Endometriosis is defined as the presence of functioning endometrial tissue (glands and stroma) outside the uterine cavity. It is an oestrogen dependent, chronic, periodically symptomatic disease affecting 10-30% of women in reproductive age and beyond [1]. It is associated with primary or secondary infertility, in 30% of the women [2]. It is responsible for symptoms including scar tenderness, chronic pelvic pain, dysmenorrhoea, dyspareunia, and subfertility that degrade the quality of life of women significantly, along with productivity loss and healthcare costs [1].
Endometriosis is usually found on the skin especially after obstetric surgical interventions (LSCS scar/episiotomy scar), within the pelvis (ovaries, followed by the pouch of Douglas, uterosacral ligaments, vesicouterine pouch, urinary bladder, outer surface of the uterus, fallopian tubes, round ligament, and rectovaginal septum), abdominal cavity (recto-sigmoid colon, appendix, caecum, distal ileum and omentum), and rarely outside (lungs/brain) [2]. Though endometriosis is a common disease, we want to emphasise the role of FNAC in diagnosing scar endometriosis, in the present era of increased LSCSs. We want to add few cases of extra genital endometriosis involving urinary bladder and sigmoid colon to the existing literature. The aim of the study was to review the cases of endometriosis with varied clinical presentation having one underlying disease, and add to the existing literature.
Materials and Methods
It was a retrospective study including all the cases diagnosed as endometriosis (either by FNAC or histopathology) in the Department of Pathology at Alluri Sitarama Raju Academy of Medical Sciences, Eluru, Andhra Pradesh, India were included in the study from the period January 2012 to November 2017. This research project was approved by the Institutional Ethical Committee of the institute (IEC/ASR/017/2017).
Statistical Analysis
Data were compiled by using Microsoft excel sheet 2010 (Microsoft Corporation, Seattle, WA, United States). Simple descriptive statistics were used and data represented in the form of tables, proportions by using pie diagrams and bar charts wherever necessary.
Results
As shown in [Table/Fig-1] a total of 29 cases with the diagnosis of endometriosis were found. The patient’s age ranged from 20 to 45 years of age, with mean age of 29.4±7.7 and median of 28.8 years. Out of 29 cases, 16 cases were diagnosed as scar endometriosis involving anterior abdominal wall at LSCS scar site (56.25% by FNAC and 43.75% by histopathology), 10 cases were diagnosed as ovarian endometriosis (bilateral in one case), two cases of urinary bladder endometriosis and one case of bowel endometriosis involving sigmoid colon. All the cases are tabulated in [Table/Fig-1].
Endometriosis cases at various sites of involement.
