The use of laser for cavity preparation as well as for conditioning of enamel and dentin surfaces instead of acid etching has increased in recent years [1,2]. The Erbium-Doped Yttrium-Aluminum-Garnet (Er:YAG) laser is one of the most preferred laser types used in preparation of dental hard tissues. Er:YAG laser ablation of tooth substance is effective and safe since its 2.94 μm wavelength is selectively absorbed by water within the hard tissue [3]. Laser ablates the hard tissues by thermomechanical interaction which is based on the microexplosions of water molecules. Therefore; laser, when used with air and water coolant, has been shown not to produce major heat-related side effects, such as cracking, melting, or carbonisation of the tooth or surrounding tissues [4]. Other benefits of using lasers for cavity preparation include antibacterial effects, minimal vibration and noise [5]. The need for local anesthesia is significantly reduced due to the less increase in intrapulpal temperature during laser preparation in comparison to the bur preparation [6]. On the other hand, some concerns have been raised about the use of lasers in cavity preparation due to the resultant surface alterations when compared to bur-prepared cavities. Er:YAG laser irradiation of dentin has shown to yield open dentinal tubules without smear layer formation and a surface with microirregularities due to the selective removal of the intertubular dentin [7,8]. Since laser irradiation alters the surface texture of dental hard tissues, adhesion of restorative materials to tooth surfaces could be poor and may result in microleakage [9].
Microleakage can be defined as the diffusion of oral fluids, bacteria and bacterial toxins between the cavity wall and the restorative material due to impaired marginal sealing [10]. This is regarded as one of the most important problems following dental restorations and therefore, the main reason for secondary caries and the resultant pulpal inflammation [11]. The key to providing an optimal marginal sealing in laser-prepared cavities is related to the successful interaction of factors such as laser irradiation, adhesive systems, and dental substrate. Since lased dentin surfaces have significantly different physiological dynamics, heterogeneous composition, and complex tubular structure, adhesion performance of current adhesive systems which were originally developed to be applied on bur-prepared surfaces may considerably differ on laser-irradiated dental surfaces [11,12]. The literature contains conflicting data regarding the microleakage of composite resin restorations when dental cavities were prepared by erbium lasers. While some studies showed favourable results with laser preparation [13,14], others reported that laser preparation did not favour the adhesion quality or even reduced it [5,15].
Since various resin-adhesive systems have been developed to be used in dental restorations and controversies exist regarding the use of lasers for cavity preparation, there is still a need for further research focusing on determining the quality of restoration margins in laser-prepared cavities bonded with different adhesive systems. It has been shown that an adhesive system showing good results in conventionally prepared cavities, can exhibit poor results in laser-prepared cavities or vice versa. SEM analysis of the resin–dentin interfaces and shear bond strength tests were carried out in a previous study by the same authors using the same adhesive systems and laser parameters [16]. Therefore, the aim of this in vitro study is to investigate the microleakage of composite restorations in Er:YAG laser and bur-prepared Class V cavities after application of three different adhesive systems. The tested hypothesis was that adhesive systems used in the study showed similar microleakage levels in the cavities prepared by laser or bur.
Materials and Methods
Tooth Preparation
The present study was an in vitro study. A total of 80 caries and restoration free human third molars, extracted as a part of dental treatment in the Oral Surgery Department, were used in the study after having obtained informed consent from the subjects. Following the extraction, residual soft tissues on the root surfaces were removed with a scaler and the teeth were cleaned with pumice-water slurry using a low-speed handpiece. The cleaned teeth were stored in distilled water at 4°C for a maximum of six months until their use. The specimens were randomly divided into eight groups of 10 teeth each according to cavity preparation and adhesive treatment modalities: Group 1: Laser preparation + Clearfil Tri-S (C3S) Bond (Kuraray Medical, Tokyo, Japan) (a one-step self-etching adhesive); Group 2: Laser preparation + Adper SE Plus (SE) Bond (3M ESPE, St. Paul. MN, USA) (a two-step self-etching adhesive); Group 3: Laser preparation + laser etching + Adper Single Bond 2 (SB2) (an etch-and-rinse adhesive); Group 4: Laser preparation + acid etching + SB2; Group 5: Laser preparation + SB2 (no etching); Group 6: Bur + acid etching + SB2; Group 7: Bur + C3S Bond; Group 8: Bur + SE Bond. The groups prepared conventionally (Groups 6-8) served as control groups.
