not taking part in the study. He opened the envelope and prepared the drug mixture to be administered in the TPVB route. Patients in group RP received 19 mL of 0.5% ropivacaine+1 mL normal saline for TPVB. Group RM received 19 mL 0.5% ropivacaine+500 mg (1 mL 50% w/v magnesium sulphate) magnesium sulphate for the same block.
In group RP, 19 mL Ropivacaine (0.5%) {Ropin® 0.5%, NEON Laboratories Ltd., Mumbai, India} and 1 mL normal saline mixture was prepared. In group RM, 19 mL Ropivacaine (0.5%) and 1 mL magnesium sulphate {Magneon® 50% w/v, NEON Laboratories Ltd., Thane, Maharashtra, India} mixture was prepared.
In all cases, preoperative fasting of minimum six hours was ensured before operation. All patients were given tablet lorazepam 1 mg and pantoprazole 40 mg orally at the night before surgery as premedication.
All baseline parameters like heart rate, Systolic BP (SBP), Diastolic BP (DBP), Mean Arterial BP (MAP), End tidal CO2 (EtCO2) and Oxygen saturation (SpO2) were noted in the observation room. Then all patients were transferred to operation theatre and continuously monitored using multipara monitor. In sitting position, same technique was used for paravertebral block in every patient. 2% lignocaine was used for skin infiltration at 2.5 cm lateral to the spinous process of third, fourth and fifth thoracic vertebra. Through the skin wheal, Tuohy needle (17G) was inserted till it comes in contact with transverse process of the intended thoracic vertebras. Tuohy needle was then walked off the cephalad edge of the transverse process and it was further advanced till it enters in paravertebral space which was identified by loss of resistance technique by using air. Local anaesthetic mixture which was already prepared by resident doctor (drawn in identical looking syringes) was injected in the desired position. The anaesthesiologist performing the paravertebral block was completely unaware of the group allocation or composition of the local anaesthetic mixture. Data collection was done by another resident doctor. The mixture was injected in three spaces in a divided manner in small aliquots with repeated aspiration. This local anaesthetic mixture administration was followed by general anaesthesia administration in supine position.
Normal Saline (NS) was started via 18 G i.v cannula done prior to transport of patient to OT. Pre-oxygenation was done with 100% O2 for five minutes. Premedication was given with inj. Glycopyrrolate (0.2 mg), inj. fentanyl (100 μg), inj ondansetron (8 mg) three minute before induction. Propofol (2 mg/kg) was used as inducing agent. Then laryngoscopy and intubation was done with the help of atracurium (0.5 mg/kg). After three minutes of atracurium application, less than 20 seconds were taken for laryngoscopy, intubation, and cuff inflation in all cases. Intraoperative muscle relaxation was maintained with intermittent intravenous atracurium (0.2 mg/kg) as per requirement. Anaesthesia workstation was used for controlled ventilation which was maintained with 66% N2O in O2 and isoflurane up to 1.5 MAC (under guidance of BIS monitoring). On completion of modified radical mastectomy, inj. glycopyrrolate 0.01 mg/kg and inj. neostigmine 0.05 mg/kg was used for reversal of anaesthesia and extubation was done with adequate spontaneous ventilation with Train-of-Four (TOF) ratio>0.9 and BIS≥70. Ten minutes after extubation, patients were all transferred to Post Anaesthesia Care Unit (PACU) for observation and postoperative pain management.
Insufficient analgesia was reflected by the presence of hypertension or tachycardia (>20% of baseline) during anaesthesia, {while BIS was 40-60 (i.e., within desired range)} and fentanyl 1 μg/kg was used to treat the condition. Propofol was supplemented with 0.2 mg/kg bolus for maximum three successive boluses at an interval of three to five minutes. This bolus propofol was given when BIS value exceeds 70. Target BIS was 40-60.
Hospital discharge (eye opening-discharge from hospital), the time for PACU stay (means time of arrival in PACU to discharge from PACU) and also the incidence of adverse events were noted. Modified Aldrete score is based on Consciousness, Respiration (breathing), Oxygen saturation, Circulation (BP), Activity. Each parameter has score (0-2). The highest cumulative score is 10. Patients were considered ready for discharge from the PACU when the modified Aldrete post anaesthesia score was ≥9. After being discharged from PACU patients were transferred to the ward. Ondansetron 0.1 mg/kg IV was administered for nausea and vomiting. Same surgeon operated all the cases. Vital parameters recording were done every 10 minutes for first two hour, then every half hourly for 12 hour, then every four hourly for two days.
