Modification of 99mTc-Thyroid Scan Protocol to Decrease the Radiation Dose to Salivary Glands
Wadah M Ali1, Entesar Zawam Dalah2, Lana Haider Ahmed3, Mohamed M Abuzaid4
1 Assistant Professor, Department of Medical Imaging, Gulf Medical University, Ajman, NAEMIA, United Arab Emirates.
2 Assistant Professor, Department of Medical Imaging, University of Sharjah, Sharjah, United Arab Emirates.
3 Assistant Professor, Department of Medical Imaging, National Ribat University, Khartoum, Sudan.
4 Assistant Professor, Department of Medical Imaging, University of Sharjah, Sharjah, United Arab Emirates.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Wadah M Ali, Assistant Professor, Department of Medical Imaging, College of Health Science, Gulf Medical University, University Street, Al Jurrf 1, Ajman, United Arab Emirates.
E-mail: dr.wadah@gmu.ac.ae
Introduction
The pharmacokinetics pathway of 99mTc-pertechnetate, used for thyroid scan, involves the absorption of 99mTc in normal non-pathologic salivary glands.
Aim
To modify the original protocol used for thyroid scan for the purpose of decreasing radiation absorbed dose to the non-target organ (Salivary Gland).
Materials and Methods
Thirty-one patients of different age, gender, and thyroid function test were gathered and analysed. Each patient underwent two thyroid scans, the first scan was performed using the original protocol, and the second scan was obtained with a slight modification to the original protocol, where patients were instructed to chew a gum and drink a cup of water before acquiring the second thyroid scan which started 5 minutes after completing the original scan protocol. Images of the original and modified protocols were acquired using SPECT scanner with the first scan starting 20 minutes post 99mTc injection. Patients lay in a supine position while undergoing both scans. The percentage of the net administrated activity localised in the thyroid and salivary glands were measured.
Results
The absorbed dose values of the thyroid and salivary glands from the original protocol and the modified protocol were estimated. There was no significant change in the thyroid gland absorbed dose between the two protocols, whereas a significant (t-test) reduction was found in the salivary gland absorbed dose using the original and modified protocol with values being 0.77±0.39 mGy and 0.28±0.09, respectively.
Conclusion
Using the modified chew gum phase protocol in imaging of the thyroid gland with 99mTc result in decreasing the absorbed dose of the salivary gland.
Absorbed doses, Effective doses, Single photon emission computed tomography, Thyroid glands, 99mTc-pertechnetate
Introduction
The functions of salivary gland are aiding in food digestion, protecting oral mucosa, facilitating re-mineralisation, and moistening the palate for articulation [1]. It is well known that all of 99mTc-pertechnetate or 123I-iodide radiopharmaceuticals are the choice for nuclear medicine thyroid gland imaging [2], where both provide excellent quality image. While 123I is more expensive than 99mTc, the latter is known to have lower patient dose [3,4] given the reasonably short physical half-life (6 hours), short retention in the thyroid gland, and the no emission of betaparticles provides low dosimetry to the thyroid gland (10,000 times less than that of 131I-iodide) as well as to the whole-body. Further, the gamma photon energy emitted by 99mTc (140 keV) is ideal for scintillation cameras, it is readily available, and is cost efficient [5]. The characteristic similarity between the iodide and pertechnetate ions explains the uptake of 99mTc-pertechnetate in the thyroid gland [2,6]. Although the thyroid does not organify 99mTc-pertechnetate, in the majority of cases the uptake and imaging data provide all the information needed for accurate diagnosis [7]. In rare instances, 123I-iodide can subsequently be used for assessment of organification defects [7]. The radionuclide used in nuclear medicine imaging should provide a high target to non-target ratio, otherwise differentiating pathology from background become a challenge despite the unnecessary radiation dose to the non-target organs. Thus, when performing a thyroid scan, ideally all the radioactivity should be in the thyroid gland and nowhere else in the neck region [8]. In an image of thyroid gland with 99mTc-pertechnetate, the radioisotope definitely appears in the salivary gland, leading to difficulties in thyroid image interpretation [4]. A 99mTc-pertechnetate is loosely bound to protein following Intravenous (IV) injection that rapidly leaves the plasma, about more than half the 99mTc-pertechnetate leaves plasma within minutes, then distributes in the extracellular fluid and rapidly concentrate in salivary glands, choroid plexus, thyroid gland, gastric mucosa, functioning breast tissue and cross placenta during pregnancy [4]. 99mTc is secreted through the kidney, and in special situation can be excreted through saliva, gastric juice, milk and sweat [9]. Thyroid gland, stomach wall, small intestine, upper and lower intestinal wall and urinary bladder are the most exposed tissues undergoing 99mTc-pertechnetate thyroid gland scan [10]. A trace of activity will still be present in the salivary glands two hours post IV injection, with peak concentration shown in the first 5 to 10 minutes [4]. Stimulation of the salivary gland with lemon juice results in rapid reduction of activity [4]. Radiation exposure from 99mTc used for thyroid scan depends on thyroid status and presence of blocking agent [10]. International Committee of Radiation Protection (ICRP-1987) reported that the effective dose to the salivary gland during thyroid scan using 99mTc is 0.013 mSv/MBq, so for use of 75 and 40 MBq of 99mTc for thyroid scan, the effective dose to salivary gland will be 1.0 and 0.5 mSv respectively. Absorbed dose without blocking agent after administering 75 MBq of 99mTc is 1.7 mGy [10]. The aim of this work was to explore the feasibility of decreasing unnecessary radiation dose to the salivary glands during a 99mTc-pertechnetate thyroid scan while maintaining image quality. Radionuclide concentration in salivary glands and the mean absorbed and effective doses were estimated.
