Hereditary Haemorrhagic Telangiectasia (HHT) also known as Osler Weber Rendu disease is a rare, under diagnosed, autosomal dominant multiorgan angio dysplasia. We present a case of 61-year-old female with complaints of abdominal distension and recurrent epistaxis. Contrast Enhanced Computed Tomography (CECT) abdomen revealed arterio portal shunt, disseminated intrahepatic telangiectasias, dilated hepatic arteries, and hepatic haemangioma. Furthermore presence of cirrhosis with focal left lobar Intra Hepatic Biliary Radical Dilatation (IHBRD) suggested advanced disease. On further investigation pulmonary involvement was also identified. HHT being clinicoradiological diagnosis, knowledge of its feature is essential for its timely correct diagnosis and to obviate inadvertent biopsy.
Arterio portal fistula, Intrahepatic, Osler Weber Rendu disease
Case Report
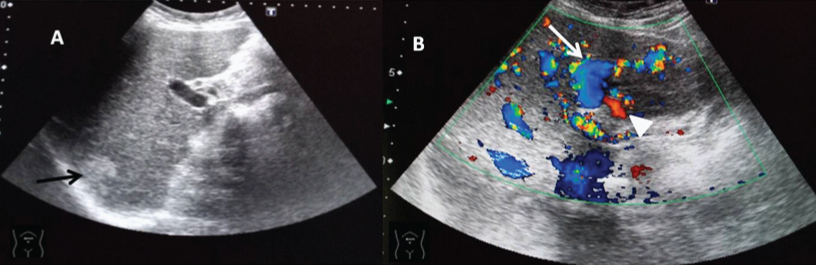
A 61-year-old female presented with slowly progressive painless abdominal distension for one month. She also complained of recurrent episodes of spontaneous epistaxis for 20 years requiring blood transfusions, and had family history of recurrent epistaxis. Clinical examination revealed nasal mucocutaneous telangiectasias, liver enlargement, firm to hard in consistency, with surface nodularity and irregular margin. Ultrasound abdomen revealed hepatomegaly, with diffusely coarse echotexture, irregular margin and abnormally dilated tortuous left portal vein, dilated left hepatic artery, and right lobe hepatic haemangioma [Table/Fig-1].
Ultrasound revealed: (A) diffusely coarse echotexture, irregular margin of liver and a well defined hyperechoic lesion in segment VI suggestive of haemangioma (black arrow); (B) dilated tortuous left portal vein (white arrow) communicating with dilated left hepatic artery (white arrow head).
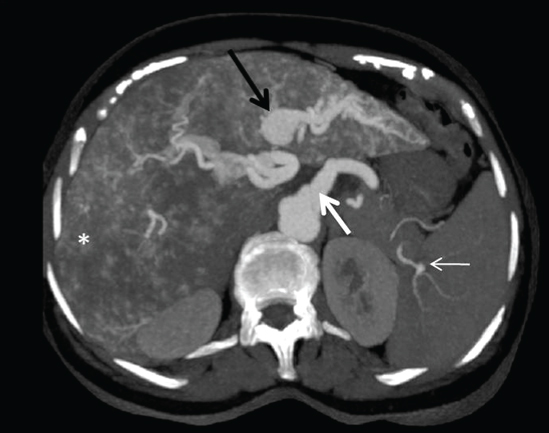
Triphasic CECT abdomen revealed, early arterial enhancement of aneurysmally dilated left portal vein with vascular ectasia suggestive of arterio portal shunt in left lobe of liver. Common hepatic artery was dilated measuring 8.5 mm in diameter [Table/Fig-2]. On arterial phase, there was evidence of mosaic type perfusion pattern, multiple intrahepatic telangiectasias diffusely in both lobes of liver, splenic artery aneurysm and right lobar hepatic haemangioma [Table/Fig-3]. There was also evidence of hepatomegaly, with underlying chronic liver disease in form of irregular contour, minimal left lobar IHBRD [Table/Fig-4] and mild ascites. Thus, as the clinicoradiological findings were suggestive of Osler Weber Rendu disease, further supplementary studies like pulmonary angiography and CECT brain were done, which revealed, pulmonary hypervascularisation in bilateral lung fields in the most peripheral segments and saccular out pouching in segmental pulmonary artery in right middle lobe [Table/Fig-5]. However, no Arterio Venous Malformation (AVM) was seen in paranasal sinuses and brain. Post processing image reconstructions, such as Maximum Intensity Projection (MIP) and 3D Volume Rendering (VR) techniques were used [Table/Fig-6]. As patient revealed family history of recurrent epistaxis, her first degree relatives were assessed, which revealed no conclusive results. Consequently, in presence of these findings, the patient was diagnosed with HHT. Epistaxis was treated by local tamponade, during episodes of severe bleeding, blood transfusion was given. Patient is under follow-up for six months and has been doing well with no other complaints.