After operation, efficacy of the paravertebral block was the primary outcome of the study which was measured by time of rescue analgesic administration among both the groups. The time of administration of first dose of rescue analgesic (Injection paracetamol 1000 mg i.v) following performance of paravertebral block was taken as the duration of block. Visual analog scale (VAS; 0=“no pain” and 10=“worst possible pain”) was used to assess the magnitude of postoperative pain for 48 hours. Injection paracetamol was given as rescue analgesia if the pain VAS >3. Perioperative adverse reactions and haemodynamic changes were also observed. Sedation was drowsy (not awake) and non-communicative state (spontaneously, but responded when asked for). Bradycardia was fall in Heart Rate (HR) ≥20% of baseline HR or HR <60/min or whichever is greater. When HR was <50/min, 0.6 mg i.v atropine was given. Hypotension was fall in Systolic Blood Pressure (SBP) ≥20% of baseline SBP or SBP<100 mm Hg or whichever is greater. When SBP <90 mm of Hg, 5 mg i.v mephentermine was given.
Statistical Analysis
Sample size was based on a crossover pilot study of 10 patients and was selected to detect a projected difference of 10% time (i.e., 1.2 hour) for administration of rescue analgesic among two groups for a type 1 error of 0.05 and a power of 0.8. On the basis of our previous study assuming within group SD of six hour and we needed to study at least 36 patients per group to be able to reject the null hypothesis which will be increased to 40 patients for possible dropouts. Raw data were entered into a MS Excel spreadsheet and analysed using standard statistical software SPSS® statistical package version 18.0 (SPSS Inc., Chicago, IL, USA). Categorical variables were analysed using the Pearson’s chi-square test. Normally distributed continuous variables were analysed using the independent sample t-test and p-value <0.05 was considered statistically significant.
Results
Each breast cancer surgery group in our study consisted of 40 patients which was greater than the calculated sample size. As the dropout cases were nil, 40 patients were in the control group (RP) and 40 patients in the magnesium group (RM) were considered for effectiveness analysis.
From [Table/Fig-1] it is evident that age, body weight, preoperative haemoglobin level, operative and anaesthetic duration were all found to be comparable (p>0.05). PACU discharge time was comparable among two groups [Table/Fig-1]. Baseline heart rate and Mean arterial pressure were also quite comparable among two groups [Table/Fig-1]. [Table/Fig-2] shows types of different breast surgeries and they were comparable too. Intraoperative mean fentanyl and propofol requirement were compared among two groups and they were much less in amount and statistically significant (p<0.05) in group RM than RP [Table/Fig-3].
Demographic profile and the preoperative hematologic status in both groups.
| Demographic factors | Group RP (n=40) | Group RM (n=40) | p-value | 95% confidence interval |
|---|
| Age (years) | 48.11±5.60 | 50.55±6.30 | 0.0710 | -5.0933 to 0.2133 |
| Weight (kg) | 60.54±9.33 | 56.92±10.02 | 0.3552 | -0.6897 to 7.9297 |
| Haemoglobin (gm%) | 11.82±3.6 | 12.55±3.91 | 0.3877 | -2.4030 to 0.9430 |
| Duration of Surgery | 55.93±8.03 | 59.11±11.13 | 0.1468 | -7.5002 to 1.1402 |
| Duration of Anaesthesia | 70.23±11.21 | 73.11±10.93 | 0.2482 | -7.8084 to 2.0484 |
| Time to reach Aldrete score ≥9 (minute) | 55.02±8.94 | 58.92±9.10 | 0.0568 | -7.9156 to 0.1156 |
| Baseline Heart rate | 68.55±12.43 | 63.72±11.93 | 0.0801 | -0.5933 to 10.2533 |
| Baseline MAP | 79.34±10.55 | 76.41±9.09 | 0.1872 | -1.4536 to 7.3136 |
Types of Surgery in Breast CA for randomised patient groups.
| Types of Surgery in Breast CA | Group RP (n=40) | Group RM (n=40) | p-value |
|---|
| Simple Mastectomy (SM) | 8 (20%) | 10 (25%) | p=0.604 |
| Modified Radical Mastectomy (MRM) | 22 (55%) | 20 (50%) | p=0.7470 |
| Partial/Segmental Mastectomy (PM) | 7 (17.5%) | 8 (20%) | |
| Lumpectomy | 3 (7.5%) | 2 (5%) | |
Data are presented as n (%).
