Gall bladder is one of the organs having a wide spectrum of diseases ranging from congenital anomalies, calculi and its complications, non-inflammatory and inflammatory lesions to neoplastic lesions [1]. The non-neoplastic lesions mainly include cholelithiasis, cholecystitis, xanthogranulomatous cholecystitis and cholesterolosis. Neoplastic category includes adenoma, carcinoma and mesenchymal tumours. The management of neoplastic and non-neoplastic lesions vary considerably with gall bladder carcinoma having very poor prognosis. Since clinical presentation of both is very similar, it is essential to diagnose gall bladder malignancy early on histopathology. Even seemingly benign chronic cholecystitis may be associated with hyperplasia and metaplasia of gall bladder epithelium and it has been suggested that metaplastic epithelium is more susceptible to malignant transformation than the normal mucosa and the intestinal metaplasia-dysplasia carcinoma sequence exists in the gall bladder [2,3].
In India gall bladder cancer is most prevalent in northern and north-eastern states of Uttar Pradesh, Bihar, Orissa, West Bengal and Assam. In fact northern India has one of the highest reported incidence of gall bladder cancer in the world (22/100,000 population) [4]. So it is essential to detect metaplastic lesions of gall bladder early and mucin histochemistry study may be useful in this regard. Ganesh IM et al., observed in their study that in normal gall bladder mucosa neutral mucins are predominant whereas in metaplastic epithelium there was significant increase in acidic mucin [5]. But Sood S et al., and Gupta SC et al., suggested in their study that metaplastic lesions showed predominantly neutral mucin [1,6]. So there are contrasting findings in different studies and hence more studies are required to arrive at a definite conclusion. All these reasons made us undertake the study. Our study was performed to find out if mucin histochemistry can be a useful tool for early detection of metaplastic lesions of gall bladder.
Materials and Methods
An observational study from February 2017 to January 2018 was conducted after approval by Institutional Ethics Committee of R.G. Kar Medical College and Hospital. A total of 1153 cholecystectomy specimen received in histopathology section of Pathology Department of RG Kar medical college were analysed. After gross examination, tissues were subjected to formalin fixation, routine processing and paraffin embedding. For cases without any gross abnormality, standard three sections including fundus, body and neck were taken. In cases with any growth, irregular mucosa, thickened wall, etc., more sections were taken. Five microns thick sections on three to four slides were prepared from each specimen. Apart from routine Haematoxylin and Eosin (H&E) stain, special stains like PAS and AB were used. Gross and microscopic features of all cases were studied in detail.
Inclusion Criteria: All the cases of histopathologically confirmed inflammatory lesions of gall bladder including those showing metaplastic changes of the gall bladder epithelium, irrespective of age and sex.
Exclusion Criteria: All the cases of histopathologically confirmed malignant gallbladder lesions were excluded from this study.
Results
Age of the patient in the present study ranged from 12-86 years and most common age group with maximum gall bladder diseases was in 5th decade i.e., 25.8% (297/1153) cases. Female to Male ratio was 3.5:1 [Table/Fig-1].
Age wise distribution of non-neoplastic gall bladder disease cases.
| Age Group (in years) | Number of cases | Percentage (%) |
|---|
| 11-20 | 30 | 2.6% |
| 21-30 | 282 | 24.5% |
| 31-40 | 276 | 23.9% |
| 41-50 | 297 | 25.8% |
| 51-60 | 164 | 14.2% |
| 61-70 | 82 | 7.1% |
| 71-80 | 15 | 1.3% |
| 81-90 | 7 | 0.6% |
Maximum cases were of chronic cholecystitis 72.9% (841/1153) cases followed by acute cholecystitis 3.8% (44/1153) cases, Xanthogranulomatous cholecystitis 3.3% (38/1153) cases, chronic cholecystitis with cholesterolosis 2.5% (29/1153) cases. Metaplastic lesions in gall bladder were found in 17.4% (201/1153) cases. Out of the metaplastic lesions 85.2% (171/201) cases was of pyloric metaplasia, 10.4 % (21/201) cases were of intestinal metaplasia and both pyloric and intestinal metaplasia was found in 4.4% (9/201) cases [Table/Fig-2].