Axial CECT arterial phase reveals opacification of aneurysmally dilated left portal vein with vascular ectasia suggestive of arterio portal shunt in left lobe of liver (thick black arrow). Dilated celiac artery (thick white arrow), disseminated telangiectasias (asterisk) and splenic artery aneurysm (white thin arrow).
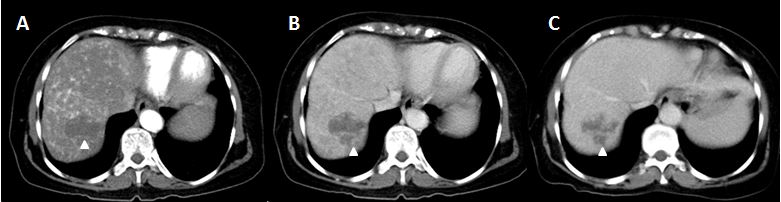
Axial CECT reveals well defined lesion with peripheral, nodular enhancement in arterial phase: (A) and progressive filling in venous; (B) and delayed; (C) phase suggestive of haemangioma (white arrow head).
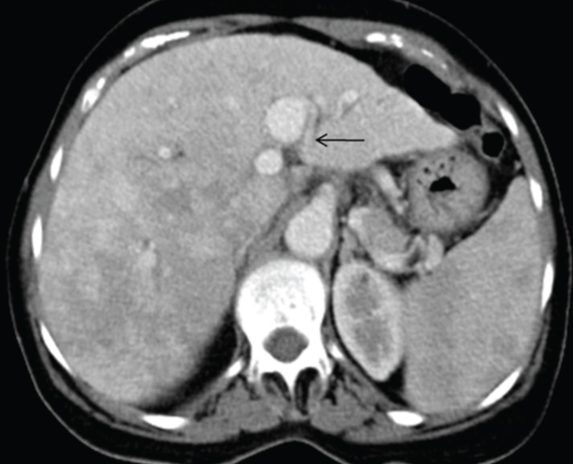
Axial CECT abdomen also revealed contour nodularity and minimal left lobar intrahepatic biliary radical dilatation IHBRD (black arrow).
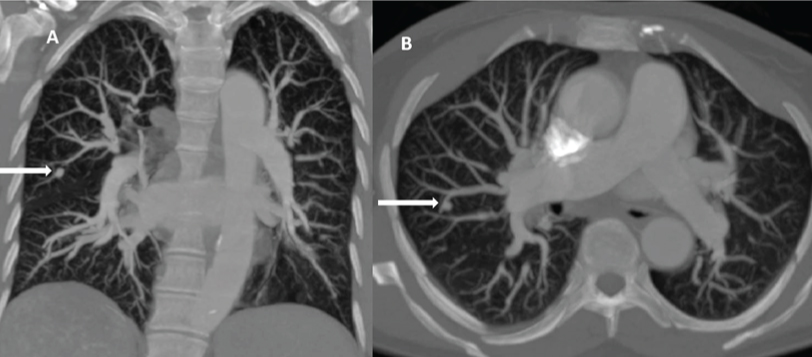
Pulmonary angiography: (A) coronal and (B) axial images revealed, pulmonary hypervascularisation in bilateral lung fields in peripheral segments and saccular outpouching in segmental pulmonary artery in right middle lobe (white arrow).
