Acrylic resins were introduced in the market in 1930s and since then they have been used to make bases for total dentures and removable dentures. Among its main characteristics are the ease of handling, good thermal conductivity, low permeability of oral fluids and chromatic stability [1].
Although, the conventional acrylic resin possesses pleasing aesthetic characteristics, the association with staples for retention of the Removable Partial Dentures (RPD) does not always please the patients. Therefore, a material was developed to convey the need of retention of the RPDs with the aesthetics demand from the patients, the flexible thermoplastic resin [2].
Some professionals question the use of these resins in making RPDs due to its basic structure, contradicting the traditional principles of biomechanics, such as absence of supports and its structural flexibility [3]. However, the use of thermoplastic resins for the manufacture of flexible RPDs has increased over the years [4,5].
There is an increasing number of thermoplastic RPDs every year, demonstrating their popularity in dental practice. However, in the academic world they are still seen with certain restrictions, especially because the number of works evaluating the physical and mechanical properties of this material are still scarce in the literature. To date, the existing works show promising results indicating that the use of this material is practical in clinical daily life [5-11].
Some studies show the success of rehabilitation with polyamide-based prostheses as well as the advantages of its properties but longitudinal and randomised studies are still needed to consolidate the rehabilitation technique [1,2,4,12,13].
The objective of this work was to test an experimental formulation of a thermoplastic flexible resin supplied by the manufacturer and to compare it with a commercially available flexible resin in order to ascertain whether the physical and mechanical properties would improve with the changes made, bringing benefits to the prostheses quality. The properties of the experimental flexible resin were also compared to a conventional thermopolymeriable acrylic resin that was (PMMA) taken as control.
Materials and Methods
This in vitro study took place at the Aracatuba Dental School/UNESP University, from July 2015 to August 2016. Two types of samples were made, 30 in circular format (3×10 mm) for color and microhardness tests, and 60 in rectangular format (64×10×3.3 mm) for the flexural strength test. The different shapes of the samples were according to the previously published studies [1,14,15]. The samples were divided into three groups (n=10) one of the experimental flexible resin tested, one with a flexible resin already marketed (Sorriflex, Peramical Com. e Ind. D e Produtos Médicos e Odontológicos e Ltda-Maringá, PR, Brazil), and a control group of a conventional thermopolymerisable acrylic resin (Lucitone, Dentisply Industria E Comercio Limitada, Petrópolis, RJ, Brazil). The samples were constructed in duplicate for the flexural strength test because the samples were lost due to fracture after the initial tests, hence, 60 samples were made to test the flexural strength.
For the preparation of the samples of the flexible resins, specimens were obtained in extrahard silicon (Zetalabor, Zhermack, Italy) with 10 mm diameter and 3 mm thick for the color and hardness test and 64×10×3.3 mm for the flexural strength test, by means of a metal pattern [1,14,15]. These samples were made in a professional prosthetic laboratory, and the extrahard silicon specimens served as a mold.
The flexible resins (Sorriflex and experimental) used in this study were made from an isopropylene thermoplastic material. They were made by a thermoinjectable system in which the samples sent were prepared in an appropriate muffle with sprus system for injection of the thermoplastic resin. These same specimens were used as a template and included in conventional muffles, into which the acrylic resin Lucitone was provided and mixed according to the manufacturer’s recommendations and carried out in hot water bath polymerisation. Each resin property and manufactures are summarised in [Table/Fig-1] and their respective images are represented in [Table/Fig-2] [2,14,16].