Materials and Methods
The present analytical descriptive study was carried out in nuclear medicine department of Khartoum Center for Oncology and Nuclear Medicine. The study includes 31 patients examined for thyroid scan during the period from January to December 2017. The study was approved by ethical research committee of National Ribat University with collaboration of Khartoum Center for Oncology and Nuclear Medicine, and all patients were informed about the study and consent form was signed before the study. Patients with thyroid goiter (toxic) and patient under radiotherapy and/or chemotherapy were excluded from this study. The gamma camera used in this study is Nucline Sprit (DHV) gamma camera, DH.004167-V0 SPECT system dual head fitted with Low Energy High Resolution (LEHR) collimator, manufactured in 2010 in Hungary, 230V/50Hz (2400VA). The software used in this camera is camera manufactured system software (MEDISO Interview XP), this camera was subject to the daily as well as routine Quality Control (QC) tests in regular bases. The radionuclide used in the study was 99mTc-pertechnetate eluted from Pole TechNet 8.0-175 GBq radionuclide generators, the radionuclide used passed the quality control tests performed before being used with the patients (radionuclide purity and chemical purity). Specifications were within the guidelines described by monographs of the USA and the European Pharmacopoeia PH 5.0-7.0 [11-13]. Radiation dosimeter was automatically calculated using Dosisrad 1.1 software, this application was developed in the Visual Basic programming language and the dosimetric calculations are based on the values given by the International Commission on Radiological Protection (ICRP) [14]. Patients were instructed to discontinue any type of thyroid medication at least one week for neomarcezole tablets and 14 days for Thyroxin tablets, no X-ray image with iodinated contrast study at least a week prior to the time of scanning appointment, and not to eat iodine-containing foods on the scan day.
Thyroid Scanning Protocols
Each individual patient underwent two protocols as described below:
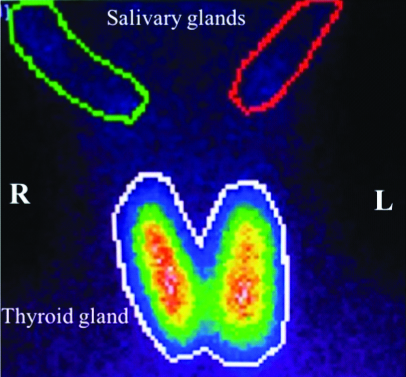
The original protocol: A dose of 74-370 MBq (2-10 mCi) of 99mTc-pertechnetate was injected depending on patient weight. Individual’s injected dose was measured according to individual’s weight and prepared in a syringe ready for injection. The syringe containing the activity was placed on the center of the gamma camera couch; 10 cm away from the gamma camera head and counted for 1 minute before injecting the patient. This was considered for Full Syringe (FS) image and data. The patient was then injected and requested to rest in the waiting room for 20 minutes prior to the first thyroid scan. Patients were recommended to hydrate well during the 20 minutes waiting period. Following injection, syringe was also counted with the same parameter for one minute and placed at 10 cm distance from the camera head to obtain what is called the Empty Syringe (ES) image and data. The subtraction value of Full Syringe and Empty Syringe (FS-ES) gives the exact net amount of activity injected to the patient. Patients lied in a supine position to acquire an anterior plane image with the thyroid gland in the center of the Field of View (FOV) of the gamma camera, and at 10 cm distance between patient head and the camera. The thyroid scan was acquired for one minute to obtain the uptake in the thyroid gland. Using MEDISO Interview XP software, a Region of Interest (ROI) was drown around the thyroid gland and around the salivary gland to calculate the counts statistics presented in each region [Table/Fig-1]. After completing the first thyroid scan protocol performed using the original protocol, individuals involved in this study underwent a second thyroid scan following a modified protocol.
