Leiomyomatous Nodule and Ovarian Mucinous Borderline Tumour: A Rare Co-existence
Sadaf Khan1, Swati Sharma2, Jyothi Shetty3
1 Postgraduate Student, Department of Pathology, Kasturba Medical College, Manipal Academy of Higher Education, Karnataka, India.
2 Associate Professor, Department of Pathology, Kasturba Medical College, Manipal Academy of Higher Education, Manipal, Karnataka, India.
3 Professor, Department of Obstetrics and Gynecology, Kasturba Medical College, Manipal Academy of Higher Education, Manipal, Karnataka, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Swati Sharma, Associate Professor, Basic Sciences Building, Department of Pathology, Kasturba Medical College, Manipal Academy of Higher Education, Manipal, Karnataka, India.
E-mail: swatisharma79@yahoo.com
Occurrence of ovarian cystic neoplasms with mural nodules are rare entities, however well established. We report a case of a leiomyomatous mural nodule co-existing with an ovarian mucinous borderline tumour. To the best of our knowledge, this is the first case of a leiomyomatous nodule co-existing with an ovarian mucinous borderline tumour in the English literature and it becomes more interesting owing to the presence of ovarian mucinous neoplasms in both the ovaries which is also a rare phenomenon. Primary leiomyomas of ovary are rare ovarian tumours and must be considered as a possible differential whenever cystic ovarian neoplasms are detected with solid component radiologically, since they have a benign course and a good prognosis.
Cystic neoplasm, Mural nodules, Ovary
Case Report
A 43-year-old diabetic female on tablet metformin 125 mg OD, came with complaints of spotting per vaginum since 12 days, abdominal pain for 15 days and post meal distension. No history of vomiting/constipation/burning micturition or breathlessness was present. On examination a soft, non-tender, vague mass per abdomen was felt. Per vaginally uterus was anteverted and normal in size, however right flank fullness was noted. Routine investigations were within normal limits. Utrasonographic examination showed a huge cystic mass measuring 21×17.6 cm with multiple loculations, echogenic fluid and moderate vascularity located behind the uterus and extending upto the xiphisternum, occupying the whole abdomen suggestive of mucinous neoplasm.
After preoperative cardiac clearance, patient underwent staging laparotomy– total abdominal hysterectomy, bilateral salpingoophorectomy, infracolicomentectomy and appendicectomy under general anesthesia. The postoperative period was uneventful and the specimens were sent for histopathological examination.
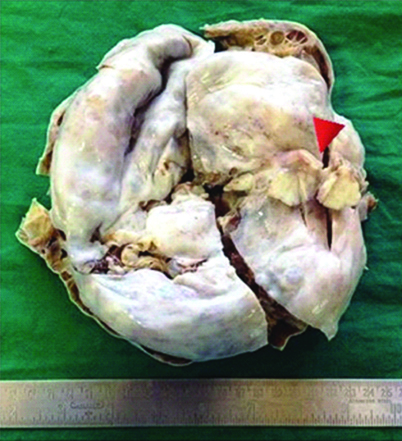
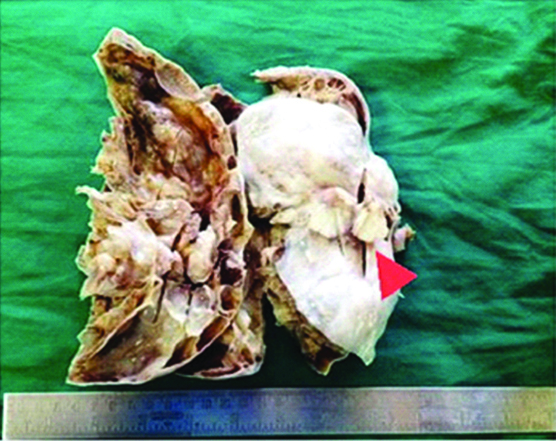
Grossly, uterus and cervix were unremarkable. Cut section of left ovary showed two cortical cysts, larger measuring 1.5×1 cm filled with mucoid material. Right ovarian mass measured 16×15×5 cm, externally showed a nodular growth measuring 2×2 cm and cut section showed grey white areas with no necrosis and haemorrhage [Table/Fig-1]. Cut section of right ovarian mass showed multiple cysts filled with seromucinous fluid and pus along with focal solid areas [Table/Fig-2]. Omentum and appendix showed no deposits grossly.
Grossly, Right ovarian mass externally showed a nodular growth measuring 2×2 cm (red arrow head).
Cut section of right ovarian mass showed multiple cysts filled with seromucinous fluid and focal solid areas. Externally nodular growth noted on the surface (red arrow head).
