Nephrocalcinosis is a disorder of increased calcium deposition in the renal parenchyma. It is a complication of various renal tubular disorders, vitamin D excess, metabolic disorders, prematurity or drug application [1].
Increasing number of patients diagnosed with NC are being reported due to higher usage of renal ultrasound as a routine diagnostic procedure. Specific therapy must be initiated for the underlying cause of NC; hence, the right diagnosis is of crucial importance [2].
The study aimed to present the aetiology and clinical presentation of NC in children, and to evaluate their growth and renal function during follow up.
Materials and Methods
Twenty-five children with bilateral NC diagnosed by ultrasonography between 1999 and 2016 in the Department of Paediatric Nephrology were included in the study. Study was approved by local Ethics Committee and informed consents were obtained.
Patients had been admitted to the department for various reasons such as failure to thrive, polyuria, polydipsia, vomiting, Urinary Tract Infection (UTI) and fever. Demographic and clinical features including age, sex, family and birth history, somatic and psychomotor development and relevant laboratory values were extracted from patients’ records and then retrospectively analysed.
In children older than 12 months, Schwartz formula was used to calculate Glomerular Filtration Rate (GFR), and a value above 90 mL/min/1.73 m2 was considered normal; in infants up to 12 months of age, age-specific values for serum creatinine to evaluate renal function were used [3,4]. Hypercalciuria was defined based on the urinary calcium to creatinine ratio (Ca/cr) values according to Matos V et al., in the second morning urine sample or as urinary calcium excretion above 4 mg/kg in 24 hours collected samples [5].
Diagnosis of the cause leading to NC was made through the following criteria: dRTA was diagnosed when a positive urinary anion gap and high urinary pH (>5.5) was associated with hyperchloremic metabolic acidosis (pH <7.35, bicarbonate <18 mEq/L); vitamin D intoxication when corresponding clinical history data were associated with hypercalcaemic (>11 mg/dL) hypercalciuria and low parathyroid hormone as well as high plasma 25-hydroxy vitamin D (>90 ng/mL); primary hyperoxaluria in patients with increased urinary oxalate excretion (>40 mg/1.73 m2/day) without any history of malabsorption; Bartter syndrome in patients with metabolic alkalosis (pH >7.45, bicarbonate >25 mEq/L), hypokalemia (<3.5 mEq/L), urinary wasting of potassium (>20 mEq/L) and chloride (>30 mEq/L) with normal blood pressure; primary hypomagnesemia with hypercalciuria when low serum magnesium was found in association with urinary magnesium wasting and hypercalciuria and IH in patients with increased urinary calcium, normal blood calcium level (9-11 mg/dL) after ruling out aforementioned tubular defects [6]. Systematic hearing screening was conducted in all patients diagnosed with dRTA.
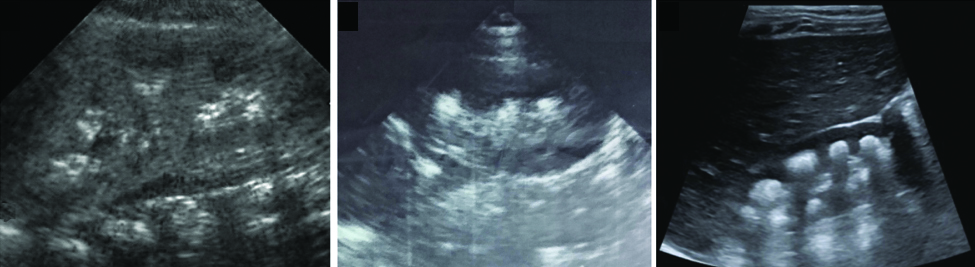
Ultrasonography was used to grade NC as follows: Grade I-slightly increase of medullary pyramids echogenicity; Grade II-diffuse increase of medullary pyramids echogenicity in the absence of acoustic shadowing; and Grade III-diffuse and more uniform increase of medullary pyramids echogenicity associated with acoustic shadowing [Table/Fig-1] [7].
Ultrasound images corresponding to Grades I, II, and III of nephrocalcinosis (left to right), respectively.
Patients diagnosed with dRTA were treated with alkali supplements and potassium citrate. Patients with IH beside dietary modifications were treated with thiazide diuretics and potassium citrate. In patient intoxicated with vitamin D, the same was withdrawn and appropriately rehydrated. Patient with hyperoxaluria was treated with vitamin B6.
Statistical Analysis
Statistical analyses were performed using SPSS for Windows version 21.0 (SPSS, Inc., Chicago, IL). Normally distributed continuous data were expressed as mean±standard deviation. Otherwise, data were presented as median (range). Student paired t-test was used to compare data of first and last examination, while paired-sample Wilcoxon signed rank test was used as a nonparametric alternative. However, when comparison was not applicable due to maturational changes, data were compared to normal range of values using one-sample t-test. One-way ANOVA was used to determine the effect of NC grade on the renal function and growth. A p-value less than 0.05 was considered as statistically significant.