Distribution of non-neoplastic gall bladder disease cases according to histomorphological spectrum.
| Histomorphological Spectrum | No. of cases | Percentage |
|---|
| Chronic Cholecystitis | 841 | 72.9% |
| Acute Cholecystitis | 44 | 3.8% |
| Chronic cholecystitis with cholesterolosis | 29 | 2.5% |
| Xanthogranulomatous Cholecystitis | 38 | 3.3% |
| Metaplasia | Pyloric | 171 | 14.8% |
| Intestinal | 21 | 1.8% |
| Pyloric+Intestinal | 9 | 0.8% |
Gall stones were present in 74.8% (862/1153) cases and 25.2% (291/1153) cases had no gall stones. Most common histomorphological lesion associated with gall stones was metaplastic lesions 85.2% (171/201) followed by chronic cholecystitis 73.4% (617/841 cases) and acute cholecystitis 66.6% (29/44) cases.
Gall stones observed in maximum number of cases were multiple stones accounting for 72.9% (628/862) cases and solitary stones in 27.1% (234/862) cases. Out of 862 gall stone cases, gall stones were more common in females 75.4% (650/862) cases than in males 24.6% (212/862) cases respectively.
Length of normal gall bladder is 7-10 cm. In this study 35.4% (408/1153) cases were of normal length while 64.6% (745/1153) were less than 7 cm. Wall thickness of normal gall bladder is less than 3 mm. In this study 21.9% (252/1153) cases had normal wall thickness while 78.1% (901/1153) cases had increased wall thickness (>3mm).
Out of 1153 cholecystectomy specimens, mucin histochemistry was done in 201cases showing metaplasia. In metaplastic group, only PAS positivity (alcian blue negative) was seen in 25.9% (52/201) cases and alcian blue positivity was seen in 74.1% (149/201) cases. Thus, metaplastic lesions showed predominantly acidic mucin [Table/Fig-3].
Type of mucin in cases of chronic cholecystitis with metaplasia.
| PAS | Alcian Blue | No. of cases | Predominant mucin |
|---|
| + | - | 52 | Neutral |
| + | + | 122 | Acidic (simple non sulphated) |
| - | + | 27 | Acidic (complex sulphated) |
In this study, all the cases of inflammatory lesions of gall bladder without metaplasia of the epithelium showed predominance of neutral mucin followed by acid mucin. The histochemical analysis of mucin showed definite altered expression in the diseased gall bladder epithelium in terms of amount and type of mucin expressed in cases of metaplasia. The acid mucin content increased while neutral mucin content decreased in cases of metaplasia [Table/Fig-4,5,6,7,8,9,10 and 11].
HPE of chronic cholecystitis with cholesterolosis (H&E stain, 10X).
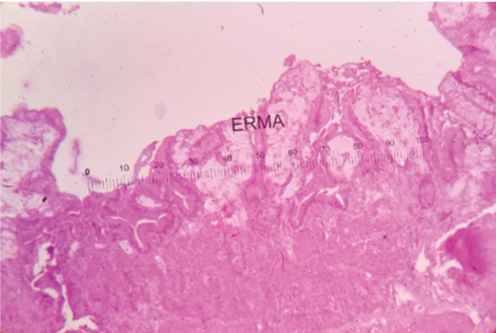
HPE of xanthogranulomatous cholecystitis (H&E stain, 40X).
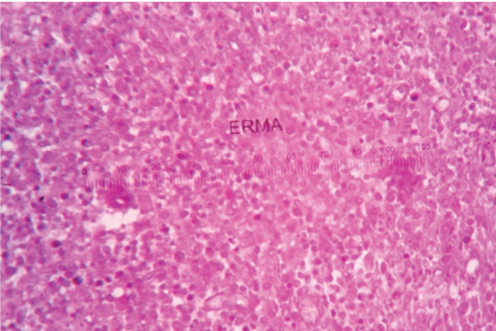
HPE of pyloric metaplasia of gall bladder mucosa (H&E stain, 40X).