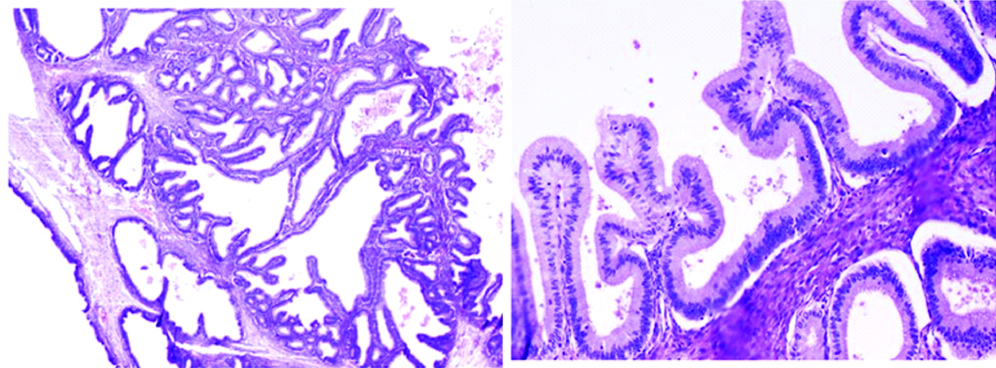
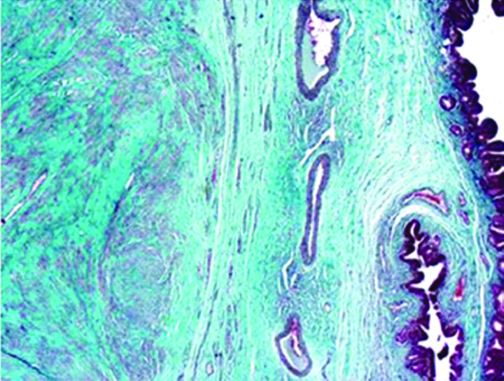
Microscopically, left ovary showed a cyst lined by mucinous tall columnar to flattened epithelium with intraluminal mucin. Section from right ovarian mass showed variably sized cystic spaces, focally thrown into papillary folds and confluent slender papillae lined by tall columnar epithelium, focally showing pseudostratification along with goblet cells [Table/Fig-3,4]. Intraluminal cell debris, foamy macrophages and brisk mitosis was also noted. Section from the attached nodular growth showed a benign tumour composed of interlacing fascicles and bundles of smooth muscle fibers with scant eosinophilic wispy cytoplasm, elongated to plump vesicular nuclei surrounded by stroma with areas of hyalinization and congested vessels [Table/Fig-5]. No necrosis or increase in mitosis was seen. Smooth muscle fibers were highlighted by staining with Masson’s trichrome stain [Table/Fig-6]. A diagnosis of left ovarian mucinous cystadenoma and right ovarian mucinous borderline tumour along with leiomyoma was given.
Microscopically, variably sized cystic spaces, focally thrown into papillary folds and confluent slender papillae lined by tall columnar epithelium, focally showing pseudostratification along with goblet cells are seen (Haematoxylin and eosin X100, X200).
Microscopically, attached nodular growth showed interlacing fascicles and bundles of smooth muscle fibers. No necrosis or increase in mitosis was seen (Haematoxylin and eosin X40).
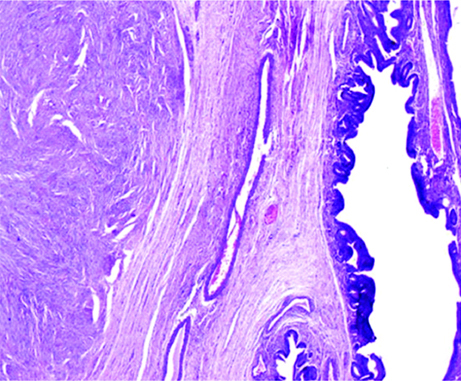
Smooth muscle fibres were highlighted by staining with Masson’s trichrome stain (Masson’s Trichrome, X40).
Discussion
The co-existence of mural nodules with serous or mucinous tumours of the ovary including benign, borderline or malignant though is rare, yet is a well reported entity with an incidence between two to five per million [1]. They are a heterogeneous group of lesions which can be reactive, benign or neoplastic and can mimic malignancy not only clinically and radiologically being misinterpreted as solid component of the accompanying cystic tumour, but also histologically in cases of reactive mural nodules. Reactive and benign mural nodules should be distinguished from malignant ones as they carry good prognostic results. Mural nodules have been divided into four varieties, i.e., Sarcoma-Like Mural Nodules (SLMN), true sarcomas, foci of anaplastic carcinoma and mixed type [2]. The existence of mucinous tumours of the ovary with “sarcoma like nodules”, which are reactive lesions histologically mimicking sarcoma, was first described by Prat and Scully [3]. In the literature, there are only few reported cases of leiomyomatous mural nodule within a benign or malignant mucinous cystic ovarian tumour. These mural nodules can project into the lumen of the cystic cavity or can be located as solid lesions on the wall of the ovarian tumour. There has also been evidence of Mucinous cystic neoplasms in other organs co-existing with secondary tumours of other histological type, such as sarcomas, anaplastic carcinomas and malignant fibrous histiocytoma [1].
