Introduction
Dry Eye Disease (DED) is one of the most common problems and reasons for referring to ophthalmology clinics, which has been exacerbated by the increased use of computers and mobile phones.
Aim
The purpose of the present systemic review was to review the effects and mechanism of medicinal plants in DED treatment.
Materials and Methods
The key words “Dry eye” or “Keratoconjunctivitis sicca” or “keratitis sicca” in combination with “medicinal plant”, “herb”, and “phyto” were used to conduct the review. Clinical and experimental published articles in English language between 2000-2018 were retrieved from databases including the Institute for Scientific Information (ISI), PubMed and Scopus.
Results
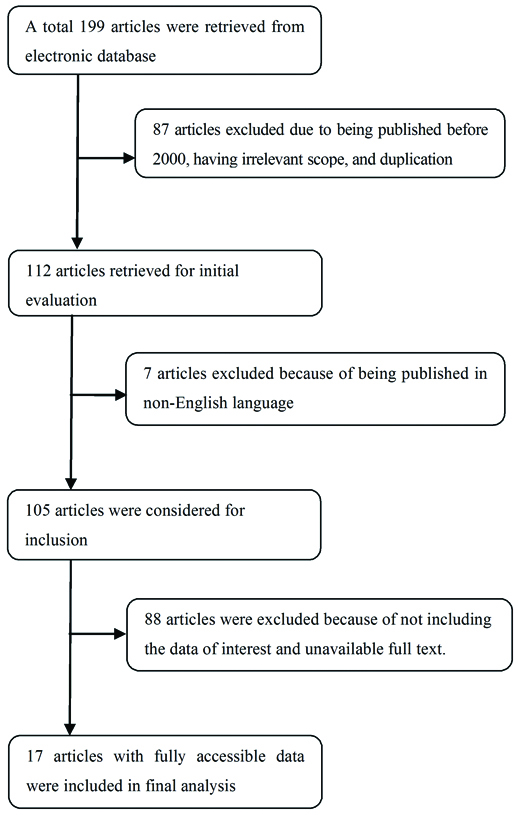
Totally, 199 articles were retrieved from the electronic database and finally 17 articles were included in the final analysis. Various plants such as Buddleja officinalis, Aristotelia chilensis, Prunus armeniaca, Hippophae rhamnoides, Lycium barbarum, and Rhynchosia volubilis Lour are effective in treating DED through different mechanisms.
Herbal derivatives such as curcumin, antioxidant glasses, phytoestrogen, ferulic acid, and kaempferol can be used as food supplement independently and in some cases along with chemical drugs. Chinese herbal formulations and compounds such as ‘Chi-Ju-Di-Huang-Wan’, ‘TriphalaGhritaNetratarpan’, and ‘ShengJinRunZaoYangXue’ granules can play a role in inducing anti-oxidant and anti-inflammatory properties in the treatment of the disease.
Conclusion
Generally, medicinal plants reduce tear film stability by decreasing osmolarity and increasing tear production. Several mechanisms, including the reduction of Reactive Oxygen Species (ROS) (antioxidant activity), the prevention of cell apoptosis, the modulation of inflammatory factors, and the regulation of androgens, can affect lacrimal glands and membrane cells, thereby helping to treat DED.
Introduction
Dry eye disease or keratoconjunctivitis sicca is a multifactorial disease that is developed due to inflammation of the ocular surface and lacrimal glands, meibomian gland dysfunction, and neuropathic pain disorder [1]. Various environmental factors such as air dryness, smoke, allergens, systemic diseases, and age (in premenopausal women) contribute to DED [2-5]. The disease has increased in recent years with the advent of monitors and prolonged use of computers and is one of the most important causes of referral to ophthalmology clinics characterised by symptoms such as scratches and foreign bodies in the eye [6-8].
Due to the high prevalence of DED, it is necessary for physicians to have important information about care, often debilitating symptoms, and preventable and curable nature of the disease [5].