Acrylic resins used in the study.
| Comercial brand | Polimerisation process | Basic chemical composition | Manufacturer |
|---|
| Sorriflex (Flexible resin) | Thermoinjection | Polyamide | Peramical Com. e Ind. D e Produtos Médicos e Odontológicos e Ltda-Maringá, PR, Brazil [16] |
| Experimental (Flexible resin) | Thermoinjection | Polyamide | ------------ |
| Lucitone 550 (Acrylic resin) | Muffle immersion into water 73°C for 90 minutes and then 100°C for 30 minutes | Monomer: Methyl methacrylate, ethylene glycol, dimethacrylate, hydroquinonePolymer: Copolymer (methyl-n-butyl), methacrylate, benzoyl peroxide, mineral dies. | Dentsply Industria E Comercio Limitada, Petrópolis, RJ, Brazil [2,14] |
Image of acrylic resins used in the study.
a-Sorriflex, b-Experimental resin, c-Lucitone 550
All the samples obtained were then polished in a sequence of metallographic sandpaper with granulation 600, 800 and 1200 (Buehler, Illinois, USA) in automatic grinder polisher (Ecomet 300 PRO; Buehler, Illinois, USA) under continuous irrigation of water for one minute with each sandpaper at 300 RPM speed. Each disc had its thickness measured with the aid of a digital caliper (500-171-20B, Mitutoyo, Tokyo, Japan), in order to obtain the proposed dimensions. After polishing, the samples were submitted to ultrasonic cleaning (Arotec, Odontobrás, São Paulo, SP) for 20 minutes in distilled water for removal of possible debris on the resin surface and then left outdoors for drying.
After this procedure, color, microhardness and flexural strength readings were performed. Afterwards the samples were submitted to the thermocycling test for 2000 cycles, simulating two clinical years use of these materials [17,18]. The samples were immersed in distilled water, undergoing alternating baths of 60 seconds at 5±1°C and 55±1°C temperature. At the end of the procedure, the samples were again submitted to color, microhardness, and flexural strength readings.
Chroma and luminosity changes were evaluated through a spectrophotometer (UV-2450, Shimadzu Corp, Kyoto, Japan), and the color alterations were calculated with the L*a*b* system established by the Commission Internationale de l’Eclairag (CIE) with standard D65 lighting [1,19].
The microhardness tests (Knoop) were performed using a microhardness tester (HMV-2T, Shimadzu Corp., Kyoto, Japan) calibrated with 25 gm load for 10 seconds [18,20]. Three readings were performed on each surface of the sample being these 500 m distant from the sample margin and, at the same time, 500 m distant from one penetration to another.
The flexural strength test was performed on a universal testing machine (EMIC, São José dos Pinhais, SP, Brazil) by means of the rupture modulus test, according to the three point bending flexural test. The samples were positioned in the machine in a holder with a distance of 50 mm between its edges, with a load cell of 100 kg and a constant velocity of 5 mm/min, until its fracture occurred.
The strength of each sample was recorded graphically, and the flexural strength was measured from the load deflection curve obtained. The value of the flexural strength, in MPa, was calculated using the following equation [1]:
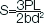
In which P is the bursting load, L is the length between the supports (50 mm), b represents the width and d is the thickness of the sample.
Statistical Analysis
The numerical values obtained from the analysis of color change, microhardness and flexural strength of the specimens mentioned above were evaluated with descriptive statistical analysis (mean and standard deviation). Then, these values were submitted to the test of adherence to the normal curve, to assess the proper parametric or non parametric tests. As a normal distribution was detected, ANOVA was performed to assess the differences among the groups and the Tukey post-test was applied when necessary to identify such differences.
Results
Color Change
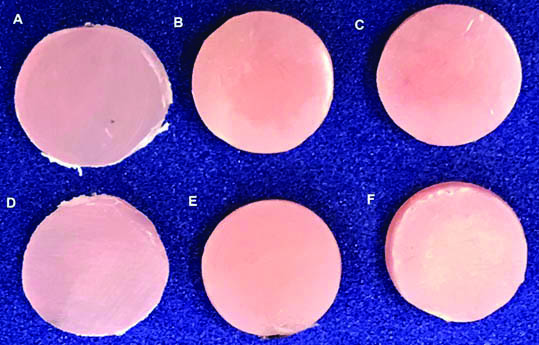
ANOVA showed no statistically significant difference (p=0.677). The ΔE at this stage was equivalent for all groups, with values ranging from 1.17 to 1.50. [Table/Fig-3] describes the mean values and the standard deviation of the color change for each resin tested. [Table/Fig-4] presents the color change for each resin before and after the thermocycling.