Shows the ROI around the thyroid gland and salivary glands in an anterior view image.
The modified protocol: Individuals were instructed to chew a gum to stimulate the salivary gland, and swallow their saliva. They were then instructed to drink a cup of water after 5 minutes after the chewing gum phase. Patient setup of the original thyroid scan protocol described above was followed, and ROIs were drawn around the thyroid and salivary glands to measure the counts in each region. Counts in the salivary glands obtained using the modified protocol was then compared to counts defined following the original protocol, reflecting the impact of chewing gum phase.
Statistical Analysis
The mean and Standard Deviation (SD) of the net injected dose (FS-ES) across the cohort samples, and the mean and SD counts in the ROIs delineated around the thyroid gland and salivary glands were calculated and measured. Pearson coefficient test was used to correlate the mean salivary absorbed dose using the original and modified protocol. t-test was used to study statistical differences between thyroid and saliva glands absorbed and effective doses using the original and modified protocol. Further, t-test was used for significant uptake differences between right and left side salivary glands.
Results
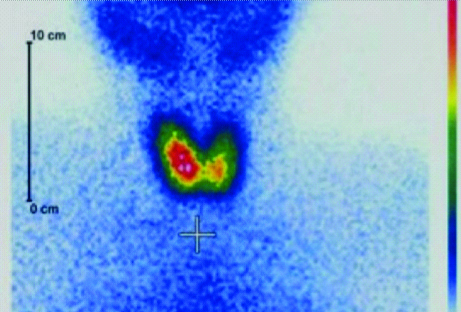
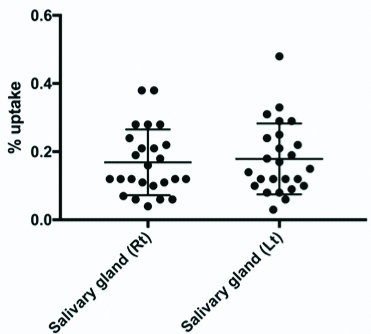
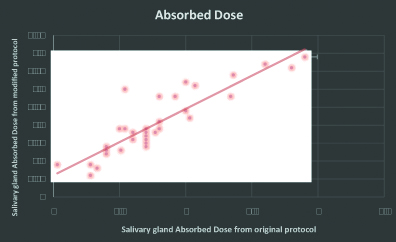
The gender, age, weight, height and the history of each patient was recorded. The peak incidence was among 24-35 year old; out of which 5 (16%) were male and 26 (84%) were female. The average age of the patients studied was 40 year old ranging from (20-80). The acquired anterior image of the thyroid gland and salivary glands showed abundance of accumulated activity in the thyroid gland and salivary glands with only minimal uptake in the neck region [Table/Fig-2]. In average, the percentage of activity trapped in the thyroid gland was 1.69% of the total dose administrated with only 0.30% being trapped in the salivary gland, implying that salivary glands receive ~20% of the dose absorbed by the thyroid gland resulting in an uptake concentration ratio of 5:1 between the thyroid (targeted) and salivary glands (non-targeted). The mean and (range) of thyroid gland absorbed dose across all 31 patients using the original and modified thyroid scan protocol were 1.90 mGy ranging from (3.95 to 1.15), and 1.89 mGy ranging from (3.94 to 1.15), respectively [Table/Fig-3a]. The mean and (range) of salivary glands absorbed dose across all 31 patients using the original and modified thyroid scan protocol were 0.77 mGy ranging from (0.95 to 0.7) and 0.28 mGy ranging from (0.70 to 0.35), respectively [Table/Fig-3b]. Making use of t-test, no significant differences were found between thyroid gland absorbed dose using the original and modified protocols. In contrast, a statistically significant difference was found in the salivary glands using the original and modified protocols, with the latter protocol demonstrating significantly lower dose to the salivary glands. No statistical differences were found between the uptake in Right (Rt) and Left (Lt) salivary glands using t-test [Table/Fig-4]. Using a Pearson coefficient test, a significant correlation was obtained between mean salivary absorbed dose using the original and modified protocol [Table/Fig-5].
Demonstrate uptake in the thyroid and salivary glands using the modified protocol.