DED treatment is based on a tear substitute. New treatments focus on certain drugs such as secretagogues, lubricants, topical androgens, antibiotics, immunosuppressive drugs, and new anti-inflammatory drugs [9,10]. Meanwhile, herbal treatments have been used in the treatment of various diseases due to being less costly and causing fewer side effects [11-19]. Although, the use of herbal medicines has been used in many countries for the treatment of eye diseases since long time [20-23], however, little information is available on the validity of the efficacy or probable side effects of these drugs. Also, regarding the high prevalence of the disease, and considering that there is still no study in this regard, the authors aimed to investigate the effect and mechanism of medicinal plants in treating DED in the present systemic review.
Materials and Methods
The key words ‘Dry eye’ or ‘Keratoconjunctivitis sicca’ or ‘keratitis sicca’ in combination with medicinal plant, herb, and phyto were used to conduct the review. Relevant articles (from 2000 till date) were retrieved by using keywords from databases including ISI, PubMed, and Scopus with EndNote software. Then, the plants and the plant-based products that were effective on DED were selected and entered to the study according to inclusion criteria. A standard form, which included items such as year of publication, aim and the title of the study, intervention, outcome, variables, journal name, period, and number, was designed. The articles contents that were in accordance with the purpose of the study were recorded in the form and entered to the study with agreement of researchers. The articles whose full texts were not accessible, the articles published in non-English languages, and those that did not address the subject of this review were excluded after the author’s agreement was achieved. [Table/Fig-1] is the flowchart that illustrates how the articles were selected for final analysis.
Flowchart of the process of selecting and analysing the articles.
Results
The 17 studies that were included in the review and were effective on treatment on DED are tabulated in [Table/Fig-2] [24-40].
Medicinal plants, phytochemicals and plant-based compounds effective in dry eye disease [25-41].
| Medicinal plants |
|---|
| References | Plants | Study Design | Type of administration | Main effects or mechanisms |
|---|
| Peng QH et al., [24] | Buddleja officinalis | Experimental (in vivo) | Extract | Displaying androgen-like activity to keep basic tears secretory volume and tear film stability |
| Hitoe Set al., [25] | Aristotelia chilensis | Clinical pilot trial | standardised extract (MaquiBright) | Increasing tear fluid volume |
| Kim CS et al., [26] | Prunus armeniaca | Experimental (in vivo) | Kernel extract | Promoting the secretion of tear fluid and mucin |
| Kimura Y et al., [27] | Hippophae rhamnoides | Experimental (in vivo) | Pulp oil | Preserving tear secretion and inflammatory cytokines in the lacrimal gland |
| Chien KJ et al., [28] | Lycium barbarum | Experimental (in vivo) | Extract | Enhancing the tear volume and repairing the damaged ocular surface cells by exhibiting antioxidant and anti-inflammatory activity. |
| Kang SW et al., [29] | Rhynchosia volubilis Lour. | Experimental (in vivo) | Ethanol extract | Inhibiting squamous metaplasia and apoptosis in the cornea and inhibiting the expression of cytochrome c and Bax, while improving that of Bcl-2. |
| Phytochemicals |
| References | Phytocompound and plant deravities | Origin | Study Design | Main effects or mechanisms |
| Chen M et al., [30] | Curcumin | Turmeric (Curcuma longa) | Experimental (in vivo) | Protecting against hyperosmoticity-induced IL-1b elevation in human corneal epithelial cell via the MAPK pathways and producing anti-inflammatory effect |
| Scuderi G et al., [31] | Lacrisek (phytoestrogen compound) | a-lipoic acid (100 mg), eicosapentaenoic acid (240 mg), and extract of fenugreek (200 mg), which contains diosgenin (1.3%), steroidal saponins (50%), and alkaloids | Randomised clinical trial | Decreasing tear osmolarity and significantly increasing tear production |
| Choi W et al., Choi W et al., and Huang JY et al., [32-34] | Antioxidant glasses | Extract of Schizonepetatenuifolia var. japonica Kitagawa, Angelica dahurica Bentham ET hooker, RehmanniaglutinosaLiboschitz var. purpurea Makino, and Cassia tora L extracts/Cassiae semen and Ophiopogonisjaponicus | Randomised Clinical trial/Experimental (in vivo) | Exerting preventive effect on membrane damage, and inflammation in DED byimprovingall clinical parameters, IL-1β, IL-6, TNF-α, and IFN-γ levels, percentage of CD4+CXCR3+ T cells, goblet cell density, number of 4-HNE-positive cells, and extracellular reactive oxygen species (ROS) production |