| Sl. no | Age | Clinical findings | Site of involvement | Diagnosis |
|---|
| 1 | 36 | P2L1D1 with cyclical pain abdomen at LSCS scar site | FNAC done from LSCS scar site | Scar endometriosis |
| 2 | 25 | P2L2 with cyclical pain abdomen at LSCS scar site | FNAC done from LSCS scar site | Scar endometriosis |
| 3 | 29 | P1L1-Lump at lateral end of LSCS scar site with cyclical pain | FNAC from nodule at lateral end of LSCS scar | Scar endometriosis |
| 4 | 20 | G3A1L2-Lump over the LSCS scar site | FNAC done from LSCS scar site | Scar endometrioma |
| 5 | 28 | Mass per abdomen below umbilical region since 6 months, associated with cyclical pain and bleeding through the sinus over the swelling during menstruation | Excised swelling at LSCS scar site in right iliac fossa | Scar endometriosis |
| 6 | 25 | P2L2-Lump over the LSCS scar site, cyclical bloody discharging sinus at the scar site, and pain | FNAC done from LSCS scar site | Scar endometriosis |
| 7 | 30 | Mass over the anterior abdominal wall at LSCS scar site, cyclical pain | Excised soft tissue mass from anterior abdominal wall LSCS scar site | Scar endometriosis |
| 8 | 25 | P1L1-Subcutaneous mass just below the LSCS scar line associated with cyclical pain during menstruation | FNAC done from subcutaneous mass in the left lower anterior abdominal wall below the LSCS scar line | Scar endometrioma |
| 9 | 28 | Mass and cyclical pain at LSCS scar site | Excised soft tissue mass of size 6×5×3 cm from anterior abdominal wall at LSCS scar site with grey white whorled cut appearance | Scar endometriosis |
| 10 | 30 | Mass arising at the LSCS suture line | Excised soft tissue mass of size 3×2×2 cm from anterior abdominal wall at LSCS scar site with grey white cut surface | Scar endometriosis |
| 11 | 32 | Mass over the anterior abdominal wall at LSCS scar site, associated with cyclical pain | Excised soft tissue mass of size 3×2×2 cm from anterior abdominal wall at LSCS scar site with grey brown cut surface | Scar endometriosis |
| 12 | 28 | Mass over the anterior abdominal wall at LSCS scar site, associated with cyclical pain | Excised soft tissue mass of size 3×2×2 cm from anterior abdominal wall at LSCS scar site with grey white cut surface | Scar endometriosis |
| 13 | 23 | P1L1 with swelling below the umbilicus since 6 months.Associated with cyclical pain during menstruation.LSCS has done 3 years back. | FNAC done anterior abdominal wall swelling below the umbilicus | Scar endometrioma |
| 14 | 23 | P1L1 with small swelling at LSCS scar site in the RIF since 1 year. LSCS has done 2 years back. | FNAC done from subcutaneous swelling in RIF beneath the LSCS scar site | Scar endometriosis |
| 15 | 24 | P1L1 with mass in the left anterior abdominal wall above suture line since 6 months.Associated with cyclical pain during menstruation. | FNAC done from swelling in the left iliac fossa LSCS scar site | Scar endometrioma |