Cavity Preparation and Restoration
Class V cavities were prepared on the buccal surfaces of the teeth either by Er:YAG laser or diamond bur in a high-speed handpiece. Cavity dimensions were standardised using a template prepared from a metal band strip for outlining the cavity with 4 mm of mesiodistal width and 3 mm of occlusogingival height. Gingival margins of the cavities were located 1mm below the cemento-enamel junction while occlusal margins were located in enamel [Table/Fig-1]. The depths of the cavities were 1.5 mm, measured with a marked periodontal probe.
a. A template prepared from a metal band strip was used for outlining the cavity; b. Cavity preparation by laser; c. Cavity preparation by bur; d. Coating with nail varnish and boxing wax; e. Sectioning with diamond saw; f. 1mm thick two slices were obtained per tooth.
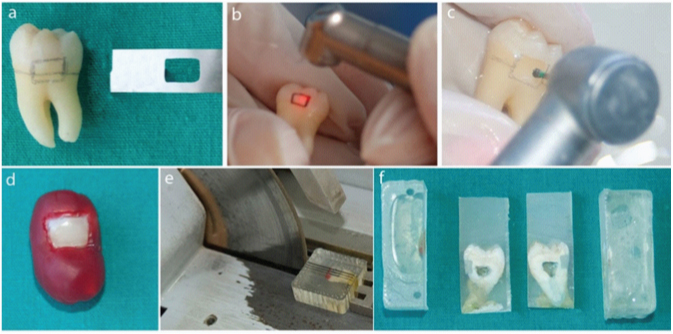
An Er:YAG laser with a wavelength of 2.94 μm (Fidelis Plus III, Fotona Medical Lasers, Ljubljana, Slovenia) and the adhesive systems were used according to manufacturers’ instructions. The laser energy was applied on specimens by a non-contact RO2-C handpiece (Fotona Medical Lasers, Ljubljana, Slovenia) with a focal spot size of 0.9 mm in diameter under continuous water spray at a distance of 7 mm from the target point. The laser energy parameters were determined as 300 mJ/20Hz for enamel preparation, 200 mJ/20Hz for dentin preparation, 120 mJ/10Hz for enamel etching and 80 mJ/10Hz for dentin-etching procedures, according to manufacturer’s instructions. The pulse duration was 100 μs (very short pulse).
Conventional cavity preparations were performed with diamond fissure (#10) and reverse cone-shaped (#12) burs (Strauss, Ra’anana, Israel) in a high-speed handpiece (KaVo Dental, Biberachan der Riß, Germany) using an air and water spray coolant. Bur was changed after every five preparations. In order to prevent excessive desiccation, the cavity surfaces were dried by cotton pellets instead of compressed air. Specimens in Groups 4 and 6 were etched with 35% phosphoric acid (Scotchbond; 3M ESPE Dental) for 15 seconds and rinsed with water for 15 seconds. The adhesives were light cured for 10 seconds (EliparFreeLight 2 LED, 3M ESPE Dental).
All cavities were restored with a universal hybrid composite resin (Filtek Z250; 3M ESPE Dental) using layering technique. Each increment was polymerised for 20 seconds. All restorations were polished using Sof-Lex XT (3M ESPE Dental) discs. A single operator performed all the procedures. All samples were kept in distilled water at 37°C for 24 hours, and then thermocycled 500 times each at 5±2°C and 55±2°C. The cycles lasted 30 seconds in each bath, with a 3 second transfer time.