The aetiology and pathogenesis of the mural nodules is still debatable. Mural nodules have not only been reported in association with ovarian cancer but also with mucinous cystic neoplasm of the pancreas and gallbladder. This can be attributed to the resemblance of the stroma neighboring the pancreatic mucinous neoplasm to the ovarian stroma by the virtue of positive stainability for Estrogen receptor, Progesterone receptor and Inhibin. These findings are suggestive of a possible derivation of the mural nodules from potential pluripotent cells in the stroma of mucinous neoplasms. The theory of ‘collision tumours’ can also explain the co-existence of mural nodules with mucinous neoplasm [1,4].
We describe a case of a benign mural nodule consistent with ovarian leiomyoma on the surface of a large ovarian cyst, diagnosed as mucinous borderline tumour of the ovary on the basis of histopathological examination and immunohistochemistry. The other ovary also had a mucinous cystadenoma. Rare cases of bilateral primary ovarian mucinous neoplasms have been reported in the literature [5]. Mucinous borderline tumour of the ovary is composed of cyst lined by gastrointestinal type epithelium with varying degrees of stratification.
As per the English medical literature, to the best of our knowledge, four cases of leiomyomatous mural nodules, rare benign, spindle cell lesion co-existing with benign mucinous cystadenoma have been described; this case being the first one where leiomyomatous nodule is noted in a mucinous borderline tumour. The first case of leiomyomatous mural nodule in a benign mucinous cystadenoma was described by Lifschitz-Mercer B et al., in 1990, second by Nichols GE et al., in 1991, third by Hameed A et al., in 1997 [6-8] and fourth by Mathew M et al., in 2013 [4]. Nichols GE et al., have emphasised on the role of IHC in distinguishing the various types of mural nodules [7]. They observed that malignant spindle cells co-expressed both vimentin and cytokeratin, however, leiomyomatous nodules were negative for cytokeratin and expressed desmin, vimentin, and muscle-specific actin. According to Hameed A et al., leiomyoma of ovary can arise from smooth muscle of mucinous cystadenoma [8].
It has been estimated that ovarian leiomyoma has an association with its uterine counterpart in almost 78 % cases [9]. However, in the present case, only a solitary ovarian leiomyoma was seen in absence of any uterine leiomyoma even on careful serial sectioning, hence making it a primary tumour of the ovary. Primary leiomyoma are rare tumours and account for less than 1% of benign ovarian tumours. Although leiomyomas have a characteristic whorling pattern on gross and microscopic examination but by virtue of its rarity, fibroma, cellular fibroma, thecoma and sclerosing stromal tumour should be included in the differential diagnosis [10,11]. Masson’s trichrome can be utilised to distinguish smooth muscles of the leiomyoma from fibrous component in the lesion. Use of desmin can aid in the diagnosis, being diffusely positive in leiomyomas whereas negative or only focally positive in fibromatous tumours. Leiomyomas are positive for alpha-Smooth Muscle Actin (α-SMA) while the comas do not express α-SMA and instead are positive for α-inhibin and calretenin. Although very rare, leiomyosarcoma should also be ruled out using criteria like cytological atypia, mitotic count, and tumour necrosis.
It is imperative to classify these mural nodules for planning the treatment of the patient, postoperative therapy and follow-up, they also have a direct impact on the prognosis of the associated ovarian tumour [12,13]. It is essential to make an accurate diagnosis since well demarcated and non-invasive leiomyomatous nodules or sarcoma-like mural nodules carry an excellent prognosis and their presence does not have an impact on the prognosis of the ovarian tumour associated with it, however presence of any sarcomatous component should be ruled out because co-existence of true sarcomatous nodules with the sarcoma like mural nodules is another rare entity which imparts a poor prognostic outcome and has been reported, thus demanding careful histological analysis [14].
Conclusion
Ovarian neoplasms of both serous and mucinous type can be associated with benign or malignant mural nodules, which can radiologically as well as grossly appear as solid lesions raising the suspicion of malignancy. Awareness of the different types of mural nodules and a complete evaluation of morphology are the key to correct diagnosis however, immunohistochemistry can assess in elucidating some of these diagnostic difficulties.
