Introduction
Anomalies of the mucin layer of the tear film have generally been studied in relation to contact lens users who represent a young population. Since the prevalence of dry eye increases with age and causes considerable discomfort to elderly patients, a proper assessment of tear film quality is required to facilitate a targeted approach in managing Dry Eye Disorder (DED) in older age.
Aim
To study the ferning pattern of the precorneal tear film in elderly individuals and compare the results with standard tests for dry eye.
Materials and Methods
A questionnaire based screened cohort of 75 individuals with DED and 75 controls, all above 50 years of age, were studied using Schirmer’s Test, Tear Break Up Time (TBUT), Lissamine green staining and ferning tests. Ferning patterns were graded by Rolando’s classification. Data were analysed by Fisher’s-exact test.
Results
Dry eye tests and tear ferning were found to be abnormal in majority of the subjects who had positive scores on screening with the dry eye questionnaire. Abnormal ferning patterns (62% Grade III and 24% Grade IV) correlated significantly with positive Schirmer’s test (p≤0.001), positive TBUT test (p≤0.001) and positive Lissamine green staining (p≤0.001) of eyes with DED. Less than 14% individuals (22 eyes) exhibited Grade II ferning in the dry eye group. Greater disturbances of ferning patterns were seen with increased age. All patients above the age of 75 (n=22 eyes) with symptomatic DED had abnormal ferning. Amongst the controls, all 150 eyes, regardless of age, showed normal ferning patterns (56% Grade 1 and 44% Grade 2).
Conclusion
A significant majority of elderly individuals with DED have an abnormal mucin layer in the tear film. Tear ferning test is a simple and inexpensive test and if combined with the other tear film tests may be helpful in a better assessment of DEDs, especially in the elderly. This is relevant especially for targeted treatment in cases of DED where the cause is not aqueous deficiency.
Introduction
Dry eye disorder is a condition that presents with multiple symptoms of ocular discomfort and is associated with increased osmolarity of the tear film and inflammation of the ocular surface. It is encountered very frequently in the ophthalmology clinic and the prevalence varies from 10.8% to 57.1%. Any abnormality in the functioning of the lacrimal gland or eyelids, or any ocular surface disorder may result in dry eye [1-5].
The presence of dry eye is influenced by age and gender, with higher incidences reported in patients above the age of 50 years. The two main reasons for development of DED are aqueous deficiency and increased evaporation of tears. Older patients can be affected by both these problems [6-10].
Decreased tear production may be due to dysfunction of the lacrimal gland. Androgen and oestrogen deficiency leads to poor lacrimal gland secretion and tear film instability in older men and women [11]. Older patients are more prone to autoimmune disorders like Sjogren’s syndrome and rheumatoid arthritis, which can result in severe dry eye [12].
It is also seen, that with age, the functioning of the conjunctival goblet cells reduces though the number remain the same [13]. The cells are also more prone to apoptosis [14]. Both these factors can lead to tear deficiency.
A large number of the population above the age of 60 years is on various systemic or topical medications for prolonged periods which may be responsible for causing a deficiency of tears. Drugs, including antidepressants, antimetabolites, diuretics and dopaminergics for Parkinson’s disease may all cause dry eyes. In addition, topical medication for glaucoma is also responsible for dry eye, especially since they are prescribed for long durations. Preservatives like Benzalkonium Chloride (BAK) common in most ophthalmic preparations, are responsible for tear film instability, loss of goblet cells and damage to the epithelium. Various factors also affect tear evaporation in old age. Anatomical disorders of the eyelids like laxity, lagophthalmous, floppy eyelid syndrome, ectropion, and entropion may lead to corneal exposure and increased evaporation of tears. Conjunctivochalasis can affect the tear meniscus and may occasionally occlude the inferior punctum [11]. Evaporative dry eye is more common than tear deficiency.
No single test of tear function can give a definite diagnosis of dry eye. Dry eye assessment is made with the help of various tests which detect tear film stability (Tear film breakup time), tear volume (Schirmer’s test), ocular surface damage (rose bengal dye test) and goblet cell deficiency (Lissamine dye test) [15].
Evaluation of the ferning pattern of the tears is also a test for mucin deficiency. Ferning of tears is seen due to a shift in the normal salt-to-macromolecules ratio on sudden evaporation of the fluid. The various ferning patterns observed are graded as per Rolando’s scale. Type I and II are predominantly seen in normal eyes and Type III and IV in DEDs [16].