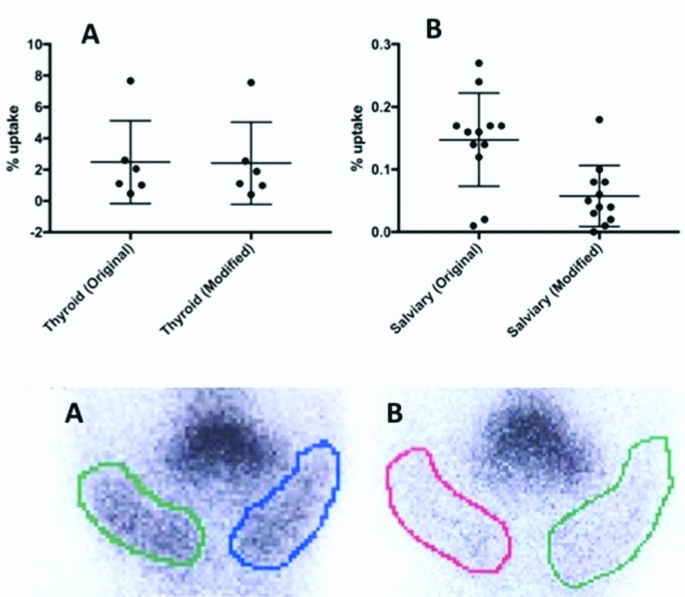
Demonstrate the mean (and range) of uptake in the thyroid (top panel, A) and salivary glands (top panel, B) using the original and modified protocol. The bottom panel demonstrate radionuclide uptake in the salivary glands (left and right sides) using the original (bottom panel, A) and modified (bottom panel, B) protocol.
Shows the uptake in the Right (Rt) and Left (Lt) salivary glands with no significant differences in the uptake between the two gland sides.
Correlation between salivary glands absorbed dose using the original and modified protocol.
Discussion
Salivary gland trapped 20% of the total activity dose administrated, even with this percentage the image is considered as high quality image [15], but the concern of radiation risk cannot be insignificant [16,17]. The result of salivary gland absorbed dose before and after the implementation of Chew protocols showed variation in the result between the two protocols used, this indicated that the modified chew protocol have a direct effect in decreasing the salivary gland absorbed dose and have no direct effect in the thyroid gland, so the modified protocols help in reducing the salivary gland absorbed dose while maintaining same image quality as of the original protocol. A significant correlation was obtained between mean salivary absorbed dose using the original and modified protocol. This minimal dose reduction observed in the thyroid gland using the two protocols can be referred to the proximity in time between the two scans (5 minutes) as the first thyroid image obtained using the original protocol was performed 20 minutes after administration of radioactivity whereas the second thyroid image obtained using the modified protocol was acquired 25 minutes after administration of radionuclide.
In contrast, the mean effective dose in the salivary glands was shown to be significantly different in the two protocols. With the mean effective dose ±SD using the original protocol to that using the modified protocol were 0.67±0.17 mSv and 0.23±0.06 mSv, respectively. The finding demonstrates the impact of gum chewing phase on the salivary glands (non-targeted organ) uptake for patients undergoing thyroid scan. The mean percentage of the total activity administrated to the patient and that absorbed by the salivary gland in term of mean absorbed dose and the mean effective dose to salivary gland were significantly reduced by 80%, 63.6%, and 68.7% from their original values respectively. There is a significant association between risk for both Malignant Salivary-gland Tumours (MSTs) and Benign Salivary-gland Tumours (BSTs) and gamma radiations. Several other case control studies have reported that various malignancies have a higher number of gamma radiation induced chromatid breaks per cell [17-19]. Herein, we demonstrate an absorbed and effective dose reduction to the salivary glands (non-targeted organ) while maintaining same image quality using chewing gum phase.
Conclusion
A 99mTc Pertechnetate is the most widely used radioisotopes in imaging of the thyroid gland. The implementation of the modified Chew protocols will help in reducing the absorbed dose of non-target organ especially the salivary gland and reduce the probability of radiation induced salivary gland cancer.
Limitation
Small sample size is one of the limitations of the study. Further studies with larger sample size are recommended.