| Chen HC et al., [35] | Ferulic acid and kaempferol | - | Experimental (in vitro) | Ferulic acid and kaempferollower IL-1B, IL-6, IL-8, and TNFα expression and produce antioxidant effect |
| Plant-based compounds |
| References | Plant-based compounds | Compounds | Study Design | Main effects or mechanisms |
| Peng QH et al., [36] | Chi-Ju-Di-Huang-Wan | Chi-Ju-Di-Huang-Wan’ is the formula ‘Liu Wei Di Huang Wan’ adds Fructus Lycii and Hos Chrysanthemi | Experimental (in vivo) | Reduce abnormalities in the corneal epithelium and be used as an artificial tear |
| Chen YL et al., [37]. | Chinese herbal treatment | Ji-Sheng-Shen-Qi-Wan, Well Zhen Zi, Dan shen, Gou Qi Zi, Well Zhen Zi, Gu Jing, Hang Bai Ju, and HuaiNiu Xi | Case-report | Effects on estrogen receptors and hormonal regulation |
| Sawant DP et al., [38]. | Triphala Ghrita Netratarpan | Phyllanthus emblica, Terminalia chebula, Terminalia bellirica in ghee base | Clinical trial | Reduced the symptoms of the computer vision syndrome |
| Hu W et al., [39]. | Sheng Jin Run ZaoYang Xue granules | Beishashen, Dihuang, Maidong, Shihu, Heshouwu, Gouqi, Juhua, Guizhencao, Ziwan and Zhigancao | Clinical trial | Treat DED, which is one of the symptoms of Sjögren’s syndrome |
| Wu SQ et al., [40] | Chinese medicinal plants | Qiwei Baizhu powder, Zhibai Dihuang decoction, Liuwei Dihuang decoction, and kidney-Qi pills | Clinical trial | Reduce levels of interleukin (IL)-1, IL-8, and TNFα and to improve eye dryness |
Discussion
Dry eye disease is a multifactorial disease. Hence, the treatment must be based on underlying cause of the disease. Medicinal plants and their derivatives can impact DED in several ways. Medicinal plants improve tear film stability by decreasing osmolality and inflammation increasing tear production. Tear hyperosmolarity has been reported to begin dry eye inflammation through the activation of epithelial and stromal cells on the ocular surface, which increase the presence of pro-inflammatory cytokines [41]. Inflammation can cause dysfunction in the lacrimal glands cells responsible for tear secretion or retention and leads to ocular surface disease. Plant compounds can be used to regulate levels of gonadal hormones by exhibiting androgen-like activities. As a result, it can inhibit topical inflammation in the lacrimal glands and reduce cell apoptosis [41,42]. In addition, plants and their compounds can contribute to the treatment process by inhibiting the expression of pro-inflammatory cytokines (IL-1 beta, IL-6, and TNFα) and reducing nitric oxide production [30,43,44]. Artificial tears are the most common treatment for eye diseases therapy; they provide temporary symptomatic relief; however, are unable inhabitation of inflammatory cytokines activity [34]. Also, reduction of ROS (antioxidant activity) and the prevention of cell apoptosis can reduce the symptom of DED. A 8-Hydroxy-2 deoxyguanosine (8-OHdG), 4-Hydroxynonenal (HNE) and Malondialdehyde (MDD) are major biomarkers that cause oxidative damage in ocular surface [41,45]. In DED patients, the levels of these in the tear and conjunctiva increase and cause disruption in the normal functioning of the eye and finally leads to oxidative damage, possibly inflammation and apoptosis [41]. Anti-apoptotic activity and increased expression of some genes in the lacrimal gland epithelial cells represent one of the mechanisms in treating DED [46]. Medicinal plants indicate androgen-like activity and can affect lacrimal glands and membrane cells, thereby contributing to DED treatment. Androgens may increase meibomian gland function and lacrimal gland function [47].
Clearly, it should be noted that some herbs did not have any positive effect on DED treatment (in humans or animals) [48,49]. Anticholinergic alkaloids are one of the strongest causes of the disease [50]. The most important plant compounds that lead to the development of DED include niacin, echinacea, and kava, and it should be noted that herbal drugs per se can cause eye complications for consumers, which is due to the natural origin of these products, are usually ignored by the consumers and even by the doctors who prescribe the herbal drugs [51,52].
