Adrenal Gland Haemangioma Mimicking a Non-functional Pheochromocytoma
Gwendolyn Fernandes1, Munde Shital2, Sanjeet Kumar3
1 Associate Professor, Department of Pathology, Seth G.S. Medical College and K.E.M. Hospital, Mumbai, Maharashtra, India.
2 Fellow, Department of Pathology, Seth G.S. Medical College and K.E.M. Hospital, Mumbai, Maharashtra, India.
3 Senior Resident, Department of Endocrinology, Seth G.S. Medical College and K.E.M. Hospital, Mumbai, Maharashtra, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Gwendolyn Fernandes, C-802, Swayam, Poonam Gardens, Mira Road, Mumbai-401107, Maharashtra, India.
E-mail: drgwenfern@yahoo.co.in
Haemangiomas of adrenal gland are rare, non-functioning benign tumours with less than 100 cases documented in literature. They are difficult to diagnose preoperatively and are often clinically mistaken for other benign adrenal lesions like myelolipoma, ganglioneuroma and non-functional pheochromocytoma. We report a case of a 38-year-old male with haemangioma of the adrenal gland which presented exclusively with pain in the right side of the abdomen.
Adrenal tumours, Histopathology, Incidentalomas
Case Report
A 38-year-old male farmer, presented with episodes of acute pain in the right side of the abdomen for six months. There was a past history of right sided renal calculous six years ago. Ultrasonography had revealed a 9 mm calculous in the pelvicalyceal system. No other abnormality was noted. The patient was put on active medical expulsive therapy with ketorolac, prednisolone and antibiotics following which the calculous was passed per urethra. There was no history of paroxysms of headache, palpitation, and diaphoresis. No cushingoid features were seen. His pulse was 98/minute and blood pressure was 120/80 mmHg. Laboratory investigations were within normal range. Plasma Free Normetanephrine (PFNMN) was 106 pg/mL, Plasma Free Metanephrine (PFMN) was 12.9 pg/mL and overnight dexamethasone suppression test with cortisol of 0.83 ug/dL, which were normal.
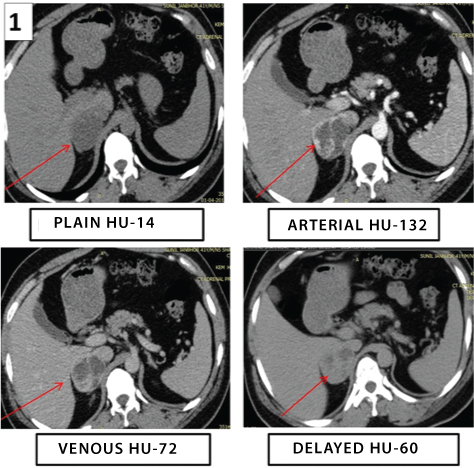
Ultrasonography revealed a 4.3x4.1 cm sized right suprarenal mass with solid-cystic structure. The kidneys were normal and no renal calculi were seen. Computed Tomography (CT) scan showed a well-defined hypodense lesion of size 5x4.5x5.1 cm arising from the right adrenal gland and showing peripheral enhancement on arterial phase with central necrosis [Table/Fig-1]. A non-functional pheochromocytoma was suspected. 131I Meta-iodobenzylguanidine (MIBG) scan showed no MIBG concentration in the whole body including the right adrenal. 18F-Fluorodeoxyglucose-Positron Emission Tomography/Computed Tomography (18FDG-PET/CT) scan showed no abnormal metabolic activity present in the whole body including the right adrenal. The patient underwent a laparoscopic right adrenalectomy. There were no intraoperative or postoperative fluctuations of blood pressure and the mass was sent for histopathological examination.
CT scan abdomen showing well-defined hypodense lesion arising from the right adrenal gland with enhancing peripheral rim in arterial phase and central necrosis (arrows).