[1]. Thienpont LM, Beastall G, Christofides ND, Faix JD, Ieiri T, Jarrige V, Proposal of a candidate international conventional reference measurement procedure for free thyroxine in serumClin Chem Lab Med 2007 45(7):934-36.10.1515/CCLM.2007.15517617044 [Google Scholar] [CrossRef] [PubMed]
[2]. Smith JJ, Croft BY, Brookeman VA, Teates CD, Estimation of 24-hour thyroid uptake of I-131 sodium iodide using a 5-minute uptake of technetium-99m pertechnetateClin Nucl Med 1990 15(2):80-83.10.1097/00003072-199002000-000032155732 [Google Scholar] [CrossRef] [PubMed]
[3]. Andros G, Harper PV, Lathrop KA, McCardle RJ, Pertechnetate-99m localization in man with applications to thyroid scanning and the study of thyroid physiologyJ Clin Endocrinol Metab 1965 25:1067-76.10.1210/jcem-25-8-106714328377 [Google Scholar] [CrossRef] [PubMed]
[4]. Mettler FA, Milton JR, Guiberteau J, Essentials of Nuclear Medicine Imaging 2012 6th EditionPhiladelphia PASaunders Elsevier:607 [Google Scholar]
[5]. Sucupira MS, Camargo EE, Nickoloff EL, Alderson PO, Wagner HN, The role of 99mTc pertechnetate uptake in the evaluation of thyroid functionInt J Nucl Med Biol 1983 10(1):29-33.10.1016/0047-0740(83)90030-X [Google Scholar] [CrossRef]
[6]. Sodee DB, The study of thyroid physiology utilizing intravenous sodium pertechnetateJ Nucl Med 1966 7:564-67. [Google Scholar]
[7]. Pesce L, Kopp P, Iodide transport: implications for health and diseaseInt J Pediatr Endocrinol 2014 2014(1):810.1186/1687-9856-2014-825009573 [Google Scholar] [CrossRef] [PubMed]
[8]. Henkin RE, Bova D, Dillehay G, Karesh SM, Halama J, Wagner R, Nuclear MedicineJournal of Nuclear Medicine 2007 48(5):846-46.10.2967/jnumed.107.040329 [Google Scholar] [CrossRef]
[9]. Hays MT, 99mTc-pertechnetate in man: absorption after subcutaneous and oral administration into saliva and gastric juiceJ Nucl Med 1973 14(6):331-35. [Google Scholar]
[10]. IiseZolleTc-99m Radiopharmaceuticals preparation and quality control in nuclear medicine 2007 HeidelberagSpringer Berlin:7-46. [Google Scholar]
[11]. Little JB, Radiation carcinogenesisCarcinogenesis 2000 21:397-404.10.1093/carcin/21.3.39710688860 [Google Scholar] [CrossRef] [PubMed]
[12]. Atcher RW, USP Public Standards for Compounded Sterile Radiopharmaceuticals: Recommendations from SNMMI 2017 [Google Scholar]
[13]. Verbruggen A, Coenen H, Deverre JR, Guilloteau D, Langstrom B, Salvadori PA, Guideline to regulations for radiopharmaceuticals in early phase clinical trials in the EUEur J Nucl Med Mol Imaging 2008 35(11):2144-51.10.1007/s00259-008-0853-718690437 [Google Scholar] [CrossRef] [PubMed]
[14]. Gómez Perales JL, Mendoza AG, Freeware for reporting radiation dosimetry following the administration of radiopharmaceuticalsApplied Radiation and Isotopes 2015 103:131-34.10.1016/j.apradiso.2015.06.00126092354 [Google Scholar] [CrossRef] [PubMed]
[15]. Cherry SR, Sorenson JA, Phelps ME, Physics in Nuclear Medicine 2012 4th Edition:78-112. [Google Scholar]
[16]. ICRP, 2012. Compendium of Dose Coefficients based on ICRP Publication 60. ICRP Publication 119. Ann. ICRP 41(Suppl.)10.1016/j.icrp.2012.06.03823025851 [Google Scholar] [CrossRef] [PubMed]
[17]. Bondy ML, Wang LE, El-Zein R, Andrade MD, Selvan MS, Bruner JM, Gamma-radiation sensitivity and risk of gliomaJ Natl Cancer Inst 2001 93(20):1553-57.10.1093/jnci/93.20.155311604478 [Google Scholar] [CrossRef] [PubMed]
[18]. Bondy ML, Kyritsis AP, Gu J, de Andrade M, Cunningham J, Levin VA, Mutagen sensitivity and risk of gliomas: a case-control analysisCancer Res 1996 56(7):1484-86. [Google Scholar]
[19]. Buchholz TA, Wu X, Radiation-induced chromatid breaks as a predictor of breast cancer riskInt J Radiat Oncol Biol Phys 2001 49(2):533-37.10.1016/S0360-3016(00)01502-9 [Google Scholar] [CrossRef]