The AKI is characterised as a rapid reduction in kidney function, resulting in failure to maintain fluid, electrolyte and acid base balance. AKI supersedes the term Acute Renal Failure (ARF) and is a spectrum, extending from a less severe form to an advanced form, which necessitates the need of Renal Replacement Therapy (RRT) [1].
Prevalence is defined as occurrence (both newly diagnosed and previously diagnosed) of an event as a proportion of total admissions. A large variation is reported in the disease prevalence (1-25%) and mortality (15-60%). The reported prevalence of AKI in United States ranges from 1% (CA-AKI) to 7% (HA-AKI) of total hospital admissions. Complications related to AKI in general healthcare facilities accounted for up to 3% of admissions [2]. It was estimated that about two million people across the globe die of AKI every year and survivors have a higher risk of developing CKD [3].
In a prospective observational study conducted in 100 critically ill patients by Eswarappa M et al., in Indian population, it was found that incidence of AKI was 17.3 cases/1000 persons. They also found that prevalence of AKI in ICU settings was >40% if sepsis was present, and the mortality rates ranged from 15% to 60% [4]. An increased risk of AKI is observed in elderly and people with multiple comorbidities [5]. Study done in Italy found that incidence of AKI is 10 times higher in hospitalised patients aged 65 years or more than their younger counterparts [6].
Studies have shown that DM alone and UTI patients with DM, septic shock or impaired baseline renal function were at higher risk of developing AKI [7,8]. Recent studies in Indian population shows nephrotoxic drugs as the most common cause of AKI among medical wards whereas sepsis among surgical wards and ICU [4,9-11]. To determine AKI; physical examination, volume status, variations in serum creatinine levels, urine output and proteinuria are crucial parameters [12,13].
The main key for the management of AKI is assuring satisfactory renal perfusion and the therapy depends on the severity of the condition. AKI is usually treated with hydration, loop diuretics and RRT [12,14]. Insulin, dextrose, sodium bicarbonate, calcium gluconate and low dose dopamine are used for treatment of complications (such as hyperkalaemia, metabolic acidosis) associated with AKI [12].
AKI is an independent risk factor in critically ill patients leading to mortality [1,15,16]. Long term follow-up studies has shown AKI as risk factor for CKD or exacerbation of underlying CKD. Many of inpatients are being affected by AKI indicating its impact on healthcare resources. According to frequency and mortality rate of AKI patients, prevention of 20% of cases of AKI, would drastically reduce number of deaths, complications and their associated costs [14].
The study was conducted with a primary objective of finding the prevalence among hospitalised patients. The secondary objective was to identify risk factors, aetiology and outcomes associated with AKI. The present study will provide us with the data needed to improve patient safety, well-being and to preserve the kidney function mitigating the long term economic burden of CKD. Early treatment of risk factors can reduce the prevalence of AKI and the morbidity and mortality associated with it.
Materials and Methods
A retrospective single centred observational study was conducted in a tertiary care teaching hospital over a period of six months (August 2015 to February 2016). Ethical clearance was obtained from Institutional Ethics Committee (KH IEC 392/2015). Data of patients aged ≥18 years admitted during January-December 2012 were collected from the medical records using the ICD 10 code for AKI, unspecified (N17.9) [17].
Renal transplant recipients and patients receiving chronic dialysis were excluded from the study. Patient data such as demographic details, social habits, history, complaints on admission, medical and medication history, aetiology, risk factors for suspecting AKI, laboratory values, urine output levels, management and outcomes were recorded in Case Record Form (CRF). Patients were grouped as CA-AKI if they were presented to the hospital with AKI, and as HA-AKI if they developed AKI after hospital admission or during their stay in hospital.