| 16 | 30 | Swelling in the anterior abdominal wall just above the LSCS scar site since 1 year. LSCS has done 4 years back. | Excised mass from anterior abdominal wall on previous LSCS scar site | Scar endometriosis |
| 17 | 41 | Pain in the left iliac fossa since 1 dayUSG-Left ovarian cyst | Excised left salpingo-oophorectomy specimen | Ovarian endometriosis |
| 18 | 25 | Pain abdomen x3 daysUSG-Left ovarian cyst | Left ovarian cyst of size 6×4×4 cm | Ovarian endometriosis with secondary fibrosis |
| 19 | 40 | Acute pain abdomen | Left salpingo-oophorectomy specimen | Ovarian endometriosis with torsion |
| 20 | 45 | Abnormal uterine bleeding and dysmenorrhoea | Total abdominal hysterectomy+bilateral salpingo-oophorectomy | Bilateral ovarian endometriosis |
| 21 | 27 | Lower pain abdomen, USG-Left ovarian cyst | Left salpingo-oophorectomy | Ovarian endometriosis (chocolate cyst) |
| 22 | 28 | Pain abdomen, USG-Right ovarian cyst | Right ovarian cyst of size 6×4×4 cm | Ovarian endometriosis (chocolate cyst) |
| 23 | 23 | Abnormal uterine bleeding and dysmenorrhoeaUSG-Right ovarian cyst | 4×3×3 cm grey brown right ovarian cystic mass | Ovarian endometriosis (chocolate cyst) |
| 24 | 25 | Lower pain abdomenUSG-Left ovarian cyst | 3×2×2 cm flap like grey-white to dark brown cut open cystic mass | Ovarian endometriosis (chocolate cyst) |
| 25 | 30 | Pain in the left iliac fossa USG-Left ovarian cyst | 6x4x4 cm flap like grey-white to dark brown cystic mass | Ovarian endometriosis (chocolate cyst) |
| 26 | 27 | Pain abdomen USG-Complex right ovarian cyst | 6×4×3 cm grey-white cystic mass with dark brown cut surface | Ovarian endometriosis (chocolate cyst) |
| 27 | 40 | Abnormal uterine bleeding with cyclical haematuria | Urinary bladder mass | Endometriosis of urinary bladder |
| 28 | 27 | Cyclical haematuria and dysmenorrhoea | Total abdominal hysterectomy+bilateral salpingo-oophorectomy and part of urinary bladder | Endometriosis of urinary bladder |
| 29 | 40 | Pain abdomen since 3 days, Constipation for 2 days | Sigmoid colon with exophytic growth | Endometriosis of sigmoid colon |
G: Gravida; P: Para; L: Live; A: Abortion; D: Death; USG: Ultra sonogram
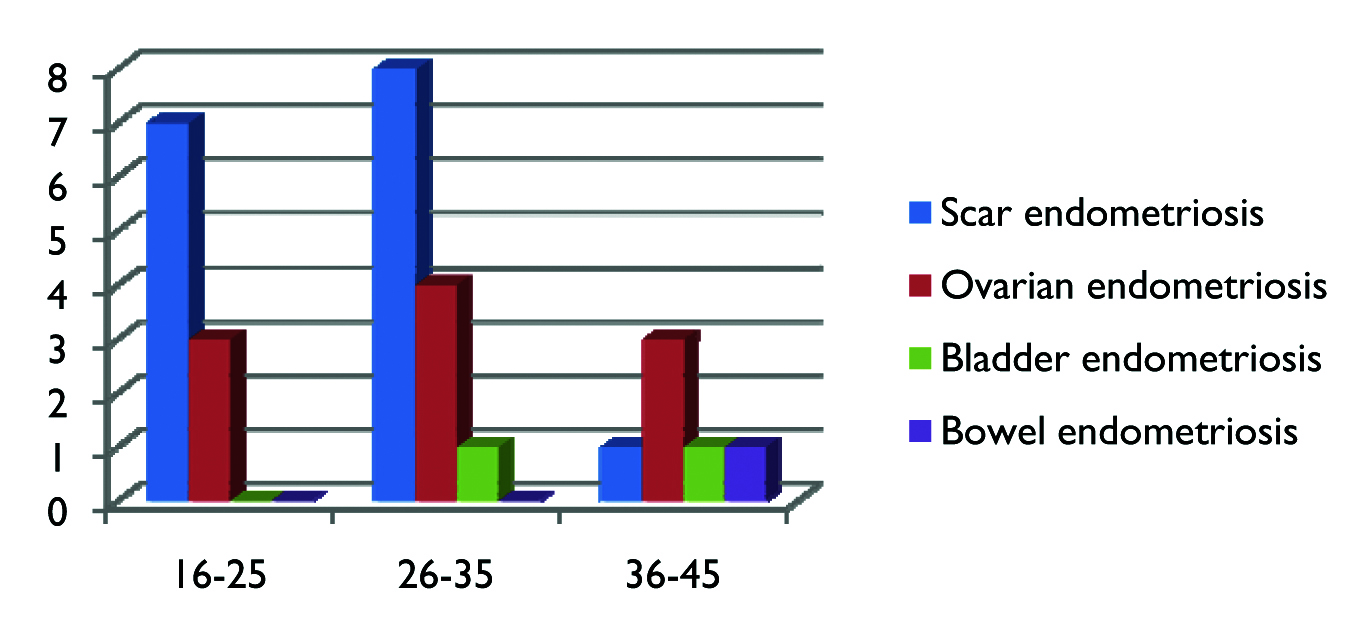
As depicted in [Table/Fig-2] scar endometriosis was the most common entity in the age group between 20-35 years followed by ovarian endometriosis. In the age group between 36-45 years ovarian endometriosis was more common than scar/bladder/bowel endometriosis. We have divided patients into three age groups because of impact of endometriosis on education in the age group of 16-25 years, employment for the age group of 26-35 years; and financial impact for those 36 years and above.
Bar chart depicting age wise distribution of cases with endometriosis.