Symptoms of dry eye may also be related to tear osmolarity, which can be detected by abnormal ferning patterns.
Studies of ferning tests have largely been performed to evaluate the safety of contact lenses as regards the tear film, and hence confined to younger populations [17].
Since the prevalence of dry eye increases with age and causes considerable discomfort to elderly patients, this study was undertaken to make a proper assessment of tear film quality with a simple tear ferning test and to facilitate a targeted approach for managing DED in older age.
The aim of the present study was the ferning pattern of the pre corneal tear film in elderly individuals and compare the results with standard tests for dry eye.
Materials and Methods
A case controlled observational study, duly approved by the Institutional Ethics Committee, was conducted on 150 adults (300 eyes) above the age of 50 years attending the eye OPD of a teaching hospital, from April 2015 to March 2016 (calculated as per prevalence based formula wherein prevalence is 5-30%) [10]. All participants were enrolled after obtaining written informed consents. Recently operated cases, individuals who were on any topical medication and with active infection were excluded. Presence of any systemic disorders was noted.
Dry eye disease has a significant impact on visual acuity, daily activities, social and physical functions and workplace productivity. Screening is best done with the help of questionnaires. In this study the initial screening for DED was determined on the basis of a Dry eye questionnaire. A pre-validated McMonnie’s questionnaire [18] was used to assign participants to one of two equal groups, i.e., Group A those with DED, and Group B those without dry eye (Non DED).
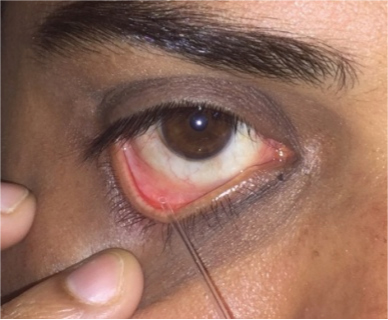
All individuals underwent routine ophthalmic evaluation. Additionally, standard dry eye tests i.e., Schirmer’s test, TBUT test and Lissamine Green staining of the ocular surface were performed on all the participants by a single investigator. Further Schirmer’s test and TBUT test were graded as mild, moderate and severe and Lissamine test as positive and negative. Ferning test was done by obtaining a drop of tear by means of a micropipette/capillary tube applied to the inferior fornix [Table/Fig-1]. The tear drop was allowed to dry on a clean slide under normal atmospheric conditions. The Ferning patterns were examined under a light microscope and graded by Rolando’s classification Grade 1 to 4 [19].
Collecting tear film for ferning.
Statistical Analysis
Data were captured on a predetermined proforma and analysed by Fisher’s-exact test using IBM SPSS version 20.0. A p-value of ≤ 0.05% was considered to be significant.
Results
A total of 150 adults (300 eyes), above the age of 50 years, participated in this study. A total of 75 individuals (150 eyes) in Group A (DED) and 75 individuals (150 eyes) in Group B (Non DED).
Mean Schirmer’s score in Group A (DED) was 7.3 mm with a range of 6 mm to 14 mm. No patients had a Schirmer’s value <5 mm (Severe dry eye). In Group B (Non DED) the mean score was 25.61 mm with a range of 18 mm to 30 mm.
Mean TBUT score in Group A (DED) was 7.6 seconds with a range of 5 second to 9 seconds. No patients had a TBUT value of <4 seconds (severe dry eye). In Group B (Non DED) the mean score was 19.63 seconds with a range of 11 seconds to 21 seconds.
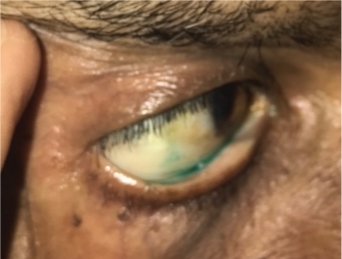
In Group A (DED), 64 individuals (n=128 eyes) tested positive with Lissamine green staining [Table/Fig-2] and 11 individuals (n=22 eyes) were negative. However, in Group B (Non DED), all patients tested negative for Lissamine green staining.