Microleakage Evaluation
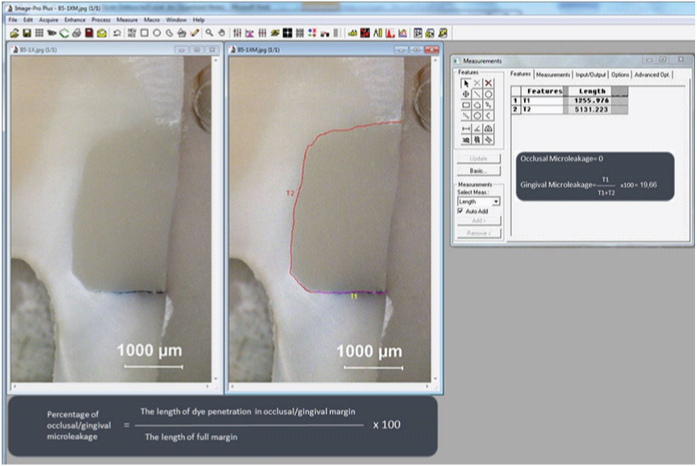
Microleakage was assessed by dye penetration based on the method of Arcoria CJ et al., [17]. After the specimens were dried, a double layer of transparent nail varnish was applied to cover the entire tooth surfaces except the restoration and its 1mm surroundings. Following drying, the varnished surfaces were also coated with boxing wax for a complete sealing. The specimens were then immersed in 0.5 % buffered methylene blue dye solution and kept in darkness for 24 hours. They subsequently were rinsed under running tap water to remove excess dye. The wax was removed and all the surfaces were brushed. After that, the teeth were embedded in clear epoxy resin (EpoFix; Struers A/S, Ballerup, Denmark) using polyethylene molds sized 3.5x4.5x1.5cm. The specimens were bisected longitudinally through the centre of the restorations with diamond saw (Discoplan TS, Struers A/S) under water cooling. Each half was then further sectioned in a vestibulolingual direction through the mesial and distal sides of the restorations, providing two 1mm thick tooth slice. Following sectioning, the cuts were flattened and polished using 400, 800, 1000 and 2000 grit wet sandpapers. Each restoration had two slices and microleakage was calculated on both sides of each slice, giving a total of four measurements per tooth [Table/Fig-1]. The surface that showed the highest degree of dye penetration per specimen was selected for observation. For analysis of dye penetration, images of each surface were taken with a digital camera (SPOT Insight; Diagnostic Instruments, Sterling Heights, MI, US) attached to a trinocular stereomicroscope (Stemi SV 6, Carl Zeiss, Göttingen, Germany) under 25x magnification. Digitised images of the sections were collected using the image analysis system (Image-Pro Plus v 4.5, Media Cybernetics, Rockville, MD, US). Gingival and occlusal microleakage were calculated according to the methods used by Bertrand MF et al., and accordingly the length of dye penetration along the occlusal margin, the length of dye penetration along the gingival margin and the total length of interface were recorded in millimeters for each specimen [18]. Occlusal and gingival microleakage percentages were calculated by dividing the length of dye penetration on either the occlusal or gingival side by the total length of the interface [Table/Fig-2].
Calculation of the percentages of gingival and occlusal microleakage.
Statistical Analysis
The sample size was calculated with G*Power 3.1.9 statistical program. The minimum sample size was calculated as six per group when alpha risk was set at 0.05 and effect size at 0.807. The sample size was increased to 10 for each group to achieve more reliable results.
Statistical analysis was performed by NCSS 2007 & PASS 2008 (NCSS Statistical Software, Kaysville, Utah, US) software. Non-parametric Kruskal–Wallis test was used to detect differences among groups and Mann–Whitney U-test was carried out for pairwise comparisons. Wilcoxon test was used to compare occlusal and gingival microleakage in each group. The significance level was set at p<0.05.