Statistical Analysis
Data analysis was performed using statistical software SPSS (Version 20.0). Descriptive statistics were used for the analysis. The data for continuous variables was summarised as median and IQR whereas categorical variables were summarised as frequency and percentage. Odds Ratio (OR) was calculated for risk factors using univariate and multivariate analysis. Multivariate logistic regression model was used for multivariate analysis of statistically significant variables from univariate analysis, to identify the association of risk factors with the outcomes (CKD and death). Statistical tests were two tailed, and p<0.05 was considered to show a statistical difference. Chi square test for association was performed between different treatment modalities used and the patient outcome at time of discharge.
Results
Of a total of 73,318 patients admitted to a tertiary care teaching hospital in southern India from January to December 2012. A total of 613 patients were found to have diagnosed with AKI, of which, 60 patients were below the age of 18 years and 25 patients met the exclusion criteria. Thus, 528 patients met the inclusion criteria and were included in the study and considered for the analysis.
Demographics of Study Population
The median (IQR) age of the study population was 56 (20) years. About 43% (n=227) of the patients were of age above 60 years. Patients in the age groups 50-59 years and 60-69 years constituted for 24.4% (n=129) and 25.2% (n=133) respectively, showing more prevalence of AKI among the age group 60-69 years. The study showed a male preponderance of 72.7% (n=384) among the admitted patients. It was observed that 23.5% (n=73) of the patients were overweight and 7.8% (n=24) were obese [Table/Fig-1]. The prevalence of AKI was found to be 8.36 cases per 1,000 people.
Demographics and patient characteristics of AKI patients.
| Variables | Number of patients (Frequency) | Percent (%) |
|---|
| Median age (IQR) | 56 (20) | |
| Age >65 years | 160 | 30.3 |
| Age (Years) |
| 18-29 | 49 | 9.3 |
| 30-39 | 45 | 8.5 |
| 40-49 | 78 | 14.8 |
| 50-59 | 129 | 24.4 |
| 60-69 | 133 | 25.2 |
| 70-79 | 68 | 12.9 |
| >80 | 26 | 4.9 |
| Gender |
| Male | 384 | 72.7 |
| Female | 144 | 27.3 |
| Body Mass Index (BMI) |
| Under weight (<18.5 Kg/m2) | 55 | 17.7 |
| Normal (18.5-24.9 Kg/m2) | 158 | 51.0 |
| Over weight (25-29.9 Kg/m2) | 73 | 23.5 |
| Obesity (>30 Kg/m2) | 24 | 7.8 |
| Social habits |
| Alcoholic | 112 | 21.2 |
| Smoker | 91 | 17.2 |
Clinical Features at Presentation
An amount of 63% (n=335) were asymptomatic, 23.3% (n=123) experienced decreased urine output and 8.7% (n=46) had pedal oedema. Elevated serum creatinine level (>2 mg/dL) was found in 92% (n=484) of patients and elevated Blood Urea Nitrogen (BUN) in 92.4% (n=477) of patients. Proteinurea (≥30 mg/dL) was found in 68.8% (n=290) of patients. Since, most of the patients were asymptomatic, laboratory investigations were prerequisite for the diagnosis and thereby for better prognosis of AKI [Table/Fig-2].
Clinical Presentation of AKI.
| Criteria (n) | Number of patients (Frequency) | Percent (%) |
|---|
| Complaints on admission (528) |
| Asymptomatic | 335 | 63.0 |
| Decreased urine output | 123 | 23.3 |
| Pedal oedema | 46 | 8.7 |
| Decreased urine output and pedal oedema. | 24 | 4.5 |
| Serum creatinine (mg/dL) (526) |
| Median (IQR) | 4.2 (4.02) | |
| Normal (<1.9) | 42 | 8 |
| Abnormal (≥2) | 484 | 92 |
| 2-3.9 | 202 | 38.4 |
| 4-5.9 | 112 | 21.3 |
| 6-7.9 | 81 | 15.4 |
| 8-9.9 | 48 | 9.1 |
| >10 | 41 | 7.8 |
| BUN (mg/dL) (516) |
| Normal (<20 mg/dL) | 39 | 7.6 |
| Abnormal (≥20 mg/dL) | 477 | 92.4 |
| Fluid input−urine output (mL) (496) |
| <300 | 177 | 35.8 |
| 300-599 | 91 | 18.3 |
| 600-899 | 75 | 15.1 |
| >900 | 153 | 30.8 |
| Proteinuria (mg/dL) (421) |
| Nil | 84 | 20.0 |
| Trace | 47 | 11.2 |
| 30 mg/dL | 147 | 34.9 |
| 100 mg/dL | 110 | 26.1 |
| >100 mg/dL | 33 | 7.8 |
BUN: Blood urea nitrogen
Aetiology
The causes of AKI among the study population are presented in [Table/Fig-3]. CA-AKI accounted for 70% (n=367) as compared to HA-AKI, which accounted for 30% (n=161) of cases. Among HA-AKI, sepsis 49.2% (n=91) and CKD 11.9% (n=22) were the most common causes, whereas CKD 23% (n=79) and sepsis 22.4% (n=77) were the most frequent causes in CA-AKI.