[1]. Yamazaki H, Matsuzawa A, Shoda T, Iguchi H, Kyushima N, Ovarian mucinous cystic tumour of borderline malignancy with a mural nodule of anaplastic spindle cell carcinoma: a case reportJ Ovarian Res 2013 6(1):8610.1186/1757-2215-6-8624305620 [Google Scholar] [CrossRef] [PubMed]
[2]. Ghosh P, Saha K, Bhowmik S, Sarcoma-like mural nodule in a borderline mucinous tumour of the ovary: A rare entityJ Midlife Health 2014 5(4):192-94.10.4103/0976-7800.14516825540570 [Google Scholar] [CrossRef] [PubMed]
[3]. Prat J, Scully RE, Ovarian mucinous tumours with sarcoma-like mural nodules: a report of seven casesCancer 1979 44(4):1332-44.10.1002/1097-0142(197910)44:4<1332::AID-CNCR2820440426>3.0.CO;2-# [Google Scholar] [CrossRef]
[4]. Mathew M, Gonsalves H, Al-Azawi S, Saparamadu PA, Asymptomatic ovarian mucinous cystadenoma with a solid mural leiomyoma: case report and brief reviewSultan Qaboos Univ Med J 2013 13(1):127-31.10.12816/000320623573393 [Google Scholar] [CrossRef] [PubMed]
[5]. Robert JK, Maria LC, Simon CH, Robert HY, WHO Classification of Tumours of Female Reproductive Organs 2014 4th edIARCLyon:26-27 [Google Scholar]
[6]. Lifschitz-Mercer B, Dgani R, Jacob N, Fogel M, Czernobilsky B, Ovarian mucinous cystadenoma with leiomyomatous mural noduleInt J Gynecol Pathol 1990 9(1):80-85.10.1097/00004347-199001000-000082294066 [Google Scholar] [CrossRef] [PubMed]
[7]. Nichols GE, Mills SE, Ulbright TM, Czernobilsky B, Roth LM, Spindle cell mural nodules in cystic ovarian mucinous tumours. A clinicopathologic and immunohistochemical study of five casesAm J Surg Pathol 1991 15(11):1055-62.10.1097/00000478-199111000-000041656803 [Google Scholar] [CrossRef] [PubMed]
[8]. Hameed A, Ying AJ, Keyhani-Rofagha S, Xie Dl, Copeland LJ, Ovarian Mucinous Cystadenoma Associated with Mural Leiomyomatous Nodule and Massive Ovarian EdemaGynecol Oncol 1997 67(2):226-29.10.1006/gyno.1997.48679367713 [Google Scholar] [CrossRef] [PubMed]
[9]. Doss BJ, Wanek SM, Jacques SM, Qureshi F, Ramirez NC, Lawrence WD, Ovarian leiomyomas: clinicopathologic features in fifteen casesInt J Gynecol Pathol 1999 18(1):63-68.10.1097/00004347-199901000-000099891243 [Google Scholar] [CrossRef] [PubMed]
[10]. Vierhout ME, Pijpers L, Tham MN, Chadha-Ajwani S, Leiomyoma of the ovaryActa Obstet Gynecol Scand 1990 69(5):445-47.10.3109/000163490090133122270773 [Google Scholar] [CrossRef] [PubMed]
[11]. Lastarria D, Sachdev RK, Babury RA, Yu HM, Nuovo GJ, Immunohistochemical analysis for desmin in normal and neoplastic ovarian stromal tissueArch Pathol Lab Med 1990 114(5):502-05. [Google Scholar]
[12]. De Rosa G, Donofrio V, De Rosa N, Fulciniti F, Zeppa P, Ovarian serous tumour with mural nodules of carcinomatous derivation (sarcomatoid carcinoma): report of a caseInt J Gynecol Pathol 1991 10(3):311-18.10.1097/00004347-199107000-000101917278 [Google Scholar] [CrossRef] [PubMed]
[13]. Guilbeau C, Soubeyrand MS, Devillebichot C, Sage M, Collin F, Arnould L, Mural nodules of anaplastic carcinoma in bilateral ovarian borderline mucinous cystadenoma: a case reportAnn Pathol 2003 23(4):340-44. [Google Scholar]
[14]. Gungor T, Altinkaya SO, Akbay S, Bilge U, Mollamahmutoglu L, Malign mural nodules associated with serous ovarian tumour of borderline malignancy: a case report and literature reviewArch Gynecol Obstet 2010 281(3):485-90.10.1007/s00404-009-1180-319597831 [Google Scholar] [CrossRef] [PubMed]