Results
The mean and standard deviation of occlusal and gingival microleakage data for each group are shown in [Table/Fig-3]. The results of each pairwise comparison are shown in [Table/Fig-4]. The Kruskal–Wallis test indicated significant differences among the experimental groups, both for the occlusal (KW:20.387, p:0.005) and the gingival scores (KW:21.164, p:0.004). The highest microleakage was observed in the gingival interface of Group 5. The lowest microleakage was seen in the occlusal interface of the SB2-applied groups subsequent to laser preparation + laser etching (Group 3) and bur preparation + acid etching (Group 6). When the effects of laser and bur preparation methods on occlusal and gingival microleakage were compared, the statistically significant difference was found between the SE Bond-applied groups on the occlusal surface (Groups 2 and 8). The laser ablated group showed less microleakage on the occlusal wall than the bur treated group (p=0.027, p<0.05). No statistically significant differences were observed (when compared amongst the same adhesive groups treated differently) between the gingival or occlusal microleakage percentages of the SB2 and the C3S Bond applied groups, regardless of whether dentin was laser ablated or bur treated (p>0.05).
Mean percentages of microleakage and standard deviation of the groups in occlusal and gingival margins.
| Occlusal | Gingival | Wilcoxon Signed test |
|---|
| Mean±SD(Median) | Mean±SD(Median) | Z-value | p-value |
|---|
| Laser+C3S Bond (G1) | 7.07±11.45 (0) | 4.35±8.42 (0) | 0.000 | 1.000 |
| Laser+SE Bond (G2) | 1.10±2.55 (0) | 6.43±10.40 (0) | -1.604 | 0.109 |
| Laser+laser etching+SB2 (G3) | 0±0 (0) | 16.12±12.29 (16.28) | -2.521 | 0.012* |
| Laser+acid etching+SB2 (G4) | 0.40±1.26 (0) | 3.71±8.65 (0) | -1.826 | 0.068 |
| Laser+SB2 (G5) | 6.75±9.50 (0) | 37.90±28.28 (53.4) | -2.100 | 0.036* |
| Bur+acid+SB2 (G6) | 0±0 (0) | 2.24±5.60 (0) | -1.342 | 0.180 |
| Bur+C3S Bond (G7) | 3.18±4.09 (1.4) | 2.62±6.17 (0) | -0.943 | 0.345 |
| Bur+SE Bond (G8) | 19.70±29.01 (11.3) | 28.43±34.78 (4.8) | -0.059 | 0.953 |
| Kruskal Wallis Test | KW:20.387 p:0.005** | KW:21.164 p:0.004** | | |
Post-Hoc Mann-Whitney U-test was used to compare two groups. Wilcoxon Signed test was used to compare occlusal and gingival microleakage.
A p-Values obtained after Post-hoc Mann-Whitney U-test comparing the microleakage means at the gingival and occlusal margins.
| G1 | G2 | G3 | G4 | G5 | G6 | G7 | G8 |
|---|
| O | G | O | G | O | G | O | G | O | G | O | G | O | G | O | G |
|---|
| G1 | O | | | 0.487 | | 0.048 | | 0.214 | | 0.535 | | 0.048 | | 0.579 | | 0.098 | |
| G | | | | 0.889 | | 0.017 | | 0.859 | | 0.007 | | 0.551 | | 0.816 | | 0.186 |
| G2 | O | | | | | 0.147 | | 0.543 | | 0.192 | | 0.147 | | 0.156 | | 0.027 | |
| G | | | | | | 0.081 | | 0.859 | | 0.014 | | 0.427 | | 0.675 | | 0.216 |
| G3 | O | | | | | | | 0.317 | | 0.031 | | 1 | | 0.013 | | 0.005 | |
| G | | | | | | | | 0.015 | | 0.129 | | 0.015 | | 0.009 | | 0.938 |
| G4 | O | | | | | | | | | 0.091 | | 0.317 | | 0.056 | | 0.013 | |
| G | | | | | | | | | | 0.008 | | 0.456 | | 0.723 | | 0.247 |
| G5 | O | | | | | | | | | | | 0.031 | | 0.679 | | 0.374 | |
| G | | | | | | | | | | | | 0.004 | | 0.006 | | 0.699 |
| G6 | O | | | | | | | | | | | | | 0.013 | | 0.005 | |
| G | | | | | | | | | | | | | | 0.691 | | 0.092 |
| G7 | O | | | | | | | | | | | | | | | 0.113 | |
| G | | | | | | | | | | | | | | | | 0.691 |
| G8 | O | | | | | | | | | | | | | | | | |
| G | | | | | | | | | | | | | | | | |
| Mann-Whitney U-Test. |
O. Occlusal; G. Gingival; Bold numbers indicate significant differences (p<0.05)
In the laser-prepared groups (G1-G5), no significant differences were found between self-etch applied groups (G1 and G2). In the laser-treated and the SB2-applied groups (G3-G5), the acid etched-applied groups (G4) showed significantly less gingival microleakage than the laser-etched (G3) (p=0.015) and the non-etched group (G5) (p=0.008). In addition, the laser-etched group (G3) demonstrated less occlusal microleakage than the non-etched group (G5) (p= 0.031). In the bur-treated groups (G6-G8), the SB2-applied group (G6) showed no microleakage on the occlusal margin and demonstrated less microleakage than the C3S (p=0.013) and SE Bond (p=0.005) groups [Table/Fig-4]. When the occlusal and gingival leakage percentages were compared in each group, statistically significant differences were observed only in the laser prepared + laser etched + SB2 applied group (G3) (p=0.012) and the laser ablated + SB2 applied group (G5) (p=0,036) [Table/Fig-3]. Significant differences in occlusal microleakage were also found between G1 and G3, G1 and G6, G3 and G7, G3 and G8, and G4 and G8. Gingival microleakage was also significantly different between G1 and G3, G1 and G5, G3 and G7, G3 and G8, and G5 and G7 [Table/Fig-4].