Lissamine green staining. (Images from left to right)
In Group A (DED), 64 individuals (n=128 eyes) had abnormal ferning pattern of Grade 4 and 3 [Table/Fig-3,4]. Of these 46 individuals (n=92 eyes) were of Grade 3 and 18 individuals (n=36 eyes) were in Grade 4. Eleven individuals (n=22 eyes) in this group had a ferning of Grade 2 [Table/Fig-5]. No patients showed Grade 1 ferning pattern.
Grade 4 of ferning pattern-No ferning is present; mucus may appear in clusters and threads.
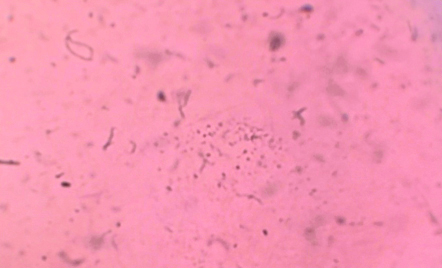
Grade 3 of ferning pattern-Ferns are little and incompletely formed with rare or no branching.
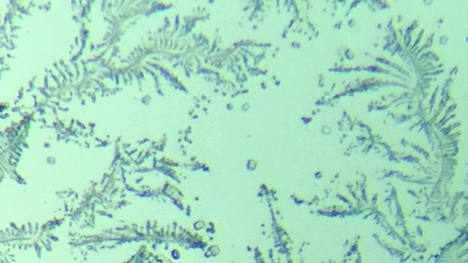
Grade 2 of ferning pattern-Arborisation is abundant, but the ferns are smaller and have a lower frequency of branching than in grade I; empty spaces appear between the ferns.

However, in Group B (Non DED) all had normal ferning patterns. A total of 42 individuals (n=84 eyes) had Grade 1 [Table/Fig-6] and 33 individuals (n=66 eyes) had Grade 2 ferning pattern.
Grade 1 of ferning pattern-Uniform arborisation in the entire field of observation without spaces between the ferns. Single ferns are big and closely branched.

Comparison of ferning pattern with schirmer’s test: Of the 150 eyes in Group A, 22 eyes had Grade 2 ferning pattern, of which 18 eyes had mild Schirmer’s test positive and 4 eyes had moderate Schirmer’s test. In the same group 92 eyes had Grade 3 Ferning pattern of which 72 eyes had mild Schirmer’s test positive and 20 eyes had moderate Schirmer’s test positive. A total of 36 eyes had Grade 4 ferning pattern of which 30 eyes had mild Schirmer’s test positive and 6 eyes had moderate Schirmer’s test positive, showing a significant positive correlation between abnormal ferning pattern and Schirmer’s test (p≤0.001) [Table/Fig-7].
Comparison of ferning pattern with schirmer’s test and TBUT in DED group.
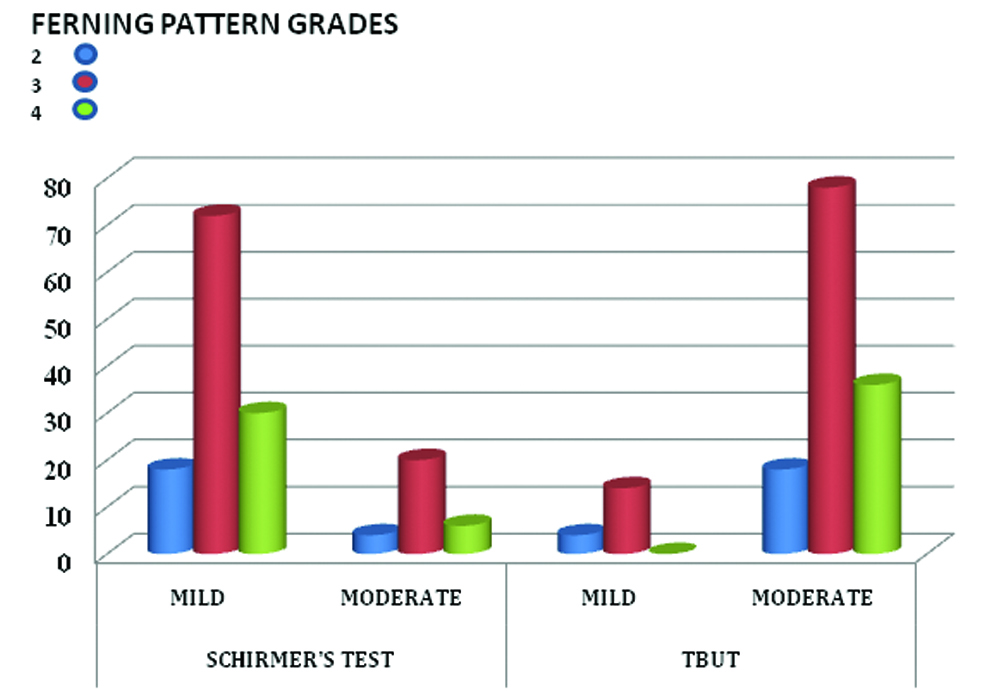
The comparison of ferning patterns and Schirmer’s test in the DED group of patients revealed that though 128 eyes had significant abnormal ferning pattern (Grade 3 and 4), the Schirmer’s test showed only mild dry eye readings in 120 eyes and moderate dry eye readings in 30 eyes. No patients had a reading of <5 mm in this group.