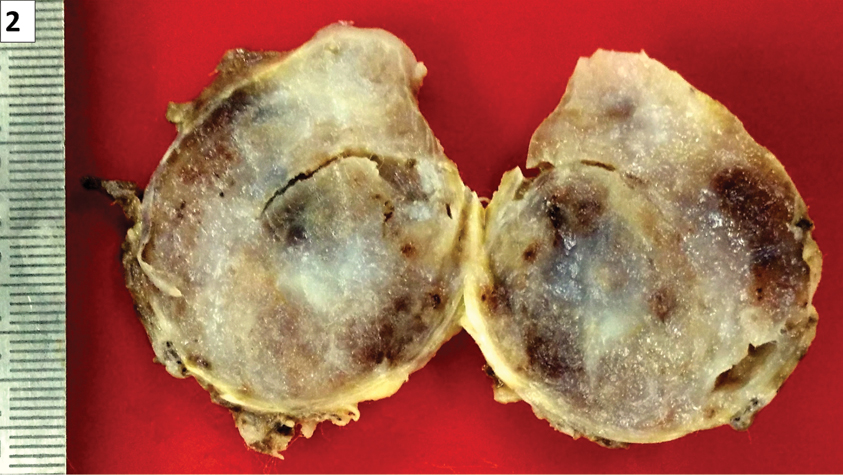
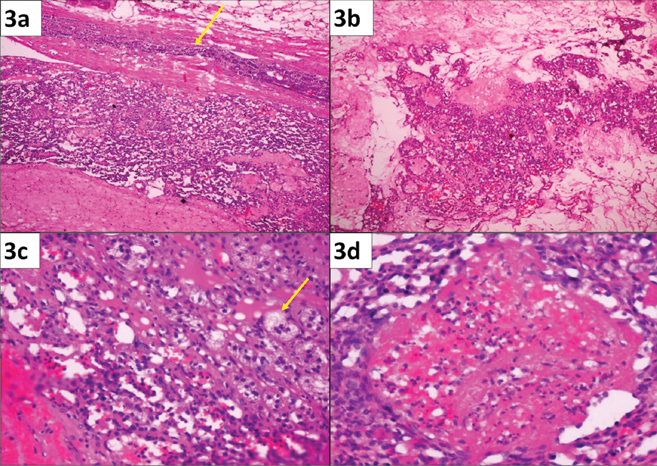
We received a right adrenalectomy specimen which was composed of a 6.5x5.5x2 cm, thinly encapsulated, a reddish-brown to grey-white, spongy mass, weighing 34 gm [Table/Fig-2]. Yellow-coloured normal adrenal tissue was seen at one focus in the periphery of the specimen. Microscopy revealed characteristic features of haemangioma. The tumour was composed of vascular channels of varying sizes from capillary sized to large vessel channels. The vascular spaces were lined by single layer of benign endothelial cells and smooth muscle was present within the vessel walls. These vascular channels showed red blood cells within their lumina and thrombi were seen within some of the vascular channels. Focal areas of haemorrhage and extensive areas of hyalinisation which corresponds to the greyish-white areas on gross were also seen [Table/Fig-3]. No other lesion or tumour was seen. Normal adrenal medulla and cortex were present at the periphery of the tumour [Table/Fig-3].
Right adrenalectomy specimen measuring 6.5x5.5x2 cm.
a) Low power magnification showing sheets of tumour composed of vascular channels varying from small to large. A strip of normal adrenal tissue is seen in the periphery (arrow) (H&E; 10X); b) Extensive areas of hyalinisation seen in the tumour which corresponds to whitish areas of the gross (H&E; 10X); c) High power magnification showing prominent vascular channels surrounded by nests of adrenocortical clear cells (arrow) (H&E; 40X); d) Thrombus is seen within the vascular lumina (H&E; 10X).