For example, pouring black tea (Camellia sinensis) directly into the cornea with the corneal epithelial defect should be prevented, as it leads to anterior stromal discolouration in individuals [53]. On the other hand, determining the effective dose of medicinal plants is another factor affecting their efficacy. Determining the effective dose in DED treatment means the determination of dose for the highest efficacy and also in the toxic dose [28,35].
In some studies, this issue has been neglected and different quantities of plant extract or derivatives at different intervals have not been studied, which was one of the major problems with these studies [54,55].
Conclusion
Medicinal plants improve tear film stability by decreasing osmolality and increasing tear production. Several mechanisms, including the reduction of ROS (antioxidant activity), the prevention of cell apoptosis, the modulation of inflammatory factors, and the regulation of androgens, can affect lacrimal glands and membrane cells, thereby contributing to DED treatment.
[1]. Javadi MA, Feizi S, Dry Eye SyndromeJ Ophthalmic Vis Res 2011 6(3):192-98. [Google Scholar]
[2]. Gayton JL, Etiology, prevalence, and treatment of dry eye diseaseClin Ophthalmol 2009 3:405-12.10.2147/OPTH.S555519688028 [Google Scholar] [CrossRef] [PubMed]
[3]. Hom MM, Nguyen AL, Bielory L, Allergic conjunctivitis and dry eye syndromeAnn Allergy Asthma Immunol 2012 108(3):163-66.10.1016/j.anai.2012.01.00622374198 [Google Scholar] [CrossRef] [PubMed]
[4]. Hessen M, Akpek EK, Dry eye: an inflammatory ocular diseaseJ Ophthalmic Vis Res 2014 9(2):240-50. [Google Scholar]
[5]. Peck T, Olsakovsky L, Aggarwal S, Dry Eye syndrome in menopause and perimenopausal age groupJ Midlife Health 2017 8(2):51-54.10.4103/jmh.JMH_41_1728706404 [Google Scholar] [CrossRef] [PubMed]
[6]. Logaraj M, Madhupriya V, Hegde S, Computer vision syndrome and associated factors among medical and engineering students in chennaiAnn Med Health Sci Res 2014 4(2):179-85.10.4103/2141-9248.12902824761234 [Google Scholar] [CrossRef] [PubMed]
[7]. Golden MI, Fries PL, Dry Eye Syndrome 2017 Treasure Island (FL)StatPearls PublishingAvailable from: https://www.ncbi.nlm.nih.gov/pubmed/29262012 [Google Scholar]
[8]. Dry Eye USA: U.S. National Library of Medicine-The World’s Largest Medical Library; 2018 [Available from: https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0023579/ [Google Scholar]
[9]. Messmer EM, The pathophysiology, diagnosis, and treatment of dry eye diseaseDtsch Arztebl Int 2015 112(5):71-82.10.3238/arztebl.2015.007125686388 [Google Scholar] [CrossRef] [PubMed]
[10]. Bhavsar AS, Bhavsar SG, Jain SM, A review on recent advances in dry eye: Pathogenesis and managementOman J Ophthalmol 2011 4(2):50-56.10.4103/0974-620X.8365321897618 [Google Scholar] [CrossRef] [PubMed]
[11]. Yavangi M, Rabiee S, Nazari S, Farimani-Sanoee M, Amiri I, Bahmanzadeh M, Comparison of the effect of oestrogen plus Foeniculum vulgare seed and oestrogen alone on increase in endometrial thickness in infertile womenJ Clin Diagn Res 2018 12(1):QC01-QC04.10.7860/JCDR/2018/30164.11020 [Google Scholar] [CrossRef]
[12]. Shabanian S, Khalili S, Lorigooini Z, Malekpour A, Heidari-Soureshjani S, The effect of vaginal cream containing ginger in users of clotrimazole vaginal cream on vaginal candidiasisJ Adv Pharm Technol Res 2017 8(2):80-84. [Google Scholar]