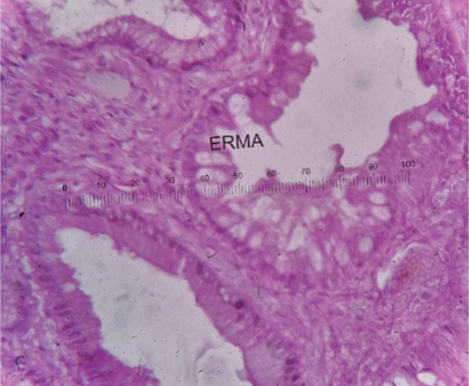
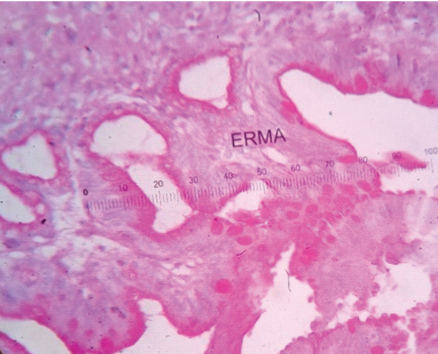
HPE of intestinal metaplasia of gall bladder mucosa (H&E stain, 10X)
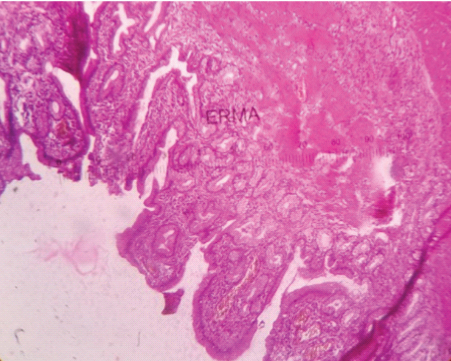
PAS positive mucin in glands in pyloric metaplasia of gall bladder (PAS stain, 10X).
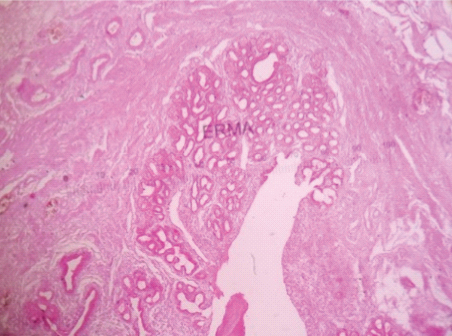
PAS positive mucin in glands in pyloric metaplasia of gall bladder (PAS stain, 40X).
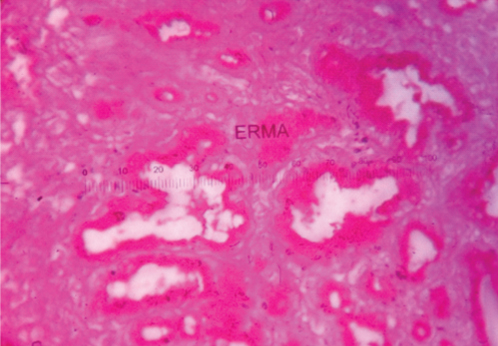
Alcian Blue positive mucin in glands of pyloric metaplasia of gall bladder (Alcian Blue stain, 40X).
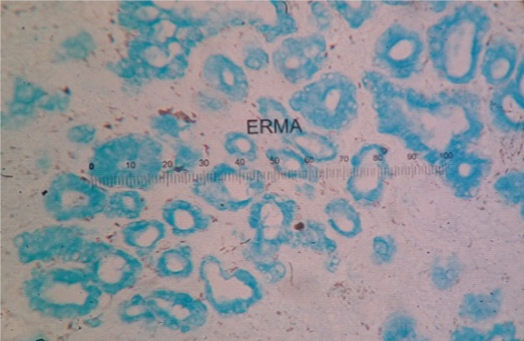
PAS positive mucin in glands of intestinal metaplasia of gall bladder (PAS stain, 40X).
Discussion
Cholecystitis is defined as inflammation of the gall bladder. The most common cause of cholecystitis is cystic duct obstruction by gall stone [7]. Cholelithiasis produces diverse changes in the gall bladder mucosa namely acute inflammation, chronic inflammation, granulomatous inflammation, cholesterolosis and glandular metaplasia [8]. Injury to the mucosal columnar epithelium by gall stones can cause changes like metaplasia, dysplasia and neoplasia [6].
In present study, age of the patients ranged from 12-86 years. Maximum cases (25.8%) of gall bladder disease were seen in fifthdecade of life, followed by (24.5%) in third decade and (23.9%) in fourth decade. The mean age in the present study was 40.3 years where as in a study by Banerjee A et al., it was observed to be 39.3 years [9]. Male to Female ratio was 1:3.5. This was in accordance with various studies where M:F ratio ranged from 1:3.2 to 1:6 [10].
In the present study on histopathological examination of gallbladder lesions, maximum cases were of chronic cholecystitis 72.9% (841/1153) followed by acute cholecystitis 3.8% (44/1153 cases). These observations were almost like Selvi TR et al., who reported chronic cholecystitis cases to be 85.8% [11].