Aetiologies of acute kidney injury.
AFI: Acute febrile illness; HF: Heart failure; CKD: Chronic kidney disease; UTI: Urinary tract infection; CA-AKI: Community acquired acute kidney injury; HA-AKI: Hospital acquired acute kidney injury
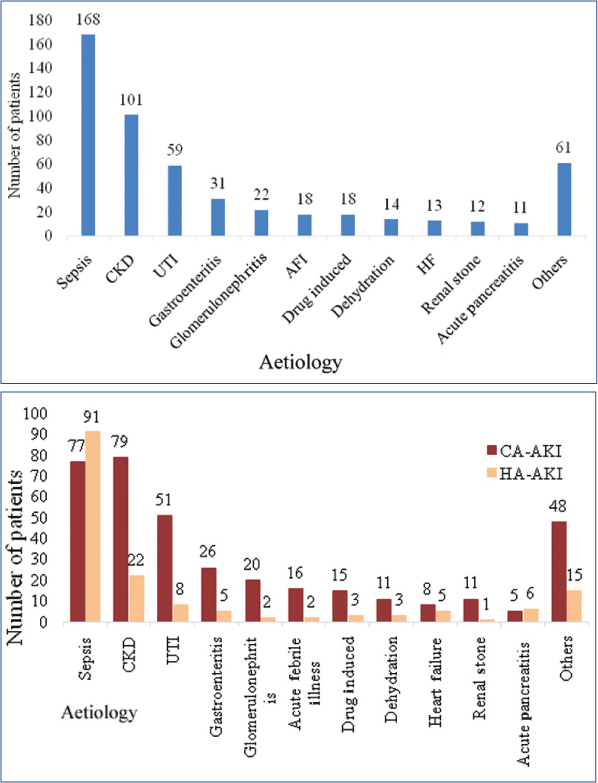
In the various aetiologies observed both in CA-AKI and HA-AKI, sepsis was found to be the most prevalent aetiology of AKI accounting for 31.9% (n=168) of patients, followed by CKD in 19.1% (n=101) of patients. Less frequently observed aetiologies of AKI are shown in the [Table/Fig-3].
Among the conditions known to increase the risk of AKI, DM was seen in 34.7% (n=183) of the patients followed by sepsis 29.5% (n=156) and UTI 15.5% (n=82). Different risk variables of AKI patients were analysed for their association with prognosis of AKI, using multinomial logistic regression. The OR was calculated using univariate and multivariate analysis to analyse the risk factors which can worsen the prognosis of AKI.
Univariate logistic regression analysis revealed that DM, Benign Prostatic Hyperplasia (BPH), UTI and male gender had a statistically significant association with the development of CKD whereas sepsis, CKD, UTI, malignancy and male gender had statistically significant association with death [Table/Fig-4].