Comparison of ferning pattern with TBUT: Of the 150 eyes in Group A, 22 eyes had Grade 2 ferning pattern of which 4 eyes had mild TBUT positive and 18 eyes had moderate TBUT positive. In the same Group 92 eyes had Grade 3 Ferning pattern of which 14 eyes had mild TBUT positive and 78 eyes had moderate TBUT positive. A total of 36 eyes had Grade 4 Ferning pattern and all had moderate TBUT positive, showing a positive correlation between abnormal Ferning pattern and TBUT (p≤0.001).
A total of 85% patients (n=126 eyes) in the DED group had a positive Lissamine green Test. All these patients had ferning pattern of Grade 3 and 4. Hence, it was seen that ferning patterns correlated best with Lissamine Green test (p≤0.001).
Discussion
It is well known that older individuals have a higher prevalence of DED [10]. Sharma A and Hindman HB, reported a DED prevalence of 8.4% in subjects younger than 60 years, 15% in 70-79 years and 20% in patients older than 80 years of age [11]. Since, present study was conducted on patients above the age of 50 years; a higher incidence of 86% DED was seen.
Most studies on ferning patterns have been done in connection with contact lens usage and, therefore, have necessarily been restricted to young populations [16,17].
To the best of our knowledge, there are no reports on this aspect of dry eye amongst the elderly. This study was, therefore, undertaken to determine mucin layer anomalies in older individuals.
Although, Meibomian Gland Dysfunction (MGD) is the most common entity in the spectrum of DEDs, both aqueous layer anomalies, as well as mucin layer deficiencies do occur, though to a lesser extent [20,21].
The quality of the mucin layer is better determined by examining its ferning pattern. There is an interaction of several factors, other than mucin, which also contribute to ferning [22]. Ferning occurs due to a shift in the ratio of salt to macromolecules that are present in mucin and the aqueous proteins.
Studies on the ferning in ophthalmology were introduced in 1982 by Tabbara K and Okumoto M [23]. Subsequently, in 1984; Rolando M introduced a grading system for classifying the ferning pattern and correlating it to the severity of the mucin deficiency [19].
Although, the Schirmer’s test is considered as the most sensitive test for aqueous deficiency in DED, it may not provide useful information regarding the mucin layer due to a paradoxical increase of compensatory aqueous secretion in more severe cases of dry eye [24].
In our study, 128 eyes belonging to DED group had Ferning pattern of Grade 3 and 4. However, Schirmer’s test of these individuals was only mildly positive in 102 eyes and moderately positive in 26 eyes. Hence a significant mucin deficiency was present despite an only mildly positive Schirmer’s. All symptomatic elderly individuals had abnormal Ferning pattern (Grade 3 and 4).
Mucin layer anomalies contribute to instability of the tear film as detected by the TBUT. In present study, ferning pattern results correlate well with TBUT (p≤0.001). However, even in those eyes with Grade 4 ferning pattern, TBUT values were not in the severe range. This indicates that some patients with significant mucin deficiency may not be detected on TBUT which is similar to the findings of the study conducted by Paderbach S and Stolze H [25].
The presence of mucin layer deficiency is easily detected by ocular surface staining with Lissamine green [15,26]. In present study, a positive Lissamine staining was seen in 85% of eyes of patients in the DED group indicating a widespread prevalence of mucin layer deficiency in the elderly. Sahai A and Malik P had reported 18.4% positivity for the same [27].