After chronic cholecystitis, acute cholecystitis cases accounted for 3.8% which was in concordance with the study by Kaur A et al., who showed it to be 2.60% cases [12], while another study by Vahini G et al., and Terada T showed 18.3% and 1.5% cases of acute cholecystitis which contrasted with present study [13,14]. This might be due to large sample size in this study. In this study, cases of cholesterolosis were 2.5% (29/1153) which was like study by Vahini G et al., who reported it to be 1.8% cases [13]. In a study by Kaur A et al., much higher percentage of cases of cholesterolosis (12.25%) was found [12]. This may be due to different dietary habits and different religions followed by the study participants.
In this study, 3.3% (38/1153) cases of xanthogranulomatous cholecystitis were found. In a study by Franco V et al., the retrospectively estimated frequency of xanthogranulomatous cholecystitis varied from 0.7 to 5.2% in surgically resected gallbladders. There was a female preponderance in xanthogranulomatous cholecystitis [15,16].
Xanthogranulomatous cholecystitis is always misdiagnosed grossly as malignant due to the appearances and thickness of the wall. Studies have been carried out to see the association or risk of developing carcinoma in them. In a study by Rao RV et al., they observed that xanthogranulomatous cholecystitis was neither associated nor does it increase risk of developing into carcinoma gallbladder [17].
In this study, chronic cholecystitis cases were mostly associated with cholelithiasis 73.4% (617/841) cases. Similar findings were noted by Baig SJ et al., Mathur SK et al., and Mohan H et al., in their studies [10,18,19]. Also, maximum cases were associated with multiple stones i.e., 78.3% (483/617) cases.
In present study, non-neoplastic lesions of gall bladder were found to be 72.9% (628/862) cases with multiple stones. Similar study was also conducted by Goyal S et al., which also observed that multiple stones are far more common than single stone accounting for 72% casesofnon-neoplastic lesions with stones, which was in concordance with our study [20].
In this study, out of 201 cases of chronic cholecystitis with metaplasia 171 cases (85.18%) were associated with gallstones which were mainly of solitary type 60.8% (104/171) cases. This was also in concordance with other studies [19,21].
In present study of mucin histochemistry, it was found that gall bladder with cholelithiasis showed acidic mucin in supranuclear region. In deeper parts neutral mucins were found. So, present studies are comparable with similar study done by Ganesh IM et al., [5]. Increased mucin secretion by gallbladder mucosa during gallstone formation has been described in various studies [22-26]. Earlier studies have shown that mucins, in addition to being a structural component of gallstones, also play an accelerator role in lithogenesis [27].
Neutral mucins are positive for PAS stain and negative for AB stain. All acidic mucins are positive for AB stain. Some acidic mucins are positive for PAS stain also (like simple non-sulphated acidic mucins) while some acidic mucins may or may not be positive for PAS stain (like complex sulphated acidic mucins). Normal gall bladder epithelium was predominantly positive for PAS stain but negative for AB stain in our study. Thus, it indicates neutral mucins are predominantly present in normal gall bladder epithelium. Also, mucin histochemistry in metaplastic group showed neutral mucin (PAS) in 25.9% cases (52/201). 74.1% (149/201) cases showed positivity for acid mucin (AB/PAS at 2.5pH). Thus, metaplastic lesions showed predominantly acid mucin. Ganesh IM et al., observed in his study that neutral mucins were predominant in normal gastric and gall bladder mucosa [5]. On the other hand, there was a significant increase of acidic mucins in intestinal metaplasia, gastric carcinoma and stone-containing gallbladder. They suggested that the sulphated mucins (acid mucin) have a greater role in gallstone formation than the neutral mucins. Also, the acid mucins play an important role in cancer progression and metastasis. But, Gupta SC et al., reported that metaplastic lesions showed predominantly neutral mucin followed by sialomucin and sulphomucin which is in contrast to our study [6].
Conclusion
Within the constraints of small sample size, we conclude that there is a definite association between metaplasia-dysplasia-carcinoma sequence. Early detection of mucin histochemistry in metaplastic cases can lead to early diagnosis of carcinoma gallbladder as acidic mucins are more predominant than neutral mucins in cases of metaplasia and carcinoma. So, irrespective of the gross appearance and seemingly benign nature of the disease affecting the organ, every gallbladder removed by cholecystectomy should be subjected to histopathological examinations and mucin histochemistry.