Association between risk factors and outcome.
| Outcome | Recoveryn (%) | CKDn (%) | Deathn (%) | Unadjusted OR 95% CI (LL, UL) | Adjusted OR 95% CI (LL, UL) |
|---|
| Risk factor | CKD | Death | CKD | Death |
|---|
| DM | Yes | 110 (32.3%) | 14 (53.8%) | 35 (42.7%) | 2.45*(1.10, 5.47)1 | 1.56(0.96, 2.56)1 | 2.26(0.93, 5.48)1 | 2.13*(1.20, 3.76)1 |
| No | 231 (67.7%) | 12 (46.2%) | 47 (57.3%) |
| CKD | Yes | 58 (16.9%) | 0 (0%) | 6 (7.3%) | - | 0.39*(0.16, 0.94)1 | - | 0.46(0.17, 1.22)1 |
| No | 286 (83.1%) | 83 (100%) | 76 (92.7%) |
| Sepsis | Yes | 65 (19.1%) | 5 (19.2%) | 50 (61.0%) | 1.01(0.37, 2.78)1 | 6.64*(3.95, 11.15)1 | 1.31(0.45, 3.81)1 | 6.37*(3.69, 11.00)1 |
| No | 276 (80.9%) | 21 (80.8%) | 32 (39.0%) |
| GE | Yes | 20 (5.9%) | 1 (3.8%) | 2 (2.4%) | 0.64(0.08, 4.98)1 | 0.40(0.09, 1.75)1 | - | - |
| No | 321 (94.1%) | 25 (96.2%) | 80 (97.6%) |
| Renal stone | Yes | 17 (5.0%) | 3 (11.5%) | 0 (0%) | 2.49(0.68, 9.11)1 | - | - | - |
| No | 324 (95.0%) | 23 (88.5%) | 82 (100%) |
| Surgery | Yes | 39 (11.4%) | 4 (15.4%) | 9 (11%) | 0.70(0.16, 3.11)1 | 0.91(0.42, 1.96)1 | - | - |
| No | 302 (88.6%) | 22 (84.6%) | 73 (89%) |
| BPH | Yes | 12 (3.5%) | 4 (15.4%) | 0 (0%) | 4.99*(1.49, 16.74)1 | - | 1.88(0.44, 8.04)1 | - |
| No | 329 (96.5%) | 22 (84.6%) | 82 (100%) |
| UTI | Yes | 61 (17.9%) | 9 (34.6%) | 4 (4.9%) | 2.43*(1.03, 5.71)1 | 0.24*(0.08, 0.67)1 | 2.01(0.73, 5.60)1 | 0.21*(0.07, 0.63)1 |
| No | 280 (82.1%) | 17 (65.4%) | 78 (95.1%) |
| Malignancy | Yes | 10 (2.9%) | 0 (0%) | 7 (8.5%) | - | 3.09*(1.14, 8.38)1 | - | 2.93(0.96, 8.81)1 |
| No | 331 (97.1%) | 26 (100%) | 75 (91.5%) |
| HF | Yes | 7 (2.1%) | 0 (0%) | 3 (3.7%) | - | 1.81(0.46, 7.16)1 | - | - |
| No | 334 (97.9%) | 26 (100%) | 79 (96.3%) |
| Drugs | Yes | 11 (3.2%) | 2 (7.7%) | 5 (6.1%) | 2.50(0.52, 11.93)1 | 1.95(0.66, 5.77)1 | - | - |
| No | 330 (96.8%) | 24 (92.3%) | 77 (93.9%) |
| Gender | Male | 250 (73.3%) | 24 (92.3%) | 49 (59.8%) | 4.37*(1.01, 18.85)1 | 0.54*(0.33, 0.89)1 | 8.35*(1.08, 64.73)1 | 0.60(0.34, 1.05)1 |
| Female | 91 (26.7%) | 2 (7.7%) | 33 (40.2%) |
| Alcohol | Yes | 73 (36%) | 4 (20%) | 18 (35.3%) | 0.45(0.14, 1.38)1 | 0.97(0.51, 1.85)1 | - | - |
| No | 130 (64%) | 16 (80%) | 33 (64.7%) |
| Smoking | Yes | 52 (25.7%) | 7 (35%) | 12 (23.5%) | 1.55(0.59, 4.10)1 | 0.89(0.43, 1.82)1 | - | - |
| No | 150 (74.3%) | 13 (65%) | 39 (76.5%) |
| BMI | Abnormal | 81 (32.8%) | 7 (35%) | 6 (28.6%) | 1.10(0.42, 2.87)1 | 0.82(0.31, 2.19)1 | - | - |
| Normal | 166 (67.2%) | 13 (65%) | 15 (71.4%) |