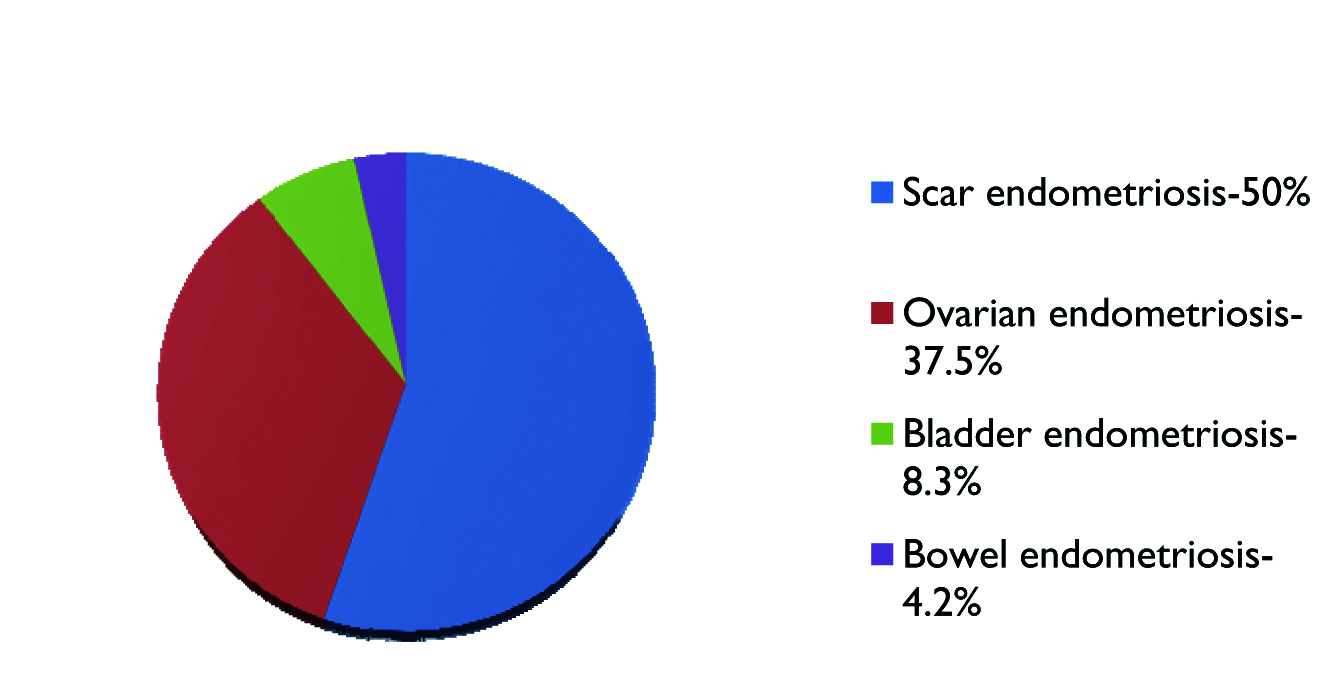
As depicted in [Table/Fig-3], scar endometriosis is the most common entity in the present study, constituting 50% of the cases occurring exclusively at the LSCS scar site. Out of 16 cases, 14 were (87.5%) presented with mass in the LSCS scar and diagnosed by preoperative FNAC in 9 cases (56.25%) [Table/Fig-4,5] and by histopathological examination of excised specimens in 7 cases (43.75%) [Table/Fig-6]. Ovarian endometriosis was the second common entity, constituting 37.5% of cases, of which one case is bilateral and one shows torsion [Table/Fig-7]. Two cases of urinary bladder endometriosis constituting 8.3% [Table/Fig-8] and only one case of bowel endometriosis involving sigmoid colon constituting 4.2% of cases [Table/Fig-9,10].
Pie chart depicting proportions of various types of endometriosis.