[13]. Solati K, Asadi-Samani M, Heidari-Soureshjani S, Medicinal plants effective on serotonin level: a systematic reviewBr J Pharm Res 2017 19(4):01-12.10.9734/JPRI/2017/36836 [Google Scholar] [CrossRef]
[14]. Shirani M, Raeisi R, Heidari-Soureshjani S, Asadi-Samani M, Luther T, A review for discovering hepatoprotective herbal drugs with least side effects on kidneyJ Nephropharmacol 2017 6(2):38-48.10.15171/npj.2017.03 [Google Scholar] [CrossRef]
[15]. Shabanian G, Heidari-Soureshjani S, Rafieian-Kopaei M, Saadat M, Shabanian M, Therapeutic effects of Quercus persica l fruit skin on healing of second-degree burn wounds in animal modelJ Zanjan Uni Med Sci Health Services 2017 25(113):81-92. [Google Scholar]
[16]. Nikfarjam M, Rakhshan R, Ghaderi H, Comparison of effect of lavandula officinalis and venlafaxine in treating depression: a double blind clinical trialJ Clin Diagn Res 2017 11(7):KC01-KC04.10.7860/JCDR/2017/20657.1023328892932 [Google Scholar] [CrossRef] [PubMed]
[17]. Heidari-Soreshjani S, Asadi-Samani M, Yang Q, Saeedi-Boroujeni A, Phytotherapy of nephrotoxicity-induced by cancer drugs: an updated reviewJ Nephropathol 2017 6(3):254-63.10.15171/jnp.2017.4128975109 [Google Scholar] [CrossRef] [PubMed]
[18]. Rafieian-Kopaei M, Baradaran A, Rafieian M, Plants antioxidants: From laboratory to clinicJ Nephropathol 2013 2(2):152-53.10.5812/nephropathol.1211624475444 [Google Scholar] [CrossRef] [PubMed]
[19]. Nasri H, Herbal drugs and new concepts on its useJ Prev Epidemiol 2016 1(1) [Google Scholar]
[20]. Ekor M, The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safetyFront Pharmacol 2013 4:17710.3389/fphar.2013.0017724454289 [Google Scholar] [CrossRef] [PubMed]
[21]. Ukponmwan CU, Momoh N, Incidence and Complications of Traditional Eye Medications in Nigeria in a Teaching HospitalMiddle East Afr J Ophthalmol 2010 17(4):315-19.10.4103/0974-9233.7159621180431 [Google Scholar] [CrossRef] [PubMed]
[22]. Shenoy R, Bialasiewicz A, Khandekar R, Al Barwani B, Al Belushi H, Traditional medicine in Oman: its role in ophthalmologyMiddle East Afr J Ophthalmol 2009 16(2):92-96.10.4103/0974-9233.5386920142969 [Google Scholar] [CrossRef] [PubMed]
[23]. Wilkinson JT, Fraunfelder FW, Use of herbal medicines and nutritional supplements in ocular disorders: an evidence-based reviewDrugs 2011 71(18):2421-34.10.2165/11596840-000000000-0000022141385 [Google Scholar] [CrossRef] [PubMed]
[24]. Peng QH, Yao XL, Peng J, Wu QL, Tan HY, Zhang JR, Effects of extract of buddleja officinalis eye drops on lacrimal gland cells apoptosis of castrated rabbits with dry eyeInt J Ophthalmol 2010 10(1):40-43. [Google Scholar]
[25]. Hitoe S, Tanaka J, Shimoda H, MaquiBright standardized maqui berry extract significantly increases tear fluid production and ameliorates dry eye-related symptoms in a clinical pilot trialPanminerva Med 2014 56(3 Suppl 1):01-06. [Google Scholar]
[26]. Kim CS, Jo K, Lee IS, Kim J, Topical application of apricot kernel extract improves dry eye symptoms in a unilateral exorbital lacrimal gland excision mouseNutrients 2016 8(11):pii:E75010.3390/nu811075027886047 [Google Scholar] [CrossRef] [PubMed]
[27]. Kimura Y, Mori D, Imada T, Izuta Y, Shibuya M, Sakaguchi H, Restoration of tear secretion in a murine dry eye model by oral administration of palmitoleic acidNutrients 2017 9(4):pii:E36410.3390/nu904036428379171 [Google Scholar] [CrossRef] [PubMed]