Intraoperative fentanyl requirement.
| Group RP (n=40) | Group RM (n=40) | p-value | 95% confidence interval |
|---|
| Intraoperative fentanyl requirement | 153.86±25.35 | 138.49±18.41 | 0.0027 | -25.2320 to -5.5080 |
| Intraoperative Propofol requirement | 150.34±18.93 | 134.23±15.02 | 0.0001 | 8.5033 to 23.7167 |
Magnesium group (group RM) received much less paracetamol (1 gm I.V) than control group (group RP) as rescue analgesic [Table/Fig-4]. The time of administration of rescue analgesic was much later in group RM than RP. Both the above mentioned results were statistically significant.
Comparison of time of rescue analgesia among two groups.
| Group RP (n=40) | Group RM (n=40) | p-value | 95% confidence interval |
|---|
| Time of administration of first rescue analgesia (hr) | 8.44±1.91 | 13.34±2.11 | 0.0001 | -5.7959 to -4.0041 |
| Total (iv) Paracetamol consumption in 24 hrs (mg) | 1592.09±220.12 | 1149.23±195.39 | 0.0001 | 350.2106 to 535.5094 |
For detection of pain, VAS score was used at different time interval. It was significantly low at rest (p<0.05) at 4,6,10 and 12 hours after surgery with a better pain control in group RM patients than in group RP patients [Table/Fig-5]. Pain was also assessed with ipsilateral shoulder movement and here also pain was significantly low (p<0.05) at 4,6,10 and 12 hours after surgery in magnesium group [Table/Fig-6].
Pain score (VAS) at rest in postoperative period.
| Postoperative Period (Hours) | Group RP (n=40) | Group RM (n=40) | p-value | 95% confidence interval |
|---|
| VAS0.5 | 2.25±0.52 | 2.02±0.61 | 0.0734 | 0.0223 to 0.4823 |
| VAS1 | 2.35±0.55 | 2.17±0.42 | 0.1040 | 0.0378 to 0.3978 |
| VAS2 | 2.91±0.60 | 2.70±0.51 | 0.0957 | -0.0379 to 0.4579 |
| VAS4 | 3.18±0.70 | 2.89±0.59 | 0.0486 | 0.0018 to 0.5782 |
| VAS6 | 3.52±0.80 | 3.10±0.72 | 0.0158 | 0.0812 to 0.7588 |
| VAS10 | 3.73±0.91 | 3.33±0.82 | 0.0422 | 0.0144 to 0.7856 |
| VAS12 | 3.84±1.04 | 3.39±0.89 | 0.0409 | 0.0191 to 0.8809 |
| VAS16 | 3.92±1.43 | 3.74±0.94 | 0.5079 | -0.3587 to 0.7187 |
| VAS24 | 3.82±1.22 | 3.49±0.83 | 0.1612 | -0.1345 to 0.7945 |
