In the present scenario where there is increased incidence of sexual assaults and crime rate, there is an ever increasing need for the development of newer techniques helping justice to find its path and law to play its role. Saliva is one of the vital fluids secreted in human beings, which is deposited on the human skin through various actions like biting, sucking etc., [1,2] and hence, saliva stands as a source for identification of accused. Bite marks analysis is very difficult because human dentition does not always leave identifying features imprinted on the skin surface [3]. The detected saliva is recognised as one of the primary source of DNA by the forensic serologist [4]. Fluorescent spectroscopy is proved to have high sensitivity and specificity, and has the advantage of being non-invasive and cost effective method. Fluorescent spectroscopy is a newer technique in detecting saliva with 100 percent specificity. Hence, this study was conducted to detect saliva on human skin, as it plays an important role in identification of suspect [5].
Materials and Methods
This is an observational study conducted at IICT (Indian Institute of Chemical Technology) Hyderabad. The institutional ethical clearance was obtained. Twenty saliva samples of healthy volunteers were tested and compared with healthy control group (water).
Determination of optimum excitation wavelength of undiluted saliva (uncontaminated) was carried out and emission spectrum of salivary samples was compared with tryptophan to standardise the wavelengths. Then emission spectrum of salivary samples and water (as a control) were compared.
The three Undiluted saliva samples were taken and placed in three wells of black well plate (triplet) separately & excited with a wavelength between 230 and 320 nm. The peak excitation wavelength of dried saliva samples and water were recorded, which were used to obtain their emission spectrum. 0.5 mg/mL of tryptophan was dissolved in 5 mM KCl and three samples of this solution were used to obtain emission spectrum. Tryptophan was excited to peak excitation wavelength of undiluted saliva i.e., 254 nm to get emission spectrum and was compared with the emission spectrum obtained from dried saliva samples and water.
The forearm was cleaned with soap and dried to prevent any contamination, before depositing saliva. An area on the forearm was marked and the volunteer was instructed to deposit saliva in the area by licking at normal temperature. Water was deposited on the other hand which was taken as control. Both the saliva and water were allowed to dry for 30-40 minutes. Fluorescent spectroscopy of saliva samples and control samples (water) were taken from skin of 20 volunteers each. In pH 7.4 phosphate buffer of 0.1M KCL fibre-free cotton dipped with an excess solution removed was rubbed over the marked area. Swab from control area was also taken. Each swab was mixed in 2ml KCl in a separate cuvette for 10 seconds and were transferred to quartz cuvette to record the fluorescence emission spectrum from 300-540 nm using spectrofluorometer.
Results
The Peak Excitation Wavelength of Undiluted Saliva Samples
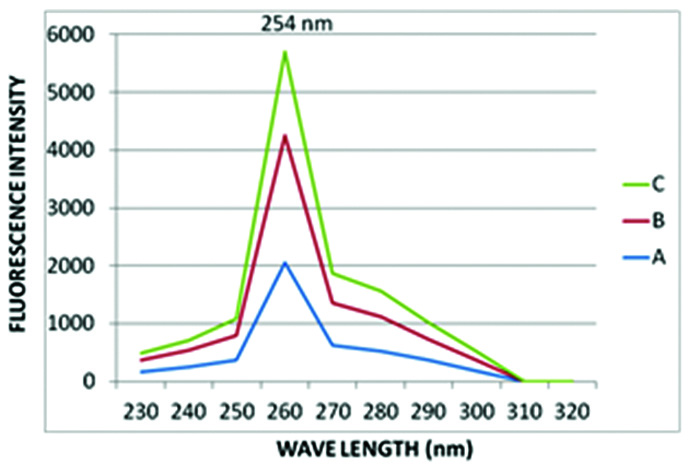
The three undiluted saliva samples were excited to wavelengths between 230-320 nm. The maximum absorption spectra was characterised by an excitation peak at 254 nm, which was considered as the maximum excitation wavelength for obtaining emission spectra of saliva samples [Table/Fig-1].
Peak excitation wavelength of undiluted saliva.
Emission Spectrum of Tryptophan
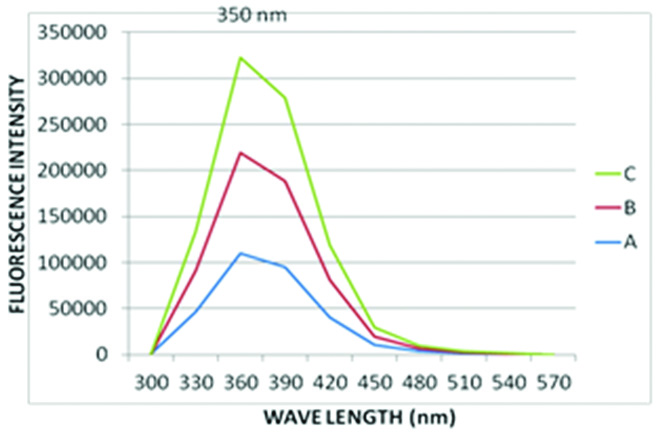
The peak emission spectrum of tryptophan was found to be 350 nm, when it is excited to its peak excitation wavelength of 254 nm [Table/Fig-2].
Emission spectrum of Tryptophan.