| Age | >65 years | 95 (27.9%) | 7 (26.9%) | 27 (32.9%) | 0.95(0.39, 2.24)1 | 1.27(0.76, 2.13)1 | - | - |
| <65 years | 246 (72.1%) | 19 (73.1%) | 55 (67.1%) |
*Statistically significant. Recovery is the reference category
LL: Lower limit; UL: Upper limit; OR: Odds ratio; CKD: Chronic kidney disease; DM: Diabetes mellitus; BPH: Benign prostatic hyperplasia; UTI: Urinary tract infection; HF: Heart failure; GE: Gastroenteritis
Multivariate logistic regression analysis showed that male gender (OR 8.35, 95%CI 1.08-64.73, p=0.042) had statistically significant association with CKD (outcome), whereas DM (OR 2.13 95% CI 1.20-3.76, p=0.010), sepsis (OR 6.37, 95% CI 3.69-11.00, p<0.001) and UTI (OR 0.21, 95% CI 0.07-0.63, p=0.005) had statistically significant association with death (outcome).
Comorbidities
Total 21% (n=112) of patient’s kidney was the sole organ affected at the time of AKI diagnosis. Along with AKI, about 29.2% (n=154) of patients had a single comorbid condition and 26.5% (n=140) had two comorbidities. Cardiovascular system was the most frequently affected non renal system, hypertension was seen in 40.7% (n=215) of patients and DM in 34.7% (n=183) followed by sepsis and UTI in 29.5% (n=156) and 15.5% (n=82) of patients respectively.
Management
Various strategies were observed in the treatment of AKI. Though, fluid replenishment with normal saline was used in 64% (n=336) of the patients. Diuretics were used in 45% (n=238), sodium bicarbonate in 33.9% (n=179), dopamine in 15% (n=80) and calcium gluconate in 6% (n=34) of patients. Haemodialysis was performed in patients with higher concentrations of serum creatinine and was seen in 39% (n=189) of patients.
Outcome
Majority, 64% (n=341) of the patients enrolled for the study got completely recovered from AKI. About 16% (n=82) of the patients died. A total of 5% (n=26) of cases were found to have progressed to CKD and about 15% (n=79) of the patients were not completely recovered but volunteered to get discharged.
Chi square test for association was performed between the different treatment modalities used and the patient outcomes at time of discharge. Statistically significant relationship between treatment and outcomes were found with 5% dextrose-0.9% normal saline χ2=10.96, p<0.05 (n=528), sodium bicarbonate χ2=7.63, p<0.05 (n=528), dopamine χ2=112.14, p<0.05 (n=528), calcium gluconate χ2=17.20, p<0.05 (n=528) and hemodialysis χ2=12.13, p<0.05 (n=528) [Table/Fig-5].
Association between management and outcomes.