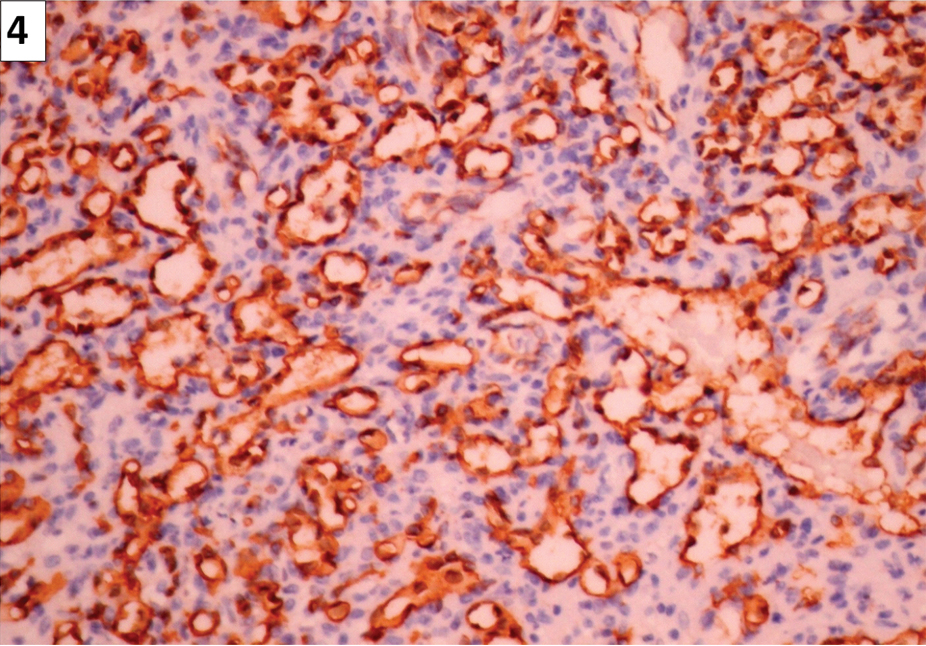
A panel of immunohistochemistry markers were done. CD34 was positive in the endothelial cells lining vascular channel [Table/Fig-4] and Smooth Muscle Antigen (SMA) was positive in the walls of the vessels. Pancytokeratin, synaptophysin and chromogranin were negative in the lesion. A final diagnosis of haemangioma of the adrenal gland was made. The patient was put on regular follow up and he is well now, seven months after the surgery with no recurrence or complications.
Vascular channels with endothelial cells strongly positive for CD34 (Immunohistochemistry, 40X).
Discussion
Haemangiomas of the adrenal gland are rare, non-functional benign tumours with less than 100 cases documented in literature [1]. They are difficult to diagnose preoperatively and are mistaken for other adrenal lesions like myelolipoma, ganglioneuroma and non-functional pheochromocytoma. Adrenal masses are an accidental finding in 1% to 5% of all abdominal CT scans performed and hence called incidentalomas [2]. Incidentalomas are usually seen between 50-70 years of age and are more common in females than males [3].
About 80% of adrenal incidentalomas are usually benign adenomas while, the remaining 20% include malignant or other benign lesions. Malignant lesions include adrenal cortical carcinoma, metastatic disease, and pheochromocytoma. Benign entities include cysts, myelolipoma, ganglioneuroma, and haemangioma [4].
In this case, a clinical diagnosis of non-functional pheochromocytoma was thought of, as the classical history of headache, palpitation and diaphoresis was absent. In addition, the blood pressure and plasma catecholamines were normal. CT findings also were indicative of a pheochromocytoma. As MIGB scans were negative, a histopathology diagnosis was sought after. The diagnosis of haemangioma was quite unexpected.
The exact aetiology of adrenal haemangioma is not known and most of the lesions are congenital which probably undergo enlargement and vascular ectasia over time [5]. Our case of adrenal haemangioma was probably not in existence six months prior to diagnosis, as ultrasonography examinations that were performed for the renal calculous disease would have detected it.
Incidentally detected adrenal haemangiomas are asymptomatic and non-functional. Some haemangiomas can bleed and undergo spontaneous rupture resulting in abdominal bleed and hypovolemic shock. Rarely, haemangiomas may show symptoms of hypercortisolism and hyperaldosteronism which are not caused by the haemangiomas as such, but because of enhanced entry of "active endocrine metabolites" into the abundant vascular channels [1,5,6].