Emission spectra and fluorescent intensity of saliva
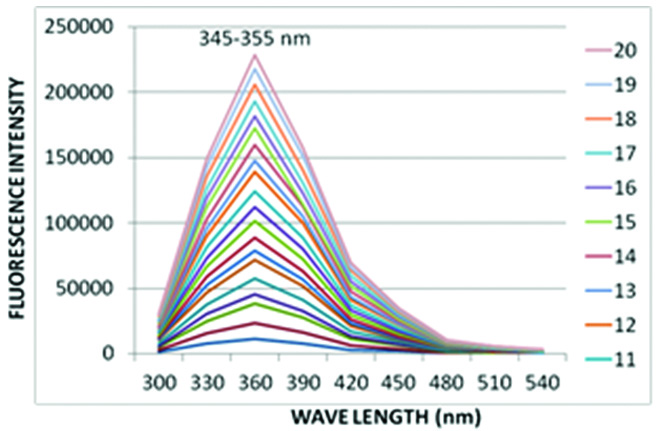
The peak emission spectrum of tryptophan was recorded at 350 nm. The 20 salivary swabs taken from dried saliva had a primary spectrum ranging from 345-355 nm. The emission spectrum of tryptophan was matched well with the emission spectrum obtained from 20 saliva swab samples from human skin [Table/Fig-3].
Emission spectrum of saliva samples.
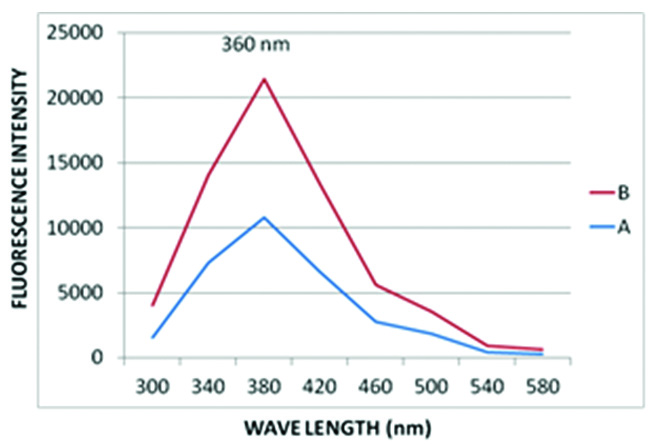
Whereas, the emission spectrum of water as a control was recorded at 360nm, which is at a higher wavelength compared to that of saliva samples [Table/Fig-4,5].
Emission spectrum of water.
| Samples | Wavelength |
|---|
| Tryptophan | 350 nm |
| Saliva samples | 345–355 nm |
| Water | 360 nm |
Discussion
Saliva plays a major role in forensic sciences because of presence of varied biological molecules such as, chemical markers (thiocynate ion, amylase), serological markers (blood group substances, macro molecular glycoproteins) for individual identification and of substance abuse, sex determination, and DNA analysis [6].
Saliva found on victims can be potentially recovered and typed from bite marks, cigarette butts, postage stamps, envelopes, edibles, and other objects [7]. The average turnover of epithelial cells in the mouth is every 2.7 hour and the mean number of epithelial cells per 1 mL of saliva is about 4.3 x 105 suggesting that there is higher probability of intact genomic DNA in saliva samples. Saliva can be used even when it is stored in different conditions [8]. Recent studies have shown that human genomic DNA can be reliably obtained from saliva [9].
Fluorescent spectroscopy is widely used to analyse structure dynamics and functional interactions of proteins [5]. It is based on the principle that when a fluorescent material is excited at a particular wavelength, it emits radiation of longer wavelength which can be recorded [10].
Tryptophan, an aromatic amino acid is present in saliva as an important constituent of salivary amylase. When it is excited at a particular peak excitation wavelength it produces a characteristic emission spectrum. This property of tryptophan was used to detect dried salivary samples. In our study, 20 salivary samples were excited at a peak excitation wave length of tryptophan i.e., 254nm and characteristic emission spectrum of 345-355nm was obtained which conferred with characteristic emission spectrum of tryptophan and hence, the samples collected were that of the saliva. The results obtained in our study were in accordance with the earlier studies conducted by Nanda KDS et al., which demonstrated that saliva had characteristic emission spectrum of 345-355nm with peak excitation spectrum at 350nm [11].
Fluorescent spectroscopy has shown 100% sensitivity in detecting dried saliva stains on skin and hence, can be a useful diagnostic tool for forensic examinations [11], especially when it is difficult to analyse the bite marks due to laceration of the skin surface [3] and is economical compared to that of other methods.
Tryptophan is one of the essential amino acid which forms an important constituent of many proteins in the body. It is present in different body fluids in different concentration. From our study it is found that, tryptophan a natural fluorophore present in saliva can be detected with 100% sensitivity and specificity by fluoroscent spectroscopy method, thus can be differentiated from other body fluids [12,13].
Limitation
Small sample size is the major limitation.
Conclusion
Saliva is one of the vital fluids secreted in the body, which has an advantage of non-invasive method of collection. Although DNA extraction from saliva has been used as one of the common method but many advanced techniques have revolutionised over a period of time in identification of different body fluids. Fluorescent spectroscopy method has established itself as a reliable method in the field of forensic science and found to be cost-effective as well. Fluorescent properties of different fluids of the body can play a major role in forensic identification.