| VAS36 | 3.20±0.78 | 2.89±0.72 | 0.0685 | -0.0241 to 0.6441 |
| VAS48 | 2.88±0.78 | 2.61±0.48 | 0.0660 | -0.0183 to 0.5583 |
Pain on shoulder (ipsilateral) movement in postoperative period.
| Postoperative Period (Hours) | Group RP (n=40) | Group RM (n=40) | p-value | 95% confidence interval |
|---|
| VAS0.5 | 2.68±0.78 | 2.37±0.81 | 0.0852 | -0.0440 to 0.6640 |
| VAS1 | 2.70±0.66 | 2.45±0.53 | 0.0655 | -0.0165 to 0.5165 |
| VAS2 | 3.08±0.68 | 2.85±0.57 | 0.1052 | -0.0493 to 0.5093 |
| VAS4 | 4.15±0.47 | 3.70±0.76 | 0.0021 | 0.1687 to 0.7313 |
| VAS6 | 4.8±0.60 | 4.45±0.56 | 0.0086 | 0.0916 to 0.6084 |
| VAS10 | 5.03±0.71 | 4.62±0.59 | 0.0063 | 0.1194 to 0.7006 |
| VAS12 | 5.23±0.83 | 4.82±0.89 | 0.0363 | 0.0269 to 0.7931 |
| VAS16 | 5.15±0.74 | 4.89±0.59 | 0.0862 | -0.0379 to 0.5579 |
| VAS24 | 4.81±0.73 | 4.55±0.51 | 0.0686 | -0.0203 to 0.5403 |
| VAS36 | 4.17±0.34 | 3.92±0.46 | 0.7844 | -1.5633 to 2.0633 |
| VAS48 | 3.30±0.43 | 3.52±0.47 | 0.8304 | -2.2577 to 1.8177 |
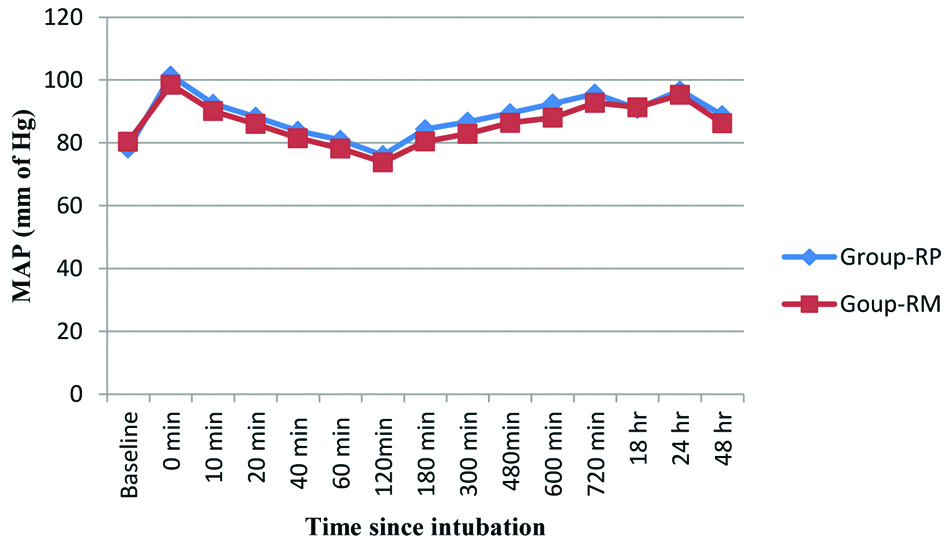
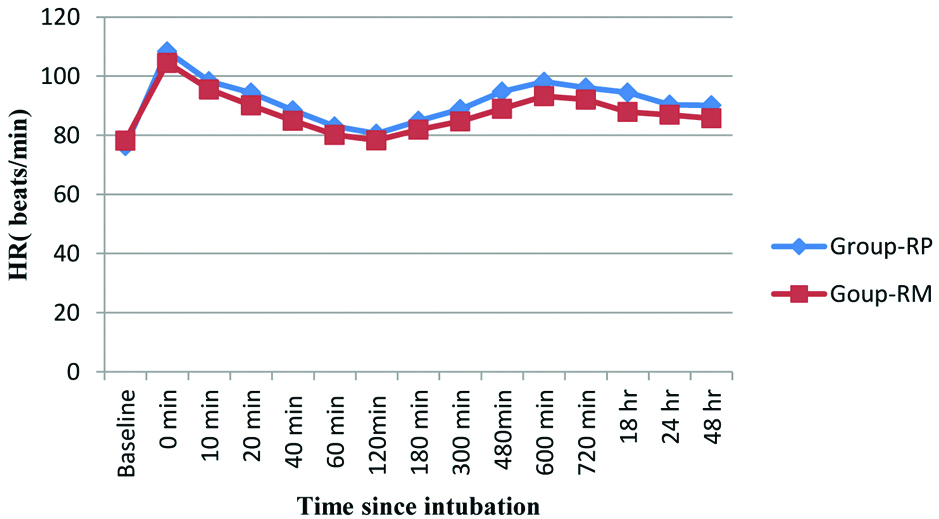
Side effects such as sedation, bradycardia, hypotension, dry mouth, nausea were all comparable among two groups (p>0.05) [Table/Fig-7]. MAP and HR were found to be quite comparable among two groups (p>0.05) [Table/Fig-8,9] respectively. The participant flow diagram is shown in [Table/Fig-10].
| Parameters | Group RP (n=40) | Group RM (n=40) | p-value |
|---|
| Nausea/Vomiting | 14 | 10 | 0.1441 |
| Sedation | 10 | 12 | 0.4901 |
| Bradycardia (HR <60 bpm) | 7 | 6 | 0.6570 |
| Hypotension (SBP <100 mm Hg) | 7 | 6 | 0.3271 |
| Dry mouth | 5 | 3 | 0.2299 |
Comparison of mean arterial pressure between two groups.
Comparison of mean heart rate between two groups.
Consort diagram showing flow of participants.

Discussion
Breast cancer as well as post-mastectomy persistent pain itself has a negative impact on quality of life in survivors [22-24]. With excellent pain management protocol, breast cancer surgery related acute post surgical pain can be alleviated; at the same time anxiety, morbidity, cost and length of hospital stay in the postoperative period can be decreased [25].