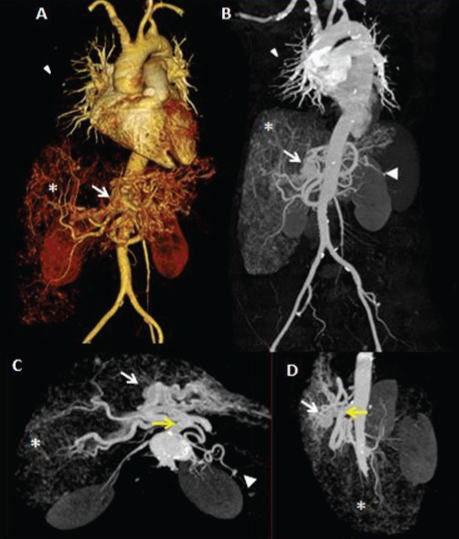
(A) 3D volume rendering image; (B) coronal MIP image; (C) axial MIP image; (D) saggital MIP image in arterial phase reveal, saccular outpouching in segmental pulmonary artery (white arrowhead), opacification of left portal vein (thick white arrow), disseminated telangiectasias (asterisk), splenic artery aneurysm (white arrow head) and dilated celiac artery (yellow arrow).
Discussion
Osler Weber Rendu disease also known as HHT, is a rare, under diagnosed autosomal dominant multisystem angio dysplasia with prevalence of 1 in 5000-10,000 [1]. According to Curacao criteria, diagnosis of HHT is made in presence of three of the four following criteria: spontaneous recurrent epistaxis, mucocutaneous telangiectasia, first degree relative with HHT, and visceral AVMs [1,2]. In present case, all these four criteria’s including hepatic AVMs were present. Hepatic involvement is usually asymptomatic, involving >60% of patients of HHT.
Ultrasound being the primary imaging modality detects hepatic and large vascular lesions; however, telangiectasias may not be visualised. Triphasic CECT can diagnose vascular abnormalities such astiny telangiectasias, intrahepatic shunts, haemangiomas, mosaic perfusion and confluent vascular lesions. Early arterial enhancement of hepatic veins is suggestive of arterio venous fistula. Early arterial enhancement of portal vein suggests arterio portal shunt, as in this case. Also, communication of dilated portal veins with systemic or hepatic vein in portal venous phase is suggestive of intrahepatic porto systemic venous shunt [3,4].
The presence of diffuse hepatic telangiectasias and dilated common hepatic artery are pathognomonic for hepatic HHT [3]. Telangiectasias is seen in about 90% of this patients. They are hypervascular arterial enhancing rounded punctate nodules which are isodense in hepatic parenchymal phase. The common hepatic artery is also abnormally dilated (diameter >7mm) due to increased volume flow in the hepatic artery due to intrahepatic shunts. Mosaic perfusion pattern is transient hepatic attenuation difference, due to alteration of blood flow induced by the arterio venous shunts and telangiectasias, thus, is an indirect sign of the presence of shunts [5]. Confluent vascular lesions appear as early arterial enhancing lesions with persistent enhancement on delayed phases. Cirrhosis secondary to extensive necrotising cholangitis, if present, is indicative of an advanced disease [3,4]. Biliary abnormalities are consequence of biliary ischemia as a result of shunting of the blood away from the peribiliary plexus and also due to compression of bile ducts by dilated vessels.
MRI reveals prominent intrahepatic and extrahepatic arterial branches, dilated hepatic and portal veins and vascular shunts. Magnetic Resonance Cholangio Pancreatography (MRCP) sequences, can detect irregular bile duct strictures with resultant IHBRD [4].
HHT being a multisystem disorder, its pulmonary manifestation includes AVM, pulmonary hypertension, peripheral hypervascularisation, dilatation of segmental pulmonary arteries, which may have delayed presentation as in the present case [5-7].
In the present case, findings were consistent with, left lobe arterio portal shunt with vascular ectasia, diffuse hepatic telangiectasias, dilated common hepatic artery and splenic artery aneurysm. Also, cirrhotic configuration of liver and minimal left lobe IHBRD, suggested advanced hepatic HHT. Furthermore, pulmonary angiography revealed saccular dilation of segmental pulmonary artery with peripheral pulmonary hypervascularisation. Agnollitto PM et al., has reported similar cases [8]. However, in this case there is an unusual late presentation of such extensive hepatic involvement with asymptomatic pulmonary involvement.
Differential diagnoses include phakomatoses with cutaneous and visceral involvement, such as RASA1 mutation, Adams Oliver syndrome (scalp and limb defects with cutaneous telangiectasias and visceral AVMs) and Wyburn Mason syndrome (retinoencephalofacial angiomatosis) [8].