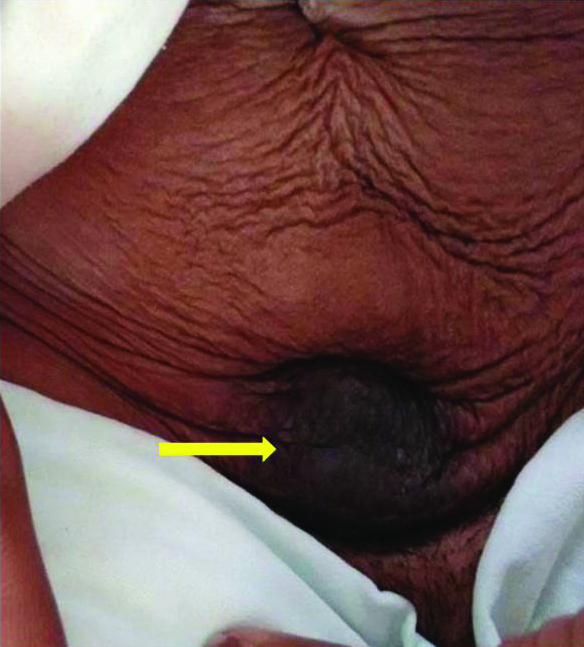
Photograph of scar endometriosis showing dark pigmented lump (yellow arrow) in the anterior abdominal wall just above the lower segment caesarean section scar site.
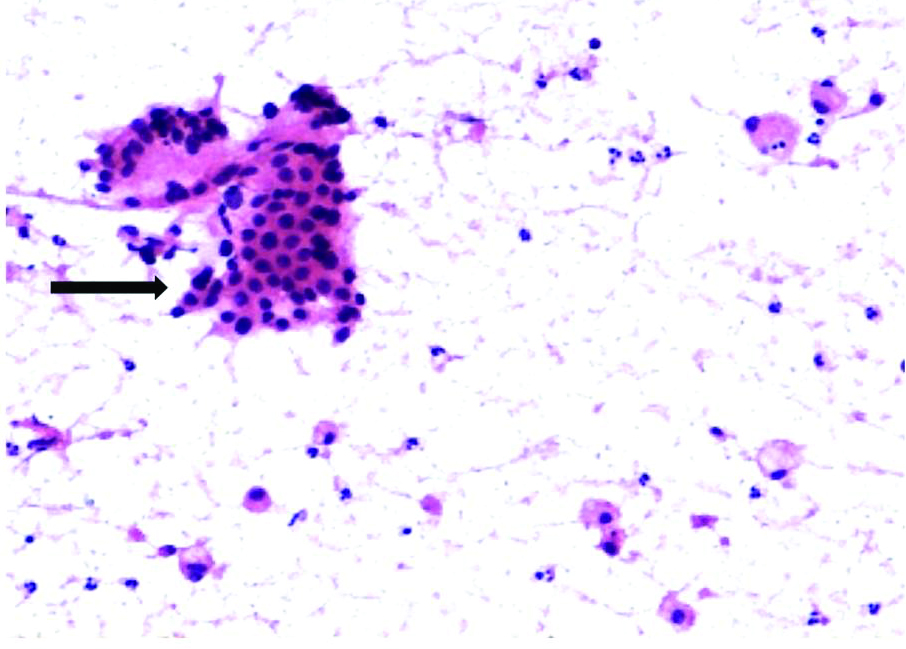
FNAC of scar endometriosis: Photomicrograph showing monolayer sheets of round to oval epithelial cells (black arrow) admixed with spindle shaped stromal cells and scattered foamy macrophages (H&E, 10X).
Photomicrograph of scar endometriosis showing endometrial glands (black arrow) and stroma with in the fibrous tissue (H&E, 4X).

Photomicrograph of ovarian endometriosis with torsion showing endometrial glands (yellow arrow) with in the ovarian stroma showing interstitial oedema and haemorrhage. Note sub nuclear vacoulations (H&E, 10X).