Results
The median age at diagnosis was 9 (3-84) months. Out of 25 patients 17 (68%) were males. The most common clinical features at presentation were failure to thrive in 8 (32%) patients; polyuria and polydipsia in 5 (20%) patients; haematuria and renal colic in 2 (8%) patients; 7 (28%) patients manifesting vomiting and/or fever were diagnosed to have UTI. In 3 (12%) cases NC was found accidentally during routine check up of children in paediatric clinic for other reasons. Positive family history for nephrolithiasis was evident in three patients (two dRTA and one IH). All patients were born on term.
Grade I NC was found in nine patients, while Grade II and Grade III in 13 and three patients, respectively. All patients had normal renal function: urea=5.8±1.2 mmol/L, creatinine=65±4.9 μmol/L and estimated GFR=101 (68-151) mL/min/1.73 m2. Mean calcium excretion was 3.76±1.51 mg/kg/day and Ca/cr ratio was 0.55 (0.06-2.2); 12 (48%) patients had hypercalciuria.
Distal renal tubular acidosis composed the largest group of patients (64%), with 11 males and five females. The median age at presentation was 6 (3-24) months. Failure to thrive was the main complaint in 44% of patients, while symptomatic polyuria and polydipsia were present in 25% of patients. The other 31% were found after diagnosis of UTI or accidentally. The median height z-score was -1.72 (-3.5 to 0.5). Laboratory values were as follows: blood pH 7.2 (6.9-7.3), bicarbonates 13.6 (8.1-16.7) mEq/L, potassium 2.9 (2.4-3.9) mEq/L, sodium 134 (126-143) mEq/L, chloride 117 (113-122) mEq/L. Citrates were low in patients with dRTA (110.6±48.2 mg/g creatinine). Grade I NC was found in six dRTA patients, while Grade II and III in eight and two patients, respectively. Hearing loss was evident in five patients (four boys and one girl) including two pairs of siblings (all boys).
Hypervitaminosis D (173 ng/mL) with hypercalcaemia (12.4 mg/dL), hypercalciuria (5.1 mg/kg/day) and Grade II NC was found in one patient with history of excess usage of vitamin D, presenting with polyuria and polydipsia. In addition, hyperoxaluria (54 mg/1.73 m2/day) as a cause of NC was also found in one patient with Grade II NC, presenting with renal colic and haematuria. IH was diagnosed in two patients, one of them had UTI, while the other one was found accidentally. Unfortunately, the aetiology of NC remained unknown in 5 (20%) patients [Table/Fig-2].
Causes of nephrocalcinosis in 25 children.
| Aetiology | N=25 (100%) |
|---|
| dRTA | 16 (64%) |
| Idiopathic hypercalciuria | 2 (8%) |
| Primary hyperoxaluria | 1 (4%) |
| Hypervitaminosis D | 1 (4%) |
| Unknown | 5 (20%) |
dRTA: distal renal tubular acidosis
Follow Up
Only 13 (52%) patients (nine dRTA, two IH, one hyperoxaluria and one hypervitaminosis D) were followed for a median duration of 99 (33-189) months. We lost contact with the other half because of the 1998-99 armed conflict in Kosovo or unknown reasons.
Of these 13 patients who were followed, 10 (77%) were boys. The median age at the diagnosis and last examination was 6.5 (3-84) and 126 (36-192) months, respectively. Height z-scores improved from −1.75 (−3.1 to 0.8) to a median of −1.14 (−2.6 to 0.8) (p<0.05). Height z-scores below −2 were noticed in 5 (42%) patients (four dRTA and one hyperoxaluria) at first examination, while 3 (25%) of them (two dRTA and one hyperoxaluria) remained growth retarded at the last examination. None of the patients received growth hormone treatment. Mean/median values of urea (6.55±0.49 mmol/L), creatinine (77±5.6 μmol/L) and GFR (98 (42-132) mL/min/1.73 m2) were still preserved; however, subnormal renal function was observed in three patients. Likewise, slightly below normal GFR values were noticed in two dRTA patients, while a remarkable decline was observed in the patient suffering from hyperoxaluria. On the other hand, improvement was noticed in the patient with vitamin D intoxication. Hypercalciuria in the first and last examination was present in 5 (42%) and 3 (25%) patients, respectively [Table/Fig-3]. The degree of NC increased in three patients, remained the same in seven and decreased in three patients. However, the NC grade change did not significantly affect renal function and growth {F (2,10)=0.682, p=0.52 and F (2,10)=1.077, p=0.37}, respectively. Furthermore, four (two pair of siblings) out of five patients diagnosed with hearing loss showed further deterioration of hearing and hearing prosthesis were needed.