Management of these patients depends upon their clinical presentation. Most of them require supportive care in form of oral iron supplements, whereas, blood transfusion is given to patients with severe bleeding as in the present case. Epistaxis was managed by local tamponade. However, liver transplant is the treatment of choice in patients with hepatic HHT, with severe organ insufficiency [9].
A long diagnostic delay of three decades is seen in patients with HHT since onset of the disease. Thus, a high index of suspicion and early diagnosis is necessary for better health outcomes and improved quality of life [10].
Conclusion
HHT is clinicoradiological diagnosis, MDCT being an essential diagnostic tool to detect intrahepatic complex vascular lesions. In presence of solid organ AVM, it is essential that the radiologist has high degree of clinical suspicion for HHT, does premptive radiological survey for possible involvement of other organs for its expedient diagnosis and prevents unadvertant biopsy.
[1]. Bayrak-Toydemir P, Mao R, Lewin S, McDonald J, Hereditary hemorrhagic telangiectasia: An overview of diagnosis and management in the molecular era for cliniciansGenet Med 2004 6(4):175-91.10.1097/01.GIM.0000132689.25644.7C15266205 [Google Scholar] [CrossRef] [PubMed]
[2]. Carette MF, Nedelcu C, Tassart M, Grange JD, Wislez M, Khalil A, Imaging of Hereditary Hemorrhagic TelangiectasiaCardiovasc Intervent Radiol 2009 32:745-57.10.1007/s00270-008-9344-218972161 [Google Scholar] [CrossRef] [PubMed]
[3]. Wu JS, Saluja S, Garcia-Tsao G, Chong A, Henderson KJ, White RI Jr, Liver involvement in hereditary hemorrhagic telangiectasia: CT and clinical findings do not correlate in symptomatic patientsAJR 2006 187(4):W399-W405.10.2214/AJR.05.106816985112 [Google Scholar] [CrossRef] [PubMed]
[4]. Bilgin M, Yildiz S, Toprak H, Ahmad IC, Kocakoc E, CT and MRI Findings of hepatic involvement in rendu-osler-weber diseaseCase Reports in Radiol 2012 2012:48408510.1155/2012/48408523198241 [Google Scholar] [CrossRef] [PubMed]
[5]. Song W, Zhao D, Li H, Ding J, He N, Chen Y, Liver Findings in patients with hereditary hemorrhagic telangiectasiaIran J Radiol 2016 13(4):e3111610.5812/iranjradiol.3111627895866 [Google Scholar] [CrossRef] [PubMed]
[6]. Abdalla SA, Gallione CJ, Barst RJ, Horn EM, Knowles JA, Marchuk DA, Primary pulmonary hypertension in families with hereditary haemorrhagic telangiectasiamEur Respir J 214 23(3):373-77.10.1183/09031936.04.0008550415065824 [Google Scholar] [CrossRef] [PubMed]
[7]. Vorselaars VMM, Velthuis S, Snijder RJ, Vos JA, Mager JJ, Post MC, Pulmonary Hypertension in Hereditary Haemorrhagic TelangiectasiaWorld J Cardiol 2015 7(5):230-37.10.4330/wjc.v7.i5.23026015855 [Google Scholar] [CrossRef] [PubMed]
[8]. Agnollitto PM, Barreto ARF, Barbieri RFP, Junior JE, Muglia VF, Rendu-Osler-Weber syndrome: what radiologists should know. Literature review and three cases reportRadiol Bras 2013 46(3):168-72.10.1590/S0100-39842013000300011 [Google Scholar] [CrossRef]
[9]. Sabba C, A rare and misdiagnosed bleeding disorder: Hereditary hemorrhagic telangiectasiaJ Thromb Haemost 215 3:2201-10.10.1111/j.1538-7836.2005.01345.x15892861 [Google Scholar] [CrossRef] [PubMed]
[10]. Pierucci P, Lenato GM, Suppressa P, Lastella P, Triggiani V, Valerio R, A long diagnostic delay in patients withhereditary haemorrhagic telangiectasia: A questionnaire based retrospective studyOrphanet J Rare Dis 2012 7:3310.1186/1750-1172-7-3322676497 [Google Scholar] [CrossRef] [PubMed]