Photomicrograph of bladder endometriosis showing endometrial glands and stroma within the detrusor muscle of the urinary bladder. Glandular lumen is filled with hemosiderin laden macrophages (yellow arrow) (H&E, 4X).

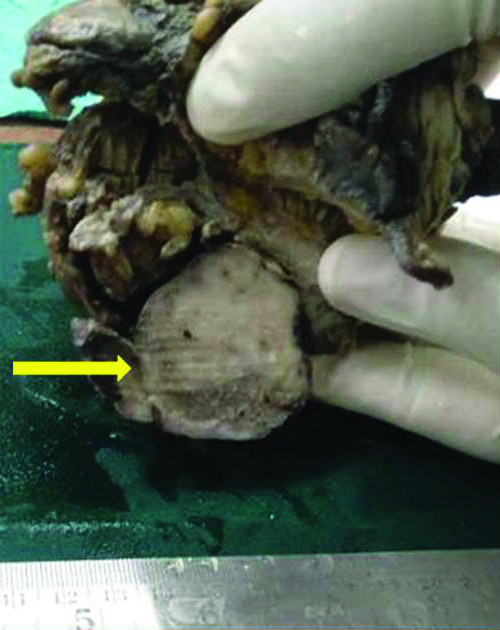
Gross photograph of sigmoid colon with growth (yellow arrow) on the serosal aspect.
Photomicrograph of bowel endometriosis showing endometrial glands and stroma within the muscularis propria (yellow arrow) of the sigmoid colon (H&E, 4X).

Discussion
Endometriosis is a condition in which endometrial tissue, composed of both endometrial glands and stroma, is found at sites outside the uterine cavity. In 1860, Carl von Rokitansky provided the first identification and detailed description of endometriosis. In 1894, Breus used the term “chocolate cyst” for the first time. In 1903, Runge provided detailed description of endometriomas, and R Meyer described endometriosis in an abdominal scar and later described intestinal endometriosis [2].
Risk factors significantly associated with endometriosis include gravidity, family history of endometriosis, pelvic surgery, dysmenorrhoea, pelvic pain, dyspareunia, premenstrual spotting, fatigue, diarrhoea and history of galactorrhoea. However, parity was negatively related to endometriosis. Increasing age, alcohol use, low body weight, early menarche, prolonged menstrual flow, and short cycle interval, intercourse during menses, infertility are also alleged risk factors [3].
Many theories have been proposed to explain the pathogenesis of endometriosis which includes: 1) Transplantation of endometrial fragments to ectopic sites through the fallopian tubes into the pelvis (retrograde menstruation) or by lymphatic, hematogeneous dissemination or mechanical dissemination (perineum or laparotomy scars); 2) Metaplasia of the multipotential celomic peritoneum, embryonic remnants; 3) Induction of undifferentiated mesenchyme in ectopic sites to form endometriotic tissues after exposure to substances released from shed endometrium; 4) Hormones such as oestrogen-driven proliferation of endometrial lesions; 5) Oxidative stress and inflammation by recruitment of immune cells and their production of cytokines that promote endometrial growth; 6) Immune dysfunction; 7) Apoptosis suppression; 8) Genetic; and 9) Stem cells. Above theories implicated in the pathogenesis of endometriosis indicate that the aetiology of endometriosis is complex and multifactorial, involving hormonal, genetic, immune, and environmental components [4].
Most common symptoms of endometriosis are dysmenorrhoea (during and at the end of menstruation), dyspareunia, chronic pelvic pain, and infertility [2]. Causes of infertility include pelvic anatomy distortion due to major pelvic or peritubal adhesions, endocrine and ovulatory abnormalities, altered peritoneal function with increased peritoneal fluid volume, and altered hormonal and cell-mediated functions in the endometrium which are affecting embryo implantation [5,6].