The present study was conducted on patients above the age of 50 and hence a higher percentage of positive Lissamine test was seen. All these patients had ferning pattern of Grade 3 and 4. Hence, it was seen that ferning patterns correlated best with Lissamine Green test (p≤0.001).
A total of 14.6% (22 eyes) of the symptomatic group had grade 2 ferning and a negative Lissamine staining, however Schirmer’s test and TBUT were abnormal. It is likely that these individuals had some other factors contributing to mucin deficiency and not just an alteration of the ratio of salts to macromolecules. Poor Ferning correlated significantly with positivity of Schirmer’s test and TBUT as well (p≤0.001).
Results of dry eye tests and ferning patterns were all within normal limits in the control group.
Limitation
There was some difficulty in collecting adequate tear samples for the ferning test. Humidity and temperature variations can alter the ferning pattern of tears and hence, results may vary. The sample size was also not very large.
Conclusion
We can therefore conclude that ferning patterns correlate well with the state of the tear film. Both for aqueous volume and tear film stability, as determined by standard tests for dry eye. Ferning pattern is aimed at evaluating mucin deficiency. It is a simple and inexpensive test and if combined with the other tear film tests may be helpful in a better assessment of DEDs, especially in the elderly. This is relevant especially for targeted treatment in cases of DED where the cause is not aqueous deficiency.
[1]. Hikichi T, Yoshida A, Fukui Y, Hamano T, Ri M, Araki K, Prevalence of dry eye in Japanese eye centersGraefes Archive for Clinical and Experimental Ophthalmology 1995 233:555-58.10.1007/BF004047058543205 [Google Scholar] [CrossRef] [PubMed]
[2]. Farrell J, Grierson DJ, Patel S, Sturrock RD, A classification for dry eyes following comparison of tear thinning time with schirmer tear testActa Ophthalmologica (Copenh) 1992 70:357-60.10.1111/j.1755-3768.1992.tb08579.x1636398 [Google Scholar] [CrossRef] [PubMed]
[3]. Toda I, Fujishima H, Tsubota K, Ocular fatigue is the major symptom of dry eyeActa Ophthalmologica (Copenh) 1993 71:347-52.10.1111/j.1755-3768.1993.tb07146.x8362634 [Google Scholar] [CrossRef] [PubMed]
[4]. Albietz JM, Prevalence of dry eye subtypes in clinical optometry practiceOptometry and Vision Science 2000 77:357-63.10.1097/00006324-200007000-00010 [Google Scholar] [CrossRef]
[5]. Versura P, Cellini M, Torreggiani A, Profazio V, Bernabini B, Caramazza R, Dryness symptoms, diagnostic protocol and therapeutic management: a report on 1200 patientsOphthalmic Research 2001 33:221-27.10.1159/00005567411464075 [Google Scholar] [CrossRef] [PubMed]
[6]. Rege A, Kulkarni V, Puthran N, Khandgave T, A clinical study of subtype-based prevalence of dry eyeJ Clin Diagn Res 2013 7(10):2207-10.10.7860/JCDR/2013/6089.347224298477 [Google Scholar] [CrossRef] [PubMed]
[7]. Gupta N, Prasad I, Himashree G, D’Souza P, Prevalence of dry eye at high altitude: a case controlled comparative studyHigh Alt Med Biol 2008 9:327-34.10.1089/ham.2007.105519115918 [Google Scholar] [CrossRef] [PubMed]
[8]. Paulsen AJ, Cruickshanks KJ, Fischer ME, Huang GH, Klein BE, Klein R, Dry eye in the beaver dam offspring study: prevalence, risk factors, and health-related quality of lifeAm J Ophthalmol 2014 157:799-806.10.1016/j.ajo.2013.12.02324388838 [Google Scholar] [CrossRef] [PubMed]
[9]. Vashisht S, Singh S, Evaluation of phenol red thread test versus schirmers test in dry eyes: a comparitive studyInt J Appl Basic Med Res 2011 1(1):40-42.10.4103/2229-516X.8197923776771 [Google Scholar] [CrossRef] [PubMed]