Tukey test analysis on mean values and standard deviation of the color change (ΔE) of the samples for each type of resin.
| Sample | ΔE Mean (SD) |
|---|
| Lucitone | 1.17 (1.1) A |
| Sorriflex | 1.47 (0.8) A |
| Tested | 1.50 (0.7) A |
Means followed by the same capital letter in the column do not differ at the 5% level of significance (p<0.05) by the Tukey test
Images of the color change (ΔE) of the samples for each type of resin and period.
A-Sorriflex initial, B-Experimental resin initial, C-Lucitone initial, D-Sorriflex after thermocycling, E-Experimental resin after thermocycling, F-Lucitone after thermocycling
Surface Hardness
In the hardness data, ANOVA demonstrated a statistical difference for time (p<0.001) and resin (p<0.001) which influenced the characteristics of the evaluated materials. In present case, we observed an increase in hardness after the thermocycling period. The resin tested showed higher hardness (7.71) when compared to the Sorriflex (6.69) resin and both showed lower hardness values than the conventional resin (17.32) [Table/Fig-5]. In the hardness results, the tested resin showed higher values than the flexible resin found in the market.
Tukey test analysis on mean values and standard deviation of samples hardness for each period (initial and thermocycling) and resin type.
| Sample | Initial | Thermocycling |
|---|
| Lucitone | 17.32 (1.3) A,a | 19.6 (1.8) A,b |
| Sorriflex | 6.69 (1.2) B,a | 7.75 (0.6) B,b |
| Tested | 7.71 (0.6) C,a | 8.72 (1.1) C,b |
Means followed by the same capital letter in the column and the same lowercase letter in the row do not differ at the 5% level of significance (p<0.05) by the Tukey test.
Flexural Strength
Only the resin type influenced the results of flexural strength on ANOVA (p<0.001), the time period (p=0.124) had no effect on the samples. We found that there was no difference between the flexible resins, the tested resin (70.7) behaved in a manner similar to the resin found in the market (68.0). Only the control group (83.5) presented difference with the others [Table/Fig-6].
Tukey test analysis on mean values and standard deviation of flexural strength of the samples for each period (initial and thermocycling) and type of resin.
| Sample | Initial | Thermocycling |
|---|
| Lucitone | 90.74 (4.5) A,a | 83.53 (5.5) A,a |
| Sorriflex | 70.71 (6.7) B,a | 68.05 (5.8) B,a |
| Tested | 76.68 (7.7) B,a | 70.75 (5.9) B,a |
Means followed by the same capital letter in the column and the same lowercase letter in the row do not differ at the 5% level of significance (p<0.05) by the Tukey test.
Discussion
Based on the tested results, the null hypothesis of the study was denied, since the resin tested had a superficial hardness higher than the already commercialised resin. Other properties, such as color change and flexural strength, were similar among the evaluated materials. The found similarity might be due to the same basic composition of evaluated resins. Regarding the results of color change, all resins behaved in a similar and adequate manner, all presented a color variation lower than 3.3, which according to some authors is considered the limit perceivable to naked eye [1,15]. Changes bigger than 3.3 are considered clinically unacceptable. Goiato et al. found similar results (ΔE ranged from 1.62 to 5.75) related to the color change of flexible resins subjected to accelerated aging, only a flexible resin evaluated (Valpast) showed clinically unfavorable results (ΔE≥3.3) [1].
Hatim NA and Al-Tahho OZ also found acceptable values for the flexible and thermopolymerizable resins tested when submitted to immersion in artificial saliva for up to 12 weeks (ΔE 1,31), but when samples were submerged in pigmented solutions such as coffee and tea, the thermoplastic resins presented unacceptable results, with a ΔE of 4,04 for coffee and 26,03 for tea [21].
In another study, two acrylic resins (Vipi Cril and Lucitone) and a nylon based thermoplastic (Transflex) were submerged in solutions with high pigment content, such as red wine, coffee, coca-cola and distilled water for control. All were influenced by red wine, but the flexible resin also suffered color change by coca-cola. In present study, immersion in distilled water had a ΔE around 1,2 [11].