Discussion
This study was carried out to assess the efficacy of the Er:YAG laser and conventional (diamond bur in a high-speed handpiece) cavity preparation methods on the marginal microleakage of Class V cavities bonded with three different adhesive systems. The null hypothesis was rejected since the results indicated that adhesive systems used in the study showed different levels of dye penetration in the cavities prepared either by laser or by diamond bur.
Dye penetration method is the most widely accepted method for assessing microleakage along the dentin–restorative interface [19]. Regarding dye penetration technique, microleakage can be measured by qualitative assessment using previously established scores or by quantitative assessment using digital images and a specific software to measure the dye length in millimeters, micrometers or percentages [20]. In the present study, the interpretation of the results has been performed quantitatively by measuring the dye penetration and then transforming the measurements into percentages. Qualitative assessment by percentages provided a more objective evaluation than scoring.
Several studies have demonstrated a higher degree of microleakage along tooth/composite interfaces when cavity preparation was performed by Er:YAG laser [15,21]. Increased microleakage in laser-prepared cavities was attributed to the non-uniform microporosities created by laser ablation and the disorganised destruction of enamel prisms. This irregular surface pattern would result in poor adhesion in the resin–dentin interface due to inadequate sealing of cavity margins [21]. It is important to note that the laser settings in these two studies were considerably higher and it is known that an increase in pulse energy results in a deeper crater pattern in the tooth surface, which adversely influences marginal integrity of the restorative material [22]. Phanombualert J et al., compared the microleakage in laser and bur prepared cavities using three different energy density parameters and concluded that less microleakage was noticed with low-energy pulses and that low laser settings contributed to a better bonding than high-power laser settings [23]. Peker S et al., investigated the effect of cavity preparation techniques (bur or laser) on the microleakage levels of three glass ionomers and one composite resin restorations [24]. Their results demonstrated that Er:YAG laser showed less microleakage then bur preparation in all groups except for a glass ionomer applied group. Some researchers have found that microleakage values in laser-prepared cavities were comparable to those of conventional preparation [13,25]. El Haddar YS et al., compared the microleakage of three self-etch adhesive systems in cavities prepared by Er:YAG laser or bur and reported that there was no significant difference between the laser or bur preparations or between the adhesive systems [26]. Their results suggests that Er:YAG laser can be used for cavity preparation effectively, as an alternative to conventional bur preparations. In the present study, Er:YAG laser-prepared cavities showed similar results to bur-prepared cavities in SB2 and S3 Bond-applied groups. The only statistically significant difference was found between SE Bond-applied groups where Er:YAG laser-prepared cavities presented less microleakage than bur-prepared cavities. This finding is also consistent with the results of authors’ previous study that used the same adhesive systems but evaluated the shear bond strength and ultrastructure of the adhesive–dentin interfaces. In SE Bond-applied groups, the hybrid layer in lased cavities was thicker and shear bond strength values was higher when compared to bur preparation. This improved adhesion of SE Bond can be explained by the chemically modified surface created by laser ablation [16]. Laser ablation increases the calcium-to-phosphorus ratio and decreases the carbon-to-phosphorus ratio in the surface [27]. Guven Y et al., proposed that carboxyl groups in SE Bond can chemically bond to hydroxyapatite and calcium, thus forming stable calcium salts that improve resin adhesion by the formation of strong ionic interactions between the tooth and adhesive layer [16]. In contrary to the present study, Yaman BC et al., compared Adper SE Bond in laser and bur prepared cavities and found no significant differences between cavity preparation methods. These different results may depend on different laser parameters used in their study or on the composite resin [28].