Ultrasonography shows non specific findings of a mass with variable size and echotexture. On CT, adrenal haemangiomas appears as well-defined, heterogeneous mass with calcifications and various enhancement patterns [7]. On T2-weighted Magnetic Resonance Imagining (MRI), these lesions are hyperintense due to the vast amount of blood into the vascular spaces [8].
Histopathological examination is diagnostic and immunohistochemistry is not required for diagnosis. The spongy appearance of haemangioma on gross is due to vascular spaces. Microscopy shows vascular channels of varying calibre lined by endothelium and containing red blood cells. In our case, many of the vascular lumen showed thrombi and haemorrhages. Calcification of these haemorrhages with phlebolith formation can also be seen; however, we did not find any phleboliths. Immunohistochemistry with endothelial markers like CD34 and CD31 highlight the endothelium lined vascular spaces and confirm the diagnosis.
Treatment for haemangiomas depends on the size of the lesion. Smaller lesions are monitored with imaging follow up, while larger lesions would require surgical excision [6,7]. Surgery is necessary for larger incidentalomas exceeding 6 cm due the risk of malignancy which is reported to be 35%-98% and their tendency to rupture [9].
Conclusion
Adrenal haemangioma, though rare, and preoperative diagnosis difficult as it is often mistaken for common benign adrenal lesions. Histopathological examination clinches the diagnosis and helps misdiagnosis from other non-functional lesions of adrenal gland.
[1]. Johnson E, Sherif A, Bermudez F, Markow M, Eatrides J, Ivancsits D, Hemorrhage; unusual presentation of adrenal hemangiomaAm J Case Rep 2016 4(7):248-50. [Google Scholar]
[2]. Aljabri KS, Bokhari SA, Alkeraithi M, Adrenal hemangioma in a 19-year-old femaleAnn Saudi Med 2011 31(4):421-23.10.4103/0256-4947.7641121293064 [Google Scholar] [CrossRef] [PubMed]
[3]. Quildrian SD, Silberman EA, Vigovich FA, Porto EA, Giant cavernous hemangioma of the adrenal glandInt J Surg Case Rep 2013 4:219-21.10.1016/j.ijscr.2012.11.02223287064 [Google Scholar] [CrossRef] [PubMed]
[4]. Galea N, Noce V, Ciolina F, Liberali S, Francone M, Images in Clinical Urology: Giant Adrenal Cavernous Hemangioma: A Rare Abdominal MassUrology 2013 82(1):e3-e4.10.1016/j.urology.2013.04.00523806410 [Google Scholar] [CrossRef] [PubMed]
[5]. Ng AC, Loh HL, Shum CF, Yip SK, A case of adrenal cavernous hemangioma presenting with progressive enlargement and apparent hormonal hypersecretionEndocr Pract 2008 14(1):104-08.10.4158/EP.14.1.10418238749 [Google Scholar] [CrossRef] [PubMed]
[6]. Edwards JP, Stuart HC, Urbanski SJ, Pasieka JL, A rare cavernous hemangioma of the adrenal glandInt J Surg Case Rep 2014 5(2):52-55.10.1016/j.ijscr.2013.11.00324441435 [Google Scholar] [CrossRef] [PubMed]
[7]. Peng J, Lv X, Lin C, Zhou J, Zhang YM, Zhang YH, Computer tomography imaging findings of adrenal cavernous hemangiomas: a report of 10 casesActa Radiol 2016 57(1):115-21.10.1177/028418511456411025585847 [Google Scholar] [CrossRef] [PubMed]
[8]. Noh JJ, Choi SH, Hwang HK, Kang CM, Lee WJ, Adrenal cavernous hemangioma: a case report with review of the literatureJOP 2014 15(3):254-57. [Google Scholar]
[9]. Arkadopoulos N, Kyriazi M, Yiallourou AI, Stafyla VK, Theodosopoulos T, Dafnios N, A rare coexistence of adrenal cavernous hemangioma with extramedullar hemopoietic tissue: A case report and brief review of the literatureWorld J Surg Oncol 2009 7:1310.1186/1477-7819-7-1319193247 [Google Scholar] [CrossRef] [PubMed]