Clinical and laboratory data at first and last examination.
| Parameter | All patients | Follow up |
|---|
| Presentation N=25 | Presentation N=13 | Follow upN=13 |
|---|
| Age (months) | 9 (3-84) | 6.5 (3-84) | 126 (36-192) |
| Weight z-score | −1.90 (−3.7 to 1.8) | −1.85 (−3.3 to 1.8) | −0.32 (−1.3 to 2.9)* |
| Height z-score | −1.88 (−3.5 to 1.2) | −1.75 (−3.1 to 0.8) | −1.14 (−2.6 to 0.8)* |
| Urea (mmol/L) | 5.8±1.2 | 5.7±0.91 | 6.55±0.49 |
| Creatinine (μmol/L) | 65±4.9 | 71±6.5 | 77±5.6 |
| Urinary Calcium (mg/kg/day) | 3.76±1.51 | 3.11±0.98 | 2.81±1.31 |
| GFR (mL/min/1.73 m2) | 101 (68-151) | 103 (72-144) | 98 (42-132) |
*p<0.05 compared to presentation
Discussion
There are two processes believed to be of crucial importance for NC development; formation of crystals in the renal tubules and their deposition in the distal tubule. Decreased urinary volume and/or increased lithogenic factors favours the development of crystals in the renal tubules. However, intact epithelium and tubular transport mechanisms as well as crystal deposition inhibitors such as magnesium, citrates and proteins, enable passage of supersaturated urine at a certain level or time duration [8,9]. Absence of these protective mechanisms lead to various disorders favouring crystal retention and hence NC development.
In the present study, dRTA was the main cause of NC (64%). Metabolic acidosis through decreased excretion of fixed acids and positive H+ balance, leading to hypercalciuria, is believed to be the main underlying mechanism. Most of the previous studies have also reported dRTA as the leading cause of NC, with an incidence range from 30.5-61.7% [7,10-12]. However, age at diagnosis was lower in the present study compared to other studies as all had presented before 24 months of age [10,11].
IH was diagnosed after excluding excessive vitamin D and/or dietary intake of calcium, as secondary causes of hypercalcaemia. One of the two patients with IH presented with renal colic while the other one was asymptomatic. The lower number of IH patients in our series could be justified by low ratio of ultrasound examination in children without symptoms in Kosovo.
Patient with hyperoxaluria progressed to renal insufficiency. Likewise, a worse prognosis of patients with NC due to hyperoxaluria as an underlying cause has been previously observed [13].
Excess usage of vitamin D was found only in one patient, perhaps due to insufficient usage of vitamin D in Kosovo in the past decade.
In 20% of patients the cause for NC remained unknown.
In earlier studies, failure to thrive/growth retardation was the most common manifestation. Rönnefarth G et al., in a multicenter German study found that 41% and 32% of children had growth failure at first and last examination, respectively [7]. Only patients with IH showed significant growth improvement. Mantan M et al., found that most of the Indian patients present with growth failure and no significant improvement was noticed at the end of the study [11]. Dogan CS et al., and Ammenti A et al., found almost half of patients presenting with growth failure, while half of them improved during the study period [10,14]. In the present study significant improvement was observed; however, three patients still had height z-score < -2 at the last examination.
In addition, two multicenter retrospective studies reported decreased GFR in 35% and 41% of patients in the first examination, while in the last examination 17.5% and 29% of patients had decreased GFR, respectively [7,14]. An Indian study reported a significant decrease of GFR during the follow up [11], while Dogan CS et al., showed increasing number of patients from one to five in the last examination with decreased GFR [10]. At the time of diagnosis all our patients had normal GFR, reflecting perhaps the importance of early age at diagnosis. In the last examination 3 (23%) patients (two dRTA and one hyperoxaluria) showed subnormal values of GFR. Inability to follow up half of the patients might suggest that patients with progressing disorders addressed more advanced centers; hence, our better results compared to other studies.
It is important to mention that growth and renal function were not affected by the progression of NC.
Moreover, hypercalciuria is considered a major predisposing factor for NC development, accounting for 42-92.2% of cases [7-9]. The results of the present study belonged to the lower limit of such range (48%). However, NC in patients with dRTA can occur without hypercalciuria. Likewise, reduced urinary citrate level, together with metabolic acidosis and persistently alkaline urine might have induced the development of NC.
Urinary tract infecion was found in seven patients. Excessive calcium excretion harming uroepithelial cells eases bacterial adherence; hence, the higher occurance of UTI among patients with hypercalciuria (six out of seven).
Hearing loss among two pairs of siblings with dRTA, might suggest that genetic transmission of mutant gene (ATP6V1B1) may also be responsible for the high incidence of dRTA. ATP6V1B1 encodes the B1 subunit of H+-ATPase proton pump that normally maintains the cochlear endolymph pH at 7.4 [15].
Limitation
The present study had several limitations. Follow up of only half of patients constitutes a major limitation. Moreover, bias may be considered because of long-term follow up, with examinations conducted by different ultrasound equipment and different observers. Finally, limited resources might be responsible for the high percentage of patients with unknown aetiology.
Conclusion
Distal renal tubular acidosis is the major cause of NC in children of Kosovo. Failure to thrive was the most frequent presenting feature. Growth and renal function depends on the underlying cause and the age at diagnosis but not on the degree on NC. Likewise, initiation of specific therapy at an earlier age prevents worsening of renal damage. Hence, more frequent ultrasound screening for renal and metabolic disorders are welcomed. Larger studies with long-term follow up are required to further understand and evaluate renal function in patients with NC.
dRTA: distal renal tubular acidosis