Scar endometriosis is the most common entity in the present study, constituting 50% of the cases occurring exclusively at the LSCS scar site. In an exclusive study of scar endometriosis done by Vellido-Cotelo R et al., endometriosis is seen at caesarean scar in 82% of cases, at episiotomy scar in 11.7% cases and at laparoscopic surgery port, 5.8% cases [7]. Increased obstetric surgical interventions are one of the most important risk factors for scar endometriosis due to direct mechanical implantation of endometrial tissue [8,9].
FNAC helps in confirming preoperative diagnosis of scar endometriosis, in clinically doubtful cases. Cytological features of scar endometriosis vary with cyclical hormonal changes. In proliferative phase, epithelial cells are small, uniform with scant cytoplasm, round to ovoid nuclei with bland chromatin and arranged in cohesive sheets along with spindle shaped stromal cells and hemosiderin laden macrophages. During secretory phase, epithelial cells gradually increase in size with cytoplasmic microvacuolations and stromal cells may have epithelioid appearance with abundant cytoplasm (predecidual change). Background usually contains inflammatory cells and hemosiderin laden macrophages [10]. The presence of any two of the three components (endometrial glands, stromal cells and hemosiderin laden macrophages) are required for the diagnosis of endometriosis. The importance of FNAC lies in excluding other lesions like abscess, suture granuloma, hematoma, desmoid tumour, nodular and proliferative fasciitis or primary and metastatic cancer [11]. So far, 23 cases of clear cell endometrial carcinoma arising from endometriosis foci located within a caesarean section scar were reported in the literature [12].
Ovarian endometriosis constitutes 37.5% of cases, which is similar to study done by de Paula Andres M et al., in adolescent women with pelvic pain [13]. Early diagnosis and intervention of cystic endometriomas in adolescent or young women with chronic pelvic pain will prevent detrimental effect on the ovarian follicular reserve. A 2-dimensional and 3-dimensional transvaginal ultrasound, transvaginal endoscopy and transvaginal hydrolaparoscopy are helpful in the early diagnosis. Transvaginal laparoscopy has been shown to be most effective in ablating endometriomas with a maximum diameter of 3 cm [14].
Ovary is the most common site for malignant transformation of endometriosis with endometrioid/clear cell carcinoma morphology, occurring in less than 1% of women [5]. Severe atypia in the glandular epithelium of ovarian endometriotic cysts may be associated with aneuploidy and may progress to invasive epithelial malignancy [15].
Urinary tract involvement in women with endometriosis is between 1% and 5%, with the bladder most often affected, followed by ureter, kidney and urethra [16,17]. Urinary bladder endometriosis constitutes 8.3% of cases in present study, which is slightly higher than reported [16,17]. Few documented cases of endometrioid endometrial carcinoma of the urinary bladder and endometrioid adenosarcoma of the bladder arising from endometriosis were reported [18].
Gastrointestinal tract involvement is known to occur in 3-37% of the cases with endometriosis and the recto-sigmoid colon is the most common site of involvement [19]. In present study, only one case of bowel endometriosis involving sigmoid colon was seen constituting 4.2%. While evaluating mucosal biopsies in female patients, a high index of suspicion is required for diagnosing intestinal endometriosis with mucosal involvement [20]. Similar to native endometrium, ectopic endometrial tissue in endometriosis cases can develop hyperplasia, dysplasia, endometrioid adenocarcinoma and carcinosarcoma. Endometrioid adenocarcinoma of bowel has to be differentiated from colorectal adenocarcinoma [21].
Malignant transformation was not seen in any of the case in our study. We lost follow-up of most of the surgically cured endometriosis cases and need more data on bladder and bowel endometriosis.
Conclusion
Endometriosis is a chronic disease and the symptoms vary depending on the organ involved but not the disease itself. FNAC is helpful for preoperative diagnosis of scar endometriosis. A high index of suspicion is required for early diagnosis and intervention of ovarian, bladder and intestinal endometriosis, to provide better quality of life and to reduce infertility.
G: Gravida; P: Para; L: Live; A: Abortion; D: Death; USG: Ultra sonogram