| S. no | Treatment | Outcome | p-value |
|---|
| Recovery n (%) | CKDn (%) | Death n (%) |
|---|
| 1. | 0.9% Normal saline | 208 (60.5%) | 13 (56.5%) | 54 (65.9%) | 0.595 |
| 2. | Loop diuretics | 150 (43.6%) | 7 (30.4%) | 42 (51.2%) | 0.178 |
| 3. | 5% Dextrose with 0.9% normal saline | 165 (48%) | 12 (52.2%) | 56 (68.3%) | 0.004* |
| 4. | Sodium bicarbonate | 101 (29.4%) | 10 (43.5%) | 36 (43.9%) | 0.022* |
| 5. | Dopamine | 22 (6.4%) | 0 (0%) | 44 (53.7%) | 0.000* |
| 6. | Calcium gluconate | 13 (3.8%) | 1 (4.3%) | 13 (15.9%) | 0.000* |
| 7. | Dialysis | 111 (32.3%) | 10 (43.5) | 43 (52.4) | 0.002* |
*Statistically significant, p-value was calculated using chi-square test
CKD: Chronic kidney disease
Discussion
As there is a paucity of data on the frequent aetiologies of AKI and its implication in human rights issue, we concentrated on identifying factors which can be modified to reduce the preventable deaths [18]. The median (IQR) age of the present study population was 56 (20) years which was similar with the study conducted by Eswarappa M et al., were the median age was 55.5 years [4]. In a study by Uchino S et al., regarding the epidemiology of AKI in ICU patients, the median age was found as 67 years [19]. The study mentioned above considered only the ICU patients, where most of the patients are elderly thus resulting in an older median age as compared to the present study.
From the present study population, more than a quarter of AKI patients were found to be elderly (>65 years). In a similar Indian study conducted by Eswarappa M et al., majority of patients with AKI belonged to the age group of 61-70 years [4]. Baraldi A et al., conducted a study in Italy on hospitalised patients and observed that patients of age above 65 years had 10 fold greater incidence of AKI compared to their younger population [20]. Wen J et al., conducted their study in Beijing at a tertiary metropolitan hospital on very elderly patients and found a 2.76 % incidence of AKI in age group 65 to 80 years and 14.8% in patients aged ≥80 years [21]. Anatomical and physiological changes that occur in the kidney with advancing age might be the reason for increased incidence of AKI among elderly patients.
The prevalence of AKI in the present study was found to be 8.36 cases /1000 people. A multicentre study by Kerr M et al., conducted at East Kent hospitals, England showed a prevalence of 15.33% [22]. Lower prevalence in the present study may be because the study was conducted in a single site. Under reporting, which resulted due to lack of proper documentation of medical/medication history, lack of proper coding system in developing countries and failure of patients to come for follow-up can be the other reasons for lower prevalence. Study by Prakash J et al., in Indian population showed an increasing incidence of AKI from 1.95 to 4.14 per 1000 admission during 1983-1995 and 1996-2008, respectively [23].
Among the study population, 23.5% were found to be overweight and 7.7% were obese. Billings FT et al., reported that an increase in BMI by 5 kg/m2 increased the risk of AKI by 26.5% [24]. Obese people must be encouraged to reduce their body weight, as obesity is a root cause for multiple medical conditions.
Majority of patients in the present study were asymptomatic and among the symptoms expressed; 23.3% had decreased urine output while 8.7% developed pedal oedema. AKI is usually asymptomatic but some patients may present with decreased urine output. Confirmation of AKI was based on the clinical examination for fluid retention, elevated levels of serum creatinine and BUN.
Median (IQR) serum creatinine among the study population was found to be 4.2 (4.02) mg/dL. Elevated serum creatinine and BUN was found in 92% and 92.4% of patients respectively. Serum creatinine being influenced by various non renal factors (age, gender, body weight, protein intake etc.,) is a poor marker for early detection of renal dysfunction [25].
Proteinuria was found in 68.8% of patients, the values were collected at the time of admission. James MT et al., reported that, presence of proteinuria is a risk factor for AKI. According to them the adjusted risk of admission with AKI was about four times higher in patients with heavy proteinuria than with no proteinuria, (rate ratio 4.4 vs. no proteinuria, 95% CI 3.7-5.2) [26]. Cho K et al., has also observed that patients with proteinuria are two to three times more likely to develop AKI, independent of Estimated Glomerular Filtration Rate (eGFR) [27].