[28]. Chien KJ, Horng CT, Huang YS, Hsieh YH, Wang CJ, Yang JS, Effects of Lycium barbarum (goji berry) on dry eye disease in ratsMol Med Rep 2018 17(1):809-18.10.3892/mmr.2017.794729115477 [Google Scholar] [CrossRef] [PubMed]
[29]. Kang SW, Kim KA, Lee CH, Yang SJ, Kang TK, Jung JH, A standardized extract of Rhynchosia volubilis Lour. exerts a protective effect on benzalkonium chloride-induced mouse dry eye modelJ Ethnopharmacol 2018 215:91-100.10.1016/j.jep.2017.12.04129288830 [Google Scholar] [CrossRef] [PubMed]
[30]. Chen M, Hu DN, Pan Z, Lu CW, Xue CY, Aass I, Curcumin protects against hyperosmoticity-induced IL-1beta elevation in human corneal epithelial cell via MAPK pathwaysExp Eye Res 2010 90(3):437-43.10.1016/j.exer.2009.12.00420026325 [Google Scholar] [CrossRef] [PubMed]
[31]. Scuderi G, Contestabile MT, Gagliano C, Iacovello D, Scuderi L, Avitabile T, Effects of phytoestrogen supplementation in postmenopausal women with dry eye syndrome: a randomized clinical trialCan J Ophthalmol 2012 47(6):489-92.10.1016/j.jcjo.2012.08.01923217501 [Google Scholar] [CrossRef] [PubMed]
[32]. Choi W, Kim JC, Kim WS, Oh HJ, Yang JM, Lee JB, Clinical effect of antioxidant glasses containing extracts of medicinal plants in patients with dry eye disease: a multi-center, prospective, randomized, double-blind, placebo-controlled trialPLoS One 2015 10(10):e013976110.1371/journal.pone.013976126457673 [Google Scholar] [CrossRef] [PubMed]
[33]. Choi W, Lee JB, Cui L, Li Y, Li Z, Choi JS, Therapeutic efficacy of topically applied antioxidant medicinal plant extracts in a mouse model of experimental dry eyeOxid Med Cell Longev 2016 2016:472741510.1155/2016/472741527313829 [Google Scholar] [CrossRef] [PubMed]
[34]. Huang JY, Yeh PT, Hou YC, A randomized, double-blind, placebo-controlled study of oral antioxidant supplement therapy in patients with dry eye syndromeClin Ophthalmol 2016 10:813-20.10.2147/OPTH.S10645527274185 [Google Scholar] [CrossRef] [PubMed]
[35]. Chen HC, Chen ZY, Wang TJ, Drew VJ, Tseng CL, Fang HW, Herbal supplement in a buffer for dry eye syndrome treatmentInt J Mol Sci 2017 18(8):pii:E169710.3390/ijms1808169728771187 [Google Scholar] [CrossRef] [PubMed]
[36]. Peng Q-H, Yao X-L, Wu Q-L, Tan H-Y, Zhang J-R, Effects of extract of Buddleja officinalis eye drops on androgen receptors of lacrimal gland cells of castrated rats with dry eyeInt J ophthalmol 2010 3(1):4310.3736/jcim2010030820226146 [Google Scholar] [CrossRef] [PubMed]
[37]. Chen Y-L, Hou MC-C, Acupuncture and Chinese herbal treatment for dry eye disease: two case reportsMed Acupunct 2012 24(1):50-55.10.1089/acu.2011.0827 [Google Scholar] [CrossRef]
[38]. Sawant DP, Parlikar GR, Binorkar SV, Efficacy of Triphala Ghrita Netratarpan in computer vision syndromeInt J Res Ayurveda Pharm 2013 4(2):244-48.10.7897/2277-4343.04232 [Google Scholar] [CrossRef]
[39]. Hu W, Qian X, Guo F, Zhang M, Lyu C, Tao J, Traditional Chinese medicine compound ShengJinRunZaoYangXue granules for treatment of primary Sjogren’s syndrome: a randomized, double-blind, placebo-controlled clinical trialChin Med J (Engl) 2014 127(15):2721-26. [Google Scholar]