Most of the studies indicated that microleakage was higher on the gingival wall than on the occlusal wall [28-32]. This difference is attributed to the fact that technique-sensitivity of bonding to dentin is higher than bonding to enamel [5]. It may also be related to the low number of dentinal tubules in gingival margins and the higher organic content of the dentin [33]. In the present study, the only statistically significant differences were obtained in the SB-applied groups without acid etching (Groups 3 and 5). Although the differences did not reach statistical significance, the SE and SB groups showed higher microleakage in the gingival wall than in the occlusal wall, in accordance with the previous studies. In contrast, the S3 bond group showed less microleakage in the gingival wall than in the occlusal wall. The reason for this finding may be related to the low acidic pH of C3S Bond (pH=2.7), which is not enough to properly etch enamel in order to achieve a durable bond to enamel.
Dentin surfaces irradiated with the Er:YAG laser are free of a smear layer and the dentinal tubules are open. This irregular morphology of the surface is believed to provide suitable surfaces for good adhesion in the resin–dentin interface [34]. However, most investigators believe that erbium laser irradiation does not replace the etching step [2,14,35,36]. In the present study, laser etching and acid etching were compared in SB2-applied groups in terms of microleakage. The highest microleakage in occlusal walls was obtained in the non-etched group while the other groups with either laser etching or acid etching showed no or very little microleakage. In the gingival walls, the acid-etched groups prepared either by bur or by laser showed significantly lower microleakage than laser etched or non-etched groups. Similar results were reported by Hossain M et al., who compared the microleakage values of a laser-prepared + laser-etched group with a bur-prepared + acid-etched group and reported no significant differences between them [37]. Chinelatti MA et al., investigated the amount of microleakage on Class V cavities prepared by bur or by laser using varying etching procedures and found that bur + acid-etched group demonstrated the lowest microleakage at all margins evaluated. Their results proves the negative effects of Er:YAG laser in cavity preparation and surface irradiation procedures in terms of microleakage [15].
Limitation
The major limitation of the present in vitro study is that the results may not directly be extrapolated to the clinical practice due to the complex nature of the oral environment. Therefore, long term clinical studies are required to verify the results of in vitro studies. In view of the fact that interaction pattern of the adhesive systems with the lased substrate can differ from those with the conventional ones and particular characteristics of the adhesive systems have a strong influence on the success of the resin–dentin bond, more research should be planned to develop bonding agents that are specifically designed for laser-prepared cavities.
Conclusion
On the basis of these results, the only significant difference between the cavity preparation methods was observed with regard to the Adper SE bond group. It is important to note that the other adhesive systems (C3S Bond and SB2) showed more leakage in laser-prepared cavities than in bur-prepared cavities, though it was not statistically significant. On the contrary, Adper SE Bond showed significantly better results in laser-prepared cavities. Therefore, the present study suggests that some adhesive system, even when showing unfavourable results with conventional preparations, may exhibit favourable results in lased cavities.
Post-Hoc Mann-Whitney U-test was used to compare two groups. Wilcoxon Signed test was used to compare occlusal and gingival microleakage.
O. Occlusal; G. Gingival; Bold numbers indicate significant differences (p<0.05)