The chromatic changes of the resins can occur extrinsically and intrinsically, the intrinsic changes are more related to aging and to changes in temperature and humidity, leading to loss of surface gloss and discoloration of the material [1]. Usually thermoplastic resins are more hydrophilic than acrylic resins, a fact that explains the greater ease of suffering pigmentation, as pointed out in different studies [11,18,21].
Regarding the hardness, we verified that the thermocycling influenced the hardness; since, the hardness values were higher for all the groups after thermocycling. Regarding the resins, we observed that the experimental resin showed higher values of hardness in relation to the Sorriflex resin and both flexible resins presented lower values of hardness than the conventional acrylic resin. These results are in agreement with other studies, in which higher values of microhardness was obtained for the methyl methacrylate resins in relation to polyamide based resins [1, 22].
This difference is due to the greater presence of crosslink agents and the presence of plasticisers in acrylic resins than in polyamide [22]. The increase in hardness after thermocycling can be attributed to complete polymerisation of the material and also the continuous temperature change, which can cause a contraction in the material [1,15]. Higher values of microhardness is positive for these materials, because the higher the hardness, the lower the incidence of scratches and possible fractures of the material [15].
Contrary to the microhardness, the flexural strength was not influenced by the thermocycling process, the only difference was in respect to the type of resin. In present case, the thermoplastic acrylic resin presented a higher resistance to thermoplastics, as reported in other studies [23-26]. Iwata Y found similar results when comparing flexural characteristics of different resins used to make denture bases, the polyamide based resins showed the lowest values of flexural strength (45.3 to 55.5 MPa) and the acrylic resins (90.1 to 95.8 MPa), the highest [27].
Shah J et al., found lower values of flexural strength for the nylon-based group and this measure decreased after immersion of these specimens in disinfectant solutions [22]. As described by other authors, we observed that thermoplastic resin samples do not suffer a disruption like thermopolymerised acrylic resins, their high flexibility causes the samples to suffer an irreversible deflection instead [22-26].
In general, the tested resin presented good physical and mechanical properties, the higher hardness of this resin, without altering its flexibility, makes it more resistant to scratches and abrasions during hygiene, which could prolong the longevity of the same, reducing the proliferation of microorganisms on its surface [1].
The thermoinjectable acrylic resins are starting to be used in large scale, therefore there are still not many studies that evaluate their properties. However, there are some reports of its successful clinical use, not only for the manufacture of removable partial dentures, but also for complete dentures and obturators [4,6,18]. It is possible to observe a great variety of formulations for these resins, they can be fabricated from nylon, polyamide, polyurethane, polyester, polycarbonate and the methyl methacrylate base [6,28,29].
It is important to mention that the experimental flexible resin is a non commercialised product from a manufacturing company whose manufacturers declared that its composition was polyamide, however, they did not give information about proportion of its basis regarding pigments and components of its formulae. Thus, this study sought to compare the properties of the experimental resin to an already commercialised resin, with the same composition, and to enable the introduction of this resin into the market, by the manufacturers, even though only few properties were studied in this experiment.
Limitation
The limitation of present study was that only one type of formulation of the thermoplastic resins (polyamide) were studied and we did not subject the resins to situations of clinical use such as immersion in pigment solutions or disinfection. Another limitation was that there was no data on the biocompatibility of the new formula with human cells. Therefore, new studies are needed to evaluate not only these but also other properties of thermoplastic resins in order to consolidate their clinical use and more properties of the experimental resin need to be studied so that this material can be introduced to the market.
We wanted to compare the properties of the experimental resin to an already commercialised resin to enable the introduction of the experimental resin into the market by the manufacturer, that was the reason of using two resins with the same composition.
Conclusion
We conclude that the experimental resin presented clinically acceptable characteristics; its color change, microhardness and flexural strength are similar to resin already marketed.
Conflicts of Interest
The experimental resin was given by Laboratório de Prótese Treco, Araçatuba, São Paulo, Brazil. The authors declare that the true composition was known and held by the company and none of the authors were employed or part of this company. The authors declare that this study was self funded and neither the company associated to this study nor the authors will have any conflict of interest in future.