Different combinations of local anaesthetics and adjuvants have been tried in paravertebral block for the patients undergoing thoracic or breast cancer surgeries to reduce the perioperative pain [10-13]. Magnesium sulphate has recently been used in neuraxial anaesthesia, peripheral nerve block and nerve plexus blockade successfully [26-28].
In this randomised, prospective placebo control trial, we have evaluated the effect of 1 mL magnesium sulphate (500 mg) in thoracic paravertebral block for the patients undergoing breast cancer surgery under GA. Here, we measured the time and dose of administering first dose of rescue analgesic as paracetamol. Total dose of intraoperative analgesic fentanyl; haemodynamics and side effects were also assessed.
Information regarding various adjuvants in TPVB were available for conducting this study (like case series, multicentre clinical trials, meta-analysis of randomised trials, computerised database, etc.,), each with specific strengths and weaknesses. This review of literature greatly helped in drawing up the research questions, identifying the common possible side effects and taking adequate measures for them, devising a data extraction plan, extracting the data, checking for errors, data analysis, and appropriate archiving and dissemination of the findings. The ethical aspects in such studies greatly helped in framing informed consent and maintaining confidentiality of each participant.
In this prospective study, we had observed that rescue analgesic was required much later in magnesium group than control. Gunduz A et al., in a placebo controlled study on the role of magnesium along with prilocaine found significant prolongation (304 vs. 253 minutes) of postoperative analgesia [29]. Ammar AS et al., while performing a placebo controlled study for the patients undergoing thoracic surgery found paravertebral magnesium addition has significantly delayed (388.8±70.6 vs. 222.2±61.6 minutes) the requirement of morphine as rescue analgesic [30]. On the contrary Lee JH et al., found that epidural administration of magnesium from before the induction of anaesthesia to 48 hours postoperatively did not significantly decrease the incidence or severity of chronic postoperative pain in patients undergoing video-assisted thoracic surgery [31].
In this study, we have found that fentanyl requirement (as intraoperative analgesic) was much less in magnesium group (RM) as compared with control group (RP) and the outcome was statistically significant. This result was in accordance with a study conducted by Kaymak C et al., who found pre-administration of magnesium sulphate bolus followed by infusion caused a significant decrease in total consumption of fentanyl and midazolam in test group in shockwave lithotripsy procedure under monitored anaesthesia care [32].
In our study, intraoperative propofol requirement was higher in control group and this finding was very much similar to Choi JC et al., who found intravenous administration of magnesium sulfate reduces propofol infusion requirement significantly in the patients undergoing elective total abdominal hysterectomy [33].
In our study, postoperative paracetamol consumption was significantly less in magnesium group than control. Similar findings were observed by Mohamed KS et al., and they found postoperative analgesic consumption as ketorolac was significantly lower in the magnesium group (17.00 vs. 30.00 mg) than in the bupivacaine group [34].
We found, VAS score was significantly higher at 4, 6, 10, 12 hours in control group. Thus in our study magnesium showed effective analgesic effects. Kaymak C et al., in their study found that intravenous magnesium had reduced VAS score significantly at 15, 20, 25 minutes time interval [32]. Similar findings were observed by Mohamed KS et al., who found intrathecal magnesium use caused a lower VAS score in study group [34].
In our study, we had found sedation was quite similar and comparable among two groups and the sedation was slightly higher in magnesium group. But on the contrary, Ammar AS et al., found lesser somnolence rate in paravertebral magnesium group than control group [30]. In our study, bradycardia and hypotension both were comparable among two groups and both the values were slightly less in magnesium group. Similar findings were also observed by Shabana R et al. They found slightly higher systolic blood pressure in magnesium group after 24 hours of operation but at the same time heart rate was slightly lower in magnesium group [35].
Again among side effects in our study nausea, vomiting was quite comparable and magnesium produced slightly lower nausea and vomiting. Mohamed KS et al., also found lesser nausea, vomiting in magnesium group and they were also statistically insignificant [34].
Limitation
Among the study limitations, though we had measured BISS (bispectral index) for anaesthesia depth assessment, we did not compare the same among two groups. We did not measure the plasma catecholamine concentration which ideally should have been decreased by magnesium. In future, a larger study with larger sample size needs to be conducted to establish author’s point of view with solidarity.
Conclusion
It can be concluded that preoperative magnesium (500 mg) administered along with thoracic paravertebral block prior to induction of general anaesthesia, will prolong intraoperative and postoperative analgesia. It also renders a lesser analgesic requirement without major haemodynamic alteration and side effects.
Data are presented as n (%).