[40]. Wu SQ, Dai Q, Xu JL, Sheng WY, Xu QB, Zhong LY, Combined effect of traditional Chinese and Western medicine on inflammatory factors in patients with diabetes-induced xerophthalmiaGenet Mol Res 2016 15(4):gmr1504903010.4238/gmr15049030 [Google Scholar] [CrossRef]
[41]. Peng QH, Yao XL, Wu QL, Chen M, Effects of extract of buddleja officinalis on prevention of dry eye in castrated rabbitsZhonghua Yan Ke Za Zhi 2008 44(11):1011-19. [Google Scholar]
[42]. Yao XL, Peng QH, Chen QL, Tang YH, Zhong Q, Effects of total flavonoid of chrysanthemum on Fas and Fasl expression in lacrimal gland cells of male castrated rabbits with dry eyeInt Eye Sci 2014 14(10):1749-54. [Google Scholar]
[43]. Wozniak A, Paduch R, Aloe vera extract activity on human corneal cellsPharm Biol 2012 50(2):147-54.10.3109/13880209.2011.57998022338121 [Google Scholar] [CrossRef] [PubMed]
[44]. Paduch R, Wozniak A, Niedziela P, Rejdak R, Assessment of Eyebright (Euphrasia Officinalis L.) Extract activity in relation to human corneal cells using in vitro testsBalkan Med J 2014 31(1):29-36.10.5152/balkanmedj.2014.837725207164 [Google Scholar] [CrossRef] [PubMed]
[45]. Barrera G, Oxidative stress and lipid peroxidation products in cancer progression and therapyISRN Oncol 2012 2012:13728910.5402/2012/13728923119185 [Google Scholar] [CrossRef] [PubMed]
[46]. Wang F, Peng QH, Li HZ, Wang F, Yao XL, Li WJ, Influence for Cell model of dry eye after intervention of plasma containing Buddleja Officinali Flavone and the expression of Bax mRNA and Bcl-2 mRNA in Lacrimal gland epithelial cellsInt Eye Sci 2012 12(10):1836-40. [Google Scholar]
[47]. Azcarate PM, Venincasa VD, Feuer W, Stanczyk F, Schally AV, Galor A, Androgen Deficiency and Dry Eye Syndrome in the Aging MaleInvest Ophth Vis Sci 2014 55(8):5046-53.10.1167/iovs.14-1468924994872 [Google Scholar] [CrossRef] [PubMed]
[48]. Abokyi S, Koffuor GA, Ofori EA, Thomford AK, Osei KA, Evaluating the effect of an aqueous extract of Pistia stratiotes Linn (Araceae) on tear secretion and tear film stability in ICR miceJ Appl Pharm Sci 2014 4(8):1-5. [Google Scholar]
[49]. Chan CC, Lau WN, Chiu SP, Chen LC, Choi WK, Tang GW, A pilot study on the effects of a Chinese herbal preparation on menopausal symptomsGynecol Endocrinol 2006 22(2):70-73.10.1080/0951359060058172316603430 [Google Scholar] [CrossRef] [PubMed]
[50]. Askeroglu U, Alleyne B, Guyuron B, Pharmaceutical and herbal products that may contribute to dry eyesPlast Reconstr Surg 2013 131(1):159-67.10.1097/PRS.0b013e318272a00e23271525 [Google Scholar] [CrossRef] [PubMed]
[51]. Fraunfelder FW, Ocular side effects from herbal medicines and nutritional supplementsAm J Ophthalmol 2004 138(4):639-47.10.1016/j.ajo.2004.04.07215488795 [Google Scholar] [CrossRef] [PubMed]
[52]. Fraunfelder FW, Ocular side effects associated with dietary supplements and herbal medicinesDrugs Today 2005 41(8):537-45.10.1358/dot.2005.41.8.89683516234877 [Google Scholar] [CrossRef] [PubMed]
[53]. Achiron A, Birger Y, Karmona L, Avizemer H, Bartov E, Rahamim Y, Corneal staining and hot black tea compressesIsr Med Assoc J 2017 19(3):152-55. [Google Scholar]
[54]. Jafari T, Antioxidants; helpful or harmful?Annals of Research in Antioxidants 2016 1(2):e13 [Google Scholar]
[55]. Baradaran A, Administration of herbal drugs in geriatric individuals; trends on its helps and hazardsGeriatrics Persia 2017 1(1):e01 [Google Scholar]