[10]. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye Work Shop (2007)Ocul Surf 2007 5(2):93-107.http://www.tearfilm.org/dewsreport/pdfs/TOS-0502-DEWS-noAds.pdf10.1016/S1542-0124(12)70082-4 [Google Scholar] [CrossRef]
[11]. Sharma A, Hindman HB, Aging: a predisposition to dry eyesJournal of Ophthalmology 2014 2014:781683Available from https://www.hindawi.com/journals/joph/2014/781683/abs/10.1155/2014/78168325197560 [Google Scholar] [CrossRef] [PubMed]
[12]. Lemp M, Foulks G, The definition & classification of dry eye disease,”in Guidelines from the 2007 International Dry Eye Workshop 2008 Available from http://www.tearfilm.org/pdfs/OM%20-%20Definition%20%26%20Classification.pdf [Google Scholar]
[13]. Zhu W, Hon Zheng J, Le Q, Xu J, Sun X, Age-related changes of human conjunctiva on in vivo confocal microscopyBritish Journal of Ophthalmology 2010 94(11):1448-53.10.1136/bjo.2008.15582020494916 [Google Scholar] [CrossRef] [PubMed]
[14]. Giebel J, Woenckhaus C, Fabian M, Tost F, Age-related differential expression of apoptosis-related genes in conjunctival epithelial cellsActa Ophthalmologica Scandinavica 2005 83(4):471-76.10.1111/j.1600-0420.2005.00472.x16029273 [Google Scholar] [CrossRef] [PubMed]
[15]. Savini G, Prabhawasat P, Kojima T, Grueterich M, The challenge of dry eye diagonosisClin Ophthalmol 2008 2(1):31-55.10.2147/OPTH.S149619668387 [Google Scholar] [CrossRef] [PubMed]
[16]. Evans KS, North RV, Purslow C, Tear ferning in contact lens wearerOphthalmic Physiol Opt 2009 29(2):199-204.10.1111/j.1475-1313.2008.00626.x19236590 [Google Scholar] [CrossRef] [PubMed]
[17]. Guillon M, Maissa C, Contact lens wear affects tear film evaporationEye Contact Lens 2008 34(6):326-30.10.1097/ICL.0b013e31818c5d0018997542 [Google Scholar] [CrossRef] [PubMed]
[18]. McMonnies C, Ho A, Wakefield D, Optimum dry eye classification using questionnaire responsesAdvances in experimental medicine and biology 1998 438:835-38.10.1007/978-1-4615-5359-5_1179634975 [Google Scholar] [CrossRef] [PubMed]
[19]. Rolando M, Tear mucus ferning test in normal and keratoconjunctivitis sicca eyesChibret Int J Ophthalmol 1984 2:32-41. [Google Scholar]
[20]. Viso E, Gude F, Rodríguez-Ares MT, The association of meibomian gland dysfunction and other common ocular diseases with dry eye: a population based study in SpainCornea 2011 30:01-06.10.1097/ICO.0b013e3181da577820847672 [Google Scholar] [CrossRef] [PubMed]
[21]. Mathers W, Ocular evaporation in meibomian gland dysfunction and dry eyeOphthalmology 1993 100(3):347-51.10.1016/S0161-6420(93)31643-X [Google Scholar] [CrossRef]
[22]. Kogbe O, Liotet S, Tiffany JM, Factors responsible for tear ferningCornea 1991 10(5):433-44.10.1097/00003226-199109000-000131935142 [Google Scholar] [CrossRef] [PubMed]
[23]. Tabbara K, Okumoto M, Ocular ferning test: a qualitative test for mucus deficiencyOphthalmology 1982 89:712-14.10.1016/S0161-6420(82)34736-3 [Google Scholar] [CrossRef]
[24]. The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye Work Shop (2007)Ocul Surf 2007 5(2):85-88.10.1016/S1542-0124(12)70081-2 [Google Scholar] [CrossRef]
[25]. Paderbach S, Stolze H, Tear ferning and other lacrimal tests in normal persons of different agesInternational Ophthalmology 1991 15:391-95.10.1007/BF001379501778670 [Google Scholar] [CrossRef] [PubMed]
[26]. Hamrah P, Alipour F, Jiang S, Sohn J-H, Foulks GN, Optimizing evaluation of lissamine green parameters for ocular surface stainingEye 2011 25:142910.1038/eye.2011.18421836630 [Google Scholar] [CrossRef] [PubMed]
[27]. Sahai A, Malik P, Dry Eye: Prevalence and attributable risk factors in a hospital based populationInd J Ophthalmol 2005 53:87-91.10.4103/0301-4738.1617015976462 [Google Scholar] [CrossRef] [PubMed]