Sepsis was found to be the most prevalent aetiology inducing AKI followed by CKD, UTI. Bagshaw SM et al., in his prospective observational study, conducted on critically ill patients at 54 hospitals in 23 countries found that, sepsis was the leading aetiology for AKI in 47.5% of patients [28]. In a study done by Ali T et al., sepsis was reported as the most common precipitating factor for AKI (47%) [29]. Liano F et al., in his prospective multicentre study, conducted at 13 tertiary care hospitals in Spain reported that, 13% of the patients presenting with AKI had CKD as aetiology [30]. Measures are to be taken to avoid contamination in hospitals and to provide sterile environment in critical care areas.
Adjusted OR for male gender had statistically significant association with CKD (outcome) whereas DM and sepsis had statistically significant association with death (outcome). A similar OR for male gender was observed in a 10-year follow-up study conducted by Yamagata K et al., in which male subject were found to have more than double Hazard Ratios (HR) for developing CKD [31]. The BPH and complicated UTI, which are risk factors for AKI are predominantly observed among male population, this can be the reason for increased risk for CKD among male gender.
Sepsis was found to have a very high odds (OR=6.37) of death among AKI patients. According to Kaul A et al., sepsis has the highest mortality (46%) among the AKI patients [32]. Patients with sepsis have compromised immunity, multiple organ dysfunctions and worsening kidney function. This might be a reason for more number of deaths in AKI patients.
The DM was found to have an OR of 2.13 and showed a statistically significant association with death (outcome). Bagshaw SM et al., reported that DM is a risk factor for End Stage Renal Disease (ESRD) and death regardless of insulin usage [33]. A lot of factors like metabolic syndrome, obesity, hypertension, DM, cardiovascular disease are associated to increase the risk of AKI. These may predispose to AKI and then usually continue to exist even after AKI recovery; leading to CKD and death [34].
Approach for management of AKI involves correction of underlying causes like fluid overload, metabolic acidosis, hyperkalaemia and haematological abnormalities. A systemic review and meta-analysis conducted by Brar SS et al., found that, hydration with sodium bicarbonate was beneficial in patients with AKI [35]. Wald R et al., proposed that an accelerated use of RRT at a lower threshold had many potential benefits in AKI patients [36]. Future studies are needed to assess the efficiency of early dialysis in comparison to conventional practice.
Complete recovery was achieved in majority (64%) of the present patients whereas a few of them volunteered to discharge due to financial and familial problems. About 5% progressed to CKD. Ali T et al., in their study reported similar results with full renal recovery observed in 68% of patients [29]. A similar result with mortality in 16-20% was observed in a study conducted in Indian population by Goldberg R et al., [37]. Sepsis being the most prevalent aetiology of AKI may be the main reason for mortality. Measures are needed to identify and treat sepsis at the earliest.
In a long term follow-up (1-10 years) study conducted by Lafrance JP et al., approximately 19-31% of AKI survivors progressed to CKD [38]. Higher percentage of AKI patients progressing to CKD in the above mentioned study might be due to a long term follow-up (1-10 years) as compared to the present study. More attention must be given to ensure complete renal recovery in order to prevent further kidney damage and worsening of patient’s quality of life.
Limitation
A few limitations of the present study were it was a retrospective study conducted over a short time frame. We had a small sample size which might not be sufficient to establish a clinically significant association between risk factors and outcomes so it can be extrapolated to larger population. Absence of control group to support the association of risk factors.
Conclusion
Prevalence was found to be 8.36 cases per 1000 persons. Most of patients were of age >65 years and showed a male predominance. Sepsis was found to be the most common aetiology for AKI and a risk factor for mortality. DM and UTI were the other risk factors associated with mortality in AKI patients. Male patients with AKI were at risk for progressing to CKD. Among all, one third of the patients required dialysis whereas rest were managed conservatively. Majority of the patients recovered from AKI, 5% progressed to CKD and about 16 % of them died.
AKI patients often do not present symptomatically. Patients of age >65 years and DM should be screened regularly for AKI. It is essential to completely cure or control the underlying cause of AKI in order to